Abstract
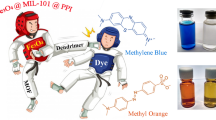
The development of effective and practical adsorbents for eliminating pollutants still remains a significant challenge. Herein, we synthesized a novel magnetically separable composite, Co0.6Fe2.4O4/MIL-101-NH2, through the in-situ growth of MIL-101-NH2 on magnetic nanoparticles, designed specifically for the removal of Congo red (CR) from aqueous solutions. MIL-101-NH2 possessed high BET surface area (240.485 m2•g−1) and facile magnetic separation function and can be swiftly separated (within 30 s) through an external magnetic field post-adsorption. The investigation systematically explored the influence of crucial parameters, including adsorbent dosage, pH, adsorption duration, temperature, and the presence of interfering ions, on CR adsorption performance. Findings indicate that CR adsorption adheres to the pseudo-second-order (PSO) kinetic model and the Langmuir isotherm model. Thermodynamic analysis reveals the spontaneity, endothermic nature, and orderly progression of the adsorption process. Remarkably, the adsorbent with 0.1 g•L−1 boasts an impressive maximum adsorption capacity of 1756.19 mg•g−1 for CR at 298.15 K, establishing its competitive advantage. The reuse of the adsorbent over 5 cycles remains 78% of the initial adsorption. The CR adsorption mechanisms were elucidated, emphasizing the roles of π-π interactions, electrostatic forces, hydrogen bonding, and metal coordination. Comparison with other dyes, such as methylene blue (MB) and methyl orange (MO), and exploration of adsorption performance in binary dye systems, demonstrates the superior capacity and selectivity of this adsorbent for CR. In conclusion, our magnetically separable metal–organic framework (MOF)based composite presents a versatile and effective solution for CR removal, with promising applications in water treatment and environmental remediation.
Graphical Abstract












Similar content being viewed by others
Data availability
Data available on request from the authors. The data that support the findings of this study are available from the corresponding author, [Li Houbin], upon reasonable request.
References
Abujazar MSS, Karaagac SU, Abu Amr SS, Alazaiza MYD, Bashir MJK (2022) Recent advancement in the application of hybrid coagulants in coagulation-flocculation of wastewater: a review. J Clean Prod 345:13. https://doi.org/10.1016/j.jclepro.2022.131133
Adebayo MA, Jabar JM, Amoko JS, Openiyi EO, Shodiya OO (2022) Coconut husk-raw clay-Fe composite: preparation, characteristics and mechanisms of Congo red adsorption. Sci Rep 12:12. https://doi.org/10.1038/s41598-022-18763-y
Al Sharabati M, Sabouni R (2020) Selective removal of dual dyes from aqueous solutions using a metal organic framework (MIL-53(Al)). Polyhedron 190:12. https://doi.org/10.1016/j.poly.2020.114762
Aly Z, Graulet A, Scales N, Hanley T (2014) Removal of aluminium from aqueous solutions using PAN-based adsorbents: characterisation, kinetics, equilibrium and thermodynamic studies. Environ Environ Sci Pollut r 21:3972–3986. https://doi.org/10.1007/s11356-013-2305-6
Banerjee S, Gautam RK, Gautam PK, Jaiswal A, Chattopadhyaya MC (2016) Recent trends and advancement in nanotechnology for water and wastewater treatment: nanotechnological approach for water purification. in: Advanced Research on Nanotechnology for Civil Engineering Applications, IGI Global, 208–252. https://doi.org/10.4018/978-1-5225-0344-6.ch007
