Abstract
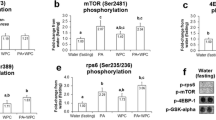
In older adults, chronic oxidative and inflammatory stresses are associated with an impaired increase in skeletal muscle protein synthesis after acute anabolic stimuli. Conjugated linoleic acid (CLA) and Protandim have been shown to activate nuclear factor erythroid-derived 2-like 2 (Nrf2), a transcription factor for the antioxidant response element and anti-inflammatory pathways. This study tested the hypothesis that compared to a placebo control (CON), CLA and Protandim would increase skeletal muscle subcellular protein (myofibrillar, mitochondrial, cytoplasmic) and DNA synthesis in older adults after 6 weeks of milk protein feeding. CLA decreased oxidative stress and skeletal muscle oxidative damage with a trend to increase messenger RNA (mRNA) expression of a Nrf2 target, NAD(P)H dehydrogenase quinone 1 (NQO1). However, CLA did not influence other Nrf2 targets (heme oxygenase-1 (HO-1), glutathione peroxidase 1 (Gpx1)) or protein or DNA synthesis. Conversely, Protandim increased HO-1 protein content but not the mRNA expression of downstream Nrf2 targets, oxidative stress, or skeletal muscle oxidative damage. Rates of myofibrillar protein synthesis were maintained despite lower mitochondrial and cytoplasmic protein syntheses after Protandim versus CON. Similarly, DNA synthesis was non-significantly lower after Protandim compared to CON. After Protandim, the ratio of protein to DNA synthesis tended to be greater in the myofibrillar fraction and maintained in the mitochondrial and cytoplasmic fractions, emphasizing the importance of measuring both protein and DNA synthesis to gain insight into proteostasis. Overall, these data suggest that Protandim may enhance proteostatic mechanisms of skeletal muscle contractile proteins after 6 weeks of milk protein feeding in older adults.




Similar content being viewed by others
References
Balage M, Averous J, Rémond D et al (2010) Presence of low-grade inflammation impaired postprandial stimulation of muscle protein synthesis in old rats. J Nutr Biochem 21:325–331. doi:10.1016/j.jnutbio.2009.01.005
Bergamo P, Maurano F, Rossi M (2007) Phase 2 enzyme induction by conjugated linoleic acid improves lupus-associated oxidative stress. Free Radic Biol Med 43:71–79. doi:10.1016/j.freeradbiomed.2007.03.023
Bergamo P, Gogliettino M, Palmieri G et al (2011) Conjugated linoleic acid protects against gliadin-induced depletion of intestinal defenses. Mol Nutr Food Res 55(Suppl 2):S248–S256. doi:10.1002/mnfr.201100295
Bruns DR, Drake JC, Biela LM et al (2015) Nrf2 signaling and the slowed aging phenotype: evidence from long-lived models. Oxidative Med Cell Longev 2015:732596. doi:10.1155/2015/732596
Busch R, Kim Y-K, Neese RA et al (2006) Measurement of protein turnover rates by heavy water labeling of nonessential amino acids. Biochim Biophys Acta 1760:730–744. doi:10.1016/j.bbagen.2005.12.023
Busch R, Neese RA, Awada M et al (2007) Measurement of cell proliferation by heavy water labeling. Nat Protoc 2:3045–3057. doi:10.1038/nprot.2007.420
Coen PM, Jubrias SA, Distefano G et al (2013) Skeletal muscle mitochondrial energetics are associated with maximal aerobic capacity and walking speed in older adults. J Gerontol A Biol Sci Med Sci 68:447–455. doi:10.1093/gerona/gls196
Crilly MJ, Tryon LD, Erlich AT, Hood DA (2016) The role of Nrf2 in skeletal muscle contractile and mitochondrial function. J Appl Physiol (1985) jap.00042.2016. doi: 10.1152/japplphysiol.00042.2016
Dhiman TR, Anand GR, Satter LD, Pariza MW (1999) Conjugated linoleic acid content of milk from cows fed different diets. J Dairy Sci 82:2146–2156. doi:10.3168/jds.S0022-0302(99)75458-5