Benmaamar Z, Boutoumi H, Hamitouche H, Benmaamar H, Benmaamar A, Aggoun A (2019) Simulation of adsorption kinetics of malachite green onto activated carbon. Port Electrochim Acta 37:93–104. https://doi.org/10.4152/pea.201902093
Biesinger MC, Payne BP, Grosvenor AP, Lau LWM, Gerson AR, Smart RS (2011) Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and Ni. Appl Surf Sci 257:2717–2730. https://doi.org/10.1016/j.apsusc.2010.10.051
Brown JP (1981) Reduction of polymeric azo and nitro dyes by intestinal bacteria. Appl Environ Microb 41:1283–1286. https://doi.org/10.1128/AEM.41.5.1283-1286.1981
Chen D, Yu H, Pan M, Pan B (2022) Hydrogen bonding-orientated selectivity of phosphate adsorption by imine-functionalized adsorbent. Chem Eng J 433:133690. https://doi.org/10.1016/j.cej.2021.133690
Cheng L, Huang R, Cao Q, Liu N, Li P, Sun M, Qin H, Wu L (2023) Magnetic metal-organic frameworks as adsorbents for the detection of azo pigments in food matrices. Food Chem 402:134134. https://doi.org/10.1016/j.foodchem.2022.134134
Christovam EM, Franco JH, Zocolo GJ, Almeida AMF, Marcelino MY, de Oliveira DP, Zanoni MVB (2022) Stability of acid black 210 dye in tannery industry effluent in aqueous solution is limited and generates harmful subproducts. Front Environ Sci 10:13. https://doi.org/10.3389/fenvs.2022.750647
Cross SN, Fataftah M, Jonderian A, McCalla E, Blum AS (2023) Preparation and characterization of ultra-small, monodisperse CoxFe3-xO4 nanoparticles. J Magn Magn Mater 567:8. https://doi.org/10.1016/j.jmmm.2022.170345
Csillag K, Emri T, Rangel DEN, Pocsi I (2023) pH-dependent effect of Congo red on the growth of Aspergillus nidulans and Aspergillus niger. Fungal Biol-UK 127:1180–1186. https://doi.org/10.1016/j.funbio.2022.05.006
Ding XX, Gutierrez L, Croue JP, Li MR, Wang LJ, Wang YR (2020) Hydroxyl and sulfate radical-based oxidation of RhB dye in UV/H2O2 and UV/persulfate systems: kinetics, mechanisms, and comparison. Chemosphere 253:11. https://doi.org/10.1016/j.chemosphere.2020.126655
Duo HX, Tang H, Ma JL, Lu XF, Wang LC, Liang XJ (2019) Iron-based metal-organic framework as an effective sorbent for the rapid and efficient removal of illegal dyes. New J Chem 43:15351–15358. https://doi.org/10.1039/c9nj03370j
Eltaweil AS, Elshishini HM, Ghatass ZF, Elsubruiti GM (2021) Ultra-high adsorption capacity and selective removal of Congo red over aminated graphene oxide modified Mn-doped UiO-66 MOF. Powder Technol 379:407–416. https://doi.org/10.1016/j.powtec.2020.10.084
Esrafili A, Ghambarian M, Yousefi M (2022) Electrospun zeolitic imidazolate framework-8/poly (lactic acid) nanofibers for pipette-tip micro-solid phase extraction of carbamate insecticides from environmental samples. Arab J Chem 15:10. https://doi.org/10.1016/j.arabjc.2022.104124
Ghazvini MF, Vahedi M, Nobar SN, Sabouri F (2021) Investigation of the MOF adsorbents and the gas adsorptive separation mechanisms. J Environ Chem Eng 9:10. https://doi.org/10.1016/j.jece.2020.104790
Gilson MK (1995) Theory of electrostatic interactions in macromolecules. Curr Opin Struc Biol 5:216–223. https://doi.org/10.1016/0959-440X(95)80079-4
Gökmen V, Serpen A (2002) Equilibrium and kinetic studies on the adsorption of dark colored compounds from apple juice using adsorbent resin. J Food Eng 53:221–227. https://doi.org/10.1016/s0260-8774(01)00160-1
Guo XL, Kong LJ, Ruan Y, Diao ZH, Shih KM, Su MH, Hou LA, Chen DY (2020) Green and facile synthesis of cobalt-based metal-organic frameworks for the efficient removal of Congo red from aqueous solution. J Colloid Interf Sci 578:500–509. https://doi.org/10.1016/j.jcis.2020.05.126
He QQ, Zhao HJ, Teng ZD, Wang Y, Sun WL, Guo YL, Ji XA, Hu W, Lam SS, Li M (2023) Efficient recovery of phosphate by Fe3O4/La-MOF: an insight of adsorption performance and mechanism from electrochemical properties. Sep Purif Technol 314:12. https://doi.org/10.1016/j.seppur.2023.123529
Hoang LP, Van HT, Nguyen LH, Mac DH, Vu TT, Ha LT, Nguyen XC (2019) Removal of Cr(VI) from aqueous solution using magnetic modified biochar derived from raw corncob. New J Chem 43:18663–18672. https://doi.org/10.1039/c9nj02661d
Hu N, Yu JZ, Hou LR, Shi CR, Li K, Hang FX, Xie CF (2022) Amine-functionalized MOF-derived carbon materials for efficient removal of Congo red dye from aqueous solutions: simulation and adsorption studies. Rsc Adv 13:1–13. https://doi.org/10.1039/d2ra06513d
Huang Q, Yang Y, Qian JJ (2023) Structure-directed growth and morphology of multifunctional metal-organic frameworks. Coord Chem Rev 484:38. https://doi.org/10.1016/j.ccr.2023.215101
Imessaoudene A, Cheikh S, Hadadi A, Hamri N, Jean-Claude B, Amrane A, Tahraoui H, Manseri A, Mouni L (2023) Adsorption performance of zeolite for the removal of Congo red dye: factorial design experiments, kinetic, and equilibrium studies. Separations 10(1):57. https://doi.org/10.3390/separations10010057
Islam M, Mostafa M (2018) Textile dyeing effluents and environment concerns-a review. J Environ Sci Nat Res 11:131–144. https://doi.org/10.3329/jesnr.v11i1-2.43380
Jaafar J, Ismail AF, Mustafa A (2007) Physicochemical study of poly(ether ether ketone) electrolyte membranes sulfonated with mixtures of fuming sulfuric acid and sulfuric acid for direct methanol fuel cell application. Mat Sci Eng A-Struct 460:475–484. https://doi.org/10.1016/j.msea.2007.02.095
Jeong C, Ansari MZ, Anwer AH, Kim SH, Nasar A, Shoeb M, Mashkoor F (2023) A review on metal-organic frameworks for the removal of hazardous environmental contaminants. Sep Purif Technol 305:40. https://doi.org/10.1016/j.seppur.2022.122416
Jiang QS, Zhao CY, Han ZL, Yang GX, Qu N, Sun LZ, Li WP, Wang MY, Cheng ZQ (2022) Amino-functionalized polyacrylonitrile/bentonite composite membranes for effective decontamination of Pb2+ and Congo Red. Sep Purif Technol 287:120606. https://doi.org/10.1016/j.seppur.2022.120606
Kazim M, Siegler MA, Lectka T (2020) Close Amide NH·F Hydrogen bonding interactions in 1, 8-disubstituted naphthalenes. J Org Chem 85:6195–6200. https://doi.org/10.1021/acs.joc.0c00553
Khan AH, Aziz HA, Khan NA, Hasan MA, Ahmed S, Farooqi IH, Dhingra A, Vambol V, Changani F, Yousefi M, Islam S, Mozaffari N, Mahtab MS (2022) Impact, disease outbreak and the eco-hazards associated with pharmaceutical residues: a Critical review. Int J Environ Sci Technol 19:677–688. https://doi.org/10.1007/s13762-021-03158-9
Kim Y, Zhao XH (2022) Magnetic soft materials and robots. Chem Rev 122:5317–5364. https://doi.org/10.1021/acs.chemrev.1c00481
Lee H, Kim DI, Kim Y, Jang A (2023) Efficient one-pot synthesis of magnetic MIL-100(Fe) using nitric acid without additional Fe ion addition and adsorption behavior of charged organic compounds. Chemosphere 314:7. https://doi.org/10.1016/j.chemosphere.2022.137696
Lei C, Wang C, Chen W, He M, Huang B (2020) Polyaniline@ magnetic chitosan nanomaterials for highly efficient simultaneous adsorption and in-situ chemical reduction of hexavalent chromium: Removal efficacy and mechanisms. Sci Total Environ 733:139316. https://doi.org/10.1016/j.scitotenv.2020.139316