Dickinson JM, Volpi E, Rasmussen BB (2013) Exercise and nutrition to target protein synthesis impairments in aging skeletal muscle. Exerc Sport Sci Rev 41:216–223. doi:10.1097/JES.0b013e3182a4e699
Donovan EL, McCord JM, Reuland DJ et al (2012) Phytochemical activation of Nrf2 protects human coronary artery endothelial cells against an oxidative challenge. Oxidative Med Cell Longev 2012:132931. doi:10.1155/2012/132931
Doyle G, Furey S, Berlin R et al (1999) Gastrointestinal safety and tolerance of ibuprofen at maximum over-the-counter dose. Aliment Pharmacol Ther 13:897–906
Drake JC, Peelor FF, Biela LM et al (2013) Assessment of mitochondrial biogenesis and mTORC1 signaling during chronic rapamycin feeding in male and female mice. J Gerontol A Biol Sci Med Sci 68:1493–1501. doi:10.1093/gerona/glt047
Drake JC, Bruns DR, Peelor FF et al (2014) Long-lived crowded-litter mice have an age-dependent increase in protein synthesis to DNA synthesis ratio and mTORC1 substrate phosphorylation. Am J Physiol - Endocrinol Metab 307:E813–E821. doi:10.1152/ajpendo.00256.2014
Drake JC, Bruns DR, Peelor FF et al (2015) Long-lived Snell dwarf mice display increased proteostatic mechanisms that are not dependent on decreased mTORC1 activity. Aging Cell 14:474–482. doi:10.1111/acel.12329
Howard C, Ferrucci L, Sun K et al (2007) Oxidative protein damage is associated with poor grip strength among older women living in the community. J Appl Physiol Bethesda Md 1985 103:17–20. doi:10.1152/japplphysiol.00133.2007
Hybertson BM, Gao B, Bose SK, McCord JM (2011) Oxidative stress in health and disease: the therapeutic potential of Nrf2 activation. Mol Asp Med 32:234–246. doi:10.1016/j.mam.2011.10.006
Kang KW, Lee SJ, Park JW, Kim SG (2002) Phosphatidylinositol 3-kinase regulates nuclear translocation of NF-E2-related factor 2 through actin rearrangement in response to oxidative stress. Mol Pharmacol 62:1001–1010
Kirkland JL (2016) Translating the science of aging into therapeutic interventions. Cold Spring Harb Perspect Med 6:a025908. doi:10.1101/cshperspect.a025908
Knekt P, Ritz J, Pereira MA et al (2004) Antioxidant vitamins and coronary heart disease risk: a pooled analysis of 9 cohorts. Am J Clin Nutr 80:1508–1520
Konopka AR, Asante A, Lanza IR et al (2015) Defects in mitochondrial efficiency and H2O2 emissions in obese women are restored to a lean phenotype with aerobic exercise training. Diabetes. doi:10.2337/db14-1701
Leiser SF, Miller RA (2010) Nrf2 signaling, a mechanism for cellular stress resistance in long-lived mice. Mol Cell Biol 30:871–884. doi:10.1128/MCB.01145-09
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods San Diego Calif 25:402–408. doi:10.1006/meth.2001.1262
Marzani B, Balage M, Vénien A et al (2008) Antioxidant supplementation restores defective leucine stimulation of protein synthesis in skeletal muscle from old rats. J Nutr 138:2205–2211. doi:10.3945/jn.108.094029
Miller BF, Robinson MM, Bruss MD et al (2012) A comprehensive assessment of mitochondrial protein synthesis and cellular proliferation with age and caloric restriction. Aging Cell 11:150–161. doi:10.1111/j.1474-9726.2011.00769.x
Miller BF, Robinson MM, Reuland DJ et al (2013) Calorie restriction does not increase short-term or long-term protein synthesis. J Gerontol A Biol Sci Med Sci 68:530–538. doi:10.1093/gerona/gls219
Miller BF, Drake JC, Naylor B et al (2014) The measurement of protein synthesis for assessing proteostasis in studies of slowed aging. Ageing Res Rev 18:106–111. doi:10.1016/j.arr.2014.09.005
Minor BD, Heusinger DE, Melanson EL et al (2012) Energy balance changes the anabolic effect of postexercise feeding in older individuals. J Gerontol A Biol Sci Med Sci 67:1161–1169. doi:10.1093/gerona/gls080