Li X, Tjiptoputro AK, Ding J, Xue JM, Zhu Y (2017) Pd-Ce nanoparticles supported on functional Fe-MIL-101-NH2: an efficient catalyst for selective glycerol oxidation. Catal Today 279:77–83. https://doi.org/10.1016/j.cattod.2016.03.044
Li YS, Wang YT, He LY, Meng LZ, Lu HJ, Li XL (2020) Preparation of poly(4-vinylpyridine)-functionalized magnetic Al-MOF for the removal of naproxen from aqueous solution. J Hazard Mater 383:12. https://doi.org/10.1016/j.jhazinat.2019.121144
Lian L-L, Jiang X-H, Deng Y-H, Lou D-W (2019) Adsorption of microcystin-LR in water with metal-organic framework MIL-101(Fe). J Instrum Anal 38:5. https://doi.org/10.3969/j.issn.1004-4957.2019.09.007
Lim J, Lee S, Sharma A, Seong J, Baek SB, Lah MS (2022) Ligand functionalization of defect-engineered Ni-MOF-74. Rsc Adv 12:31451–31455. https://doi.org/10.1039/d2ra06587h
Liu D, Gu W, Zhou L, Lei J, Wang L, Zhang J, Liu Y (2023) From biochar to functions: Lignin induced formation of Fe3C in carbon/Fe composites for efficient adsorption of tetracycline from wastewater. Sep Purif Technol. 304. https://doi.org/10.1016/j.seppur.2022.122217
Liu RJ, Xie YD, Cui KF, Xie J, Zhang YX, Huang YP (2022a) Adsorption behavior and adsorption mechanism of glyphosate in water by amino-MIL-101(Fe). J Phys Chem Solids 161:10. https://doi.org/10.1016/j.jpcs.2021.110403
Liu Y, Qiu GY, Liu YF, Niu YZ, Qu RJ, Ji CN, Wang Y, Zhang Y, Sun CM (2022b) Fabrication of CoFe-MOF materials by different methods and adsorption properties for Congo red. J Mol Liq 360:10. https://doi.org/10.1016/j.molliq.2022.119405
Lu FF, Dong AQ, Ding GJ, Xu K, Li JM, You LJ (2019) Magnetic porous polymer composite for high performance adsorption of acid red 18 based on melamine resin and chitosan. J Mol Liq 294:12. https://doi.org/10.1016/j.molliq.2019.111515
Lu ZH, Tian Q, Zhou DD, Chen M, Cao YW, Zhuang LY, Liu X, Yang ZH, Senosy IA (2022) Magnetic MXene based metal organic frameworks composites: synthesis, characterization and application. J Environ Chem Eng 10:10. https://doi.org/10.1016/j.jece.2022.108037
Mahmoudian MH, Azari A, Jahantigh A, Sarkhosh M, Yousefi M, Razavinasab SA, Afsharizadeh M, Shahraji FM, Pasandi AP, Zeidabadi A, Bardsiri TI, Ghasemian M (2023) Statistical modeling and optimization of dexamethasone adsorption from aqueous solution by Fe3O4@NH2-MIL88B nanorods: isotherm, kinetics, and thermodynamic. Environ Res 236:16. https://doi.org/10.1016/j.envres.2023.116773
Marnani NN, Shahbazi A (2019) A novel environmental-friendly nanobiocomposite synthesis by EDTA and chitosan functionalized magnetic graphene oxide for high removal of rhodamine B: adsorption mechanism and separation property. Chemosphere 218:715–725. https://doi.org/10.1016/j.chemosphere.2018.11.109
Mazloomi S, Yousefi M, Nourmoradi H, Shams M (2019) Evaluation of phosphate removal from aqueous solution using metal organic framework; isotherm, kinetic and thermodynamic study. J Environ Health Sci Eng 17:209–218. https://doi.org/10.1007/s40201-019-00341-6
Munagapati VS, Kim DS (2017) Equilibrium isotherms, kinetics, and thermodynamics studies for congo red adsorption using calcium alginate beads impregnated with nano-goethite. Ecotox Environ Safe 141:226–234. https://doi.org/10.1016/j.ecoenv.2017.03.036
Olusegun SJ, Mohallem NDS (2020) Comparative adsorption mechanism of doxycycline and Congo red using synthesized kaolinite supported CoFe2O4 nanoparticles. Environ Pollut 260:11. https://doi.org/10.1016/j.envpol.2020.114019
Pallach R, Keupp J, Terlinden K, Frentzel-Beyme L, Kloss M, Machalica A, Kotschy J, Vasa SK, Chater PA, Sternemann C, Wharmby MT, Linser R, Schmid R, Henke S (2021) Frustrated flexibility in metal-organic frameworks. Nat Commun 12:12. https://doi.org/10.1038/s41467-021-24188-4
Razavi L, Raissi H, Hashemzadeh H, Farzad F (2022) Strategy to improve Cu-BTC metal-organic frameworks performance in removal of rhodamine B: MD and WT-MtD simulations assessment. Npj Clean Water 5:8. https://doi.org/10.1038/s41545-022-00195-w
Sarkodie B, Amesimeku J, Frimpong C, Howard EK, Feng Q, Xu ZZ (2023) Photocatalytic degradation of dyes by novel electrospun nanofibers: a review. Chemosphere 313:16. https://doi.org/10.1016/j.chemosphere.2022.137654
Selvaraj V, Karthika TS, Mansiya C, Alagar M (2021) An over review on recently developed techniques, mechanisms and intermediate involved in the advanced azo dye degradation for industrial applications. J Mol Struct 1224:15. https://doi.org/10.1016/j.molstruc.2020.129195
Shi YT, Zou YT, Khan MS, Zhang MG, Yan J, Zheng XH, Wang WQ, Xie ZG (2023) Metal-organic framework-derived photoelectrochemical sensors: structural design and biosensing technology. J Mater Chem c 11:3692–3709. https://doi.org/10.1039/d2tc05338a
Song Y, He L, Zhang S, Liu X, Chen K, Jia Q, Zhang Z, Du M (2021) Novel impedimetric sensing strategy for detecting ochratoxin A based on NH2-MIL-101(Fe) metal-organic framework doped with cobalt phthalocyanine nanoparticles. Food Chem 351:129248. https://doi.org/10.1016/j.foodchem.2021.129248
Stockton W, Rubner M (1997) Molecular-level processing of conjugated polymers. 4. Layer-by-layer manipulation of polyaniline via hydrogen-bonding interactions. Macromolecules 30:2717–2725. https://doi.org/10.1021/ma9700486
Tan YM, Sun ZQ, Meng H, Han YD, Wu JB, Xu JL, Xu Y, Zhang X (2019) Efficient and selective removal of congo red by mesoporous amino -modified MIL-101(Cr) nanoadsorbents. Powder Technol 356:162–169. https://doi.org/10.1016/j.powtec.2019.08.017
Tee GT, Gok XY, Yong WF (2022) Adsorption of pollutants in wastewater via biosorbents, nanoparticles and magnetic biosorbents: a review. Environ Res 212:20. https://doi.org/10.1016/j.envres.2022.113248
Valadi FM, Ekramipooya A, Gholami MR (2020) Selective separation of Congo red from a mixture of anionic and cationic dyes using magnetic-MOF: experimental and DFT study. J Mol Liq 318:13. https://doi.org/10.1016/j.molliq.2020.114051
Vermohlen K, Lewandowski H, Narres HD, Schwuger MJ (2000) Adsorption of polyelectrolytes onto oxides - the influence of ionic strength, molar mass, and Ca2+ ions. Colloid Surface a 163:45–53. https://doi.org/10.1016/s0927-7757(99)00429-x
Wang C, Xiong C, He YL, Yang C, Li XT, Zheng JZ, Wang SX (2021a) Facile preparation of magnetic Zr-MOF for adsorption of Pb(II) and Cr(VI) from water: adsorption characteristics and mechanisms. Chem Eng J 415:11. https://doi.org/10.1016/j.cej.2021.128923
Wang HT, Yang L, Qin YH, Chen Z, Wang TL, Sun W, Wang CW (2023) Highly effective removal of methylene blue from wastewater by modified hydroxyl groups materials: adsorption performance and mechanisms. Colloid Surface a 656:12. https://doi.org/10.1016/j.colsurfa.2022.130290
Wang L, Wang AQ (2008) Adsorption properties of Congo red from aqueous solution onto surfactant-modified montmorillonite. J Hazard Mater 160:173–180. https://doi.org/10.1016/j.jhazmat.2008.02.104
Wang QZ