Neese RA, Misell LM, Turner S et al (2002) Measurement in vivo of proliferation rates of slow turnover cells by 2H2O labeling of the deoxyribose moiety of DNA. Proc Natl Acad Sci U S A 99:15345–15350. doi:10.1073/pnas.232551499
Nelson SK, Bose SK, Grunwald GK et al (2006) The induction of human superoxide dismutase and catalase in vivo: a fundamentally new approach to antioxidant therapy. Free Radic Biol Med 40:341–347. doi:10.1016/j.freeradbiomed.2005.08.043
Nightingale AK, Crilley JG, Pegge NC et al (2007) Chronic oral ascorbic acid therapy worsens skeletal muscle metabolism in patients with chronic heart failure. Eur J Heart Fail 9:287–291. doi:10.1016/j.ejheart.2006.06.006
Nuernberg K, Dannenberger D, Ender K, Nuernberg G (2007) Comparison of different methylation methods for the analysis of conjugated linoleic acid isomers by silver ion HPLC in beef lipids. J Agric Food Chem 55:598–602. doi:10.1021/jf061865k
Ochala J, Frontera WR, Dorer DJ et al (2007) Single skeletal muscle fiber elastic and contractile characteristics in young and older men. J Gerontol A Biol Sci Med Sci 62:375–381
Orgel LE (1963) The maintenance of the accuracy of protein synthesis and its relevance to ageing. Proc Natl Acad Sci U S A 49:517–521
Reuland DJ, Khademi S, Castle CJ et al (2013) Upregulation of phase II enzymes through phytochemical activation of Nrf2 protects cardiomyocytes against oxidant stress. Free Radic Biol Med 56:102–111. doi:10.1016/j.freeradbiomed.2012.11.016
Rieu I, Magne H, Savary-Auzeloux I et al (2009) Reduction of low grade inflammation restores blunting of postprandial muscle anabolism and limits sarcopenia in old rats. J Physiol 587:5483–5492. doi:10.1113/jphysiol.2009.178319
Robinson MM, Richards JC, Hickey MS et al (2010) Acute {beta}-adrenergic stimulation does not alter mitochondrial protein synthesis or markers of mitochondrial biogenesis in adult men. Am J Physiol Regul Integr Comp Physiol 298:R25–R33. doi:10.1152/ajpregu.00524.2009
Robinson MM, Turner SM, Hellerstein MK et al (2011) Long-term synthesis rates of skeletal muscle DNA and protein are higher during aerobic training in older humans than in sedentary young subjects but are not altered by protein supplementation. FASEB J 25:3240–3249. doi:10.1096/fj.11-186437
Safdar A, deBeer J, Tarnopolsky MA (2010) Dysfunctional Nrf2-Keap1 redox signaling in skeletal muscle of the sedentary old. Free Radic Biol Med 49:1487–1493. doi:10.1016/j.freeradbiomed.2010.08.010
Santanasto AJ, Glynn NW, Jubrias SA et al (2015) Skeletal muscle mitochondrial function and fatigability in older adults. J Gerontol A Biol Sci Med Sci 70:1379–1385. doi:10.1093/gerona/glu134
Scalzo RL, Peltonen GL, Binns SE et al (2014) Greater muscle protein synthesis and mitochondrial biogenesis in males compared with females during sprint interval training. FASEB J Off Publ Fed Am Soc Exp Biol 28:2705–2714. doi:10.1096/fj.13-246595
Schaap LA, Pluijm SMF, Deeg DJH et al (2009) Higher inflammatory marker levels in older persons: associations with 5-year change in muscle mass and muscle strength. J Gerontol A Biol Sci Med Sci 64:1183–1189. doi:10.1093/gerona/glp097
Schwanhäusser B, Busse D, Li N et al (2011) Global quantification of mammalian gene expression control. Nature 473:337–342. doi:10.1038/nature10098
Semba RD, Ferrucci L, Sun K et al (2007a) Oxidative stress is associated with greater mortality in older women living in the community. J Am Geriatr Soc 55:1421–1425. doi:10.1111/j.1532-5415.2007.01308.x
Semba RD, Ferrucci L, Sun K et al (2007b) Oxidative stress and severe walking disability among older women. Am J Med 120:1084–1089. doi:10.1016/j.amjmed.2007.07.028
Shenton D, Smirnova JB, Selley JN et al (2006) Global translational responses to oxidative stress impact upon multiple levels of protein synthesis. J Biol Chem 281:29011–29021. doi:10.1074/jbc.M601545200
Steinbaugh MJ, Sun LY, Bartke A, Miller RA (2012) Activation of genes involved in xenobiotic metabolism is a shared signature of mouse models with extended lifespan. Am J Physiol Endocrinol Metab 303:E488–E495. doi:10.1152/ajpendo.00110.2012
Strong R, Miller RA, Antebi A et al (2016) Longer lifespan in male mice treated with a weakly estrogenic agonist, an antioxidant, an α-glucosidase inhibitor or a Nrf2-inducer. Aging Cell. doi:10.1111/acel.12496
Velmurugan K, Alam J, McCord JM, Pugazhenthi S (2009) Synergistic induction of heme oxygenase-1 by the components of the antioxidant supplement Protandim. Free Radic Biol Med 46:430–440. doi:10.1016/j.freeradbiomed.2008.10.050
Yu R, Lei W, Mandlekar S et al (1999) Role of a mitogen-activated protein kinase pathway in the induction of phase II detoxifying enzymes by chemicals. J Biol Chem 274:27545–27552
Zhang L, Kimball SR, Jefferson LS, Shenberger JS (2009) Hydrogen peroxide impairs insulin-stimulated assembly of mTORC1. Free Radic Biol Med 46:1500–1509. doi:10.1016/j.freeradbiomed.2009.03.001
Acknowledgements
This work was supported by the Dairy Research Institute and LifeVantage, Inc. (to KLH and BFM). The authors would like to thank the participants for their time and commitment to this study. We are grateful for the assistance of Gary Luckasen, M.D., and Jon Matthews, M.D., and their associates for providing medical oversight. We would also like to acknowledge the technical assistance provided by Gaia Bublitz, Kim Cox-York, PhD., and the Metabolomics and Proteomics Facility at Colorado State University. Analysis for C-reactive protein was performed at the University of Colorado Hospital Clinical and Translational Research Centers, which is a service within the Colorado Clinical and Translational Sciences Institute (CCTSI), supported in part by NIH/NCATS Colorado CTSI Grant Number UL1 TR001082.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the Institutional Review Board at Colorado State University (13-4191H). Prior to beginning participation, each participant was informed of the study design, potential risk, and benefits and provided written consent.
Additional information
Karyn L. Hamilton and Benjamin F. Miller are co-senior authors.
Electronic supplementary material
Online Resource 1
Plasma concentration of C18:2 isomers (A) and cis-9, trans-11 and trans-10, cis-12 isomers (B). Cis-9, trans-11 and trans-10, cis-12 isomers co-eluted at the same retention time. Therefore, the overlapping CLA isomers were calculated using both standard curves and an average was taken. Undetectable levels were observed in CON and pre CLA. Therefore, individual data are presented. Detectable levels were observed in each participant post CLA. (PDF 28 kb)
Online Resource 2
Sexual dimorphism in the ratio of myofibrillar protein to DNA synthesis after 6 weeks of milk protein feeding with control (CON; n=4 Men, n=8 Women) and Protandim (n=5 men, n=10 women). Data presented as mean ± SEM. *P<0.05 vs. Protandim women; P=0.06 vs. CON Men. (PDF 35 kb)
About this article
Cite this article
Konopka, A.R., Laurin, J.L., Musci, R.V. et al. Influence of Nrf2 activators on subcellular skeletal muscle protein and DNA synthesis rates after 6 weeks of milk protein feeding in older adults. GeroScience 39, 175–186 (2017). https://doi.org/10.1007/s11357-017-9968-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-017-9968-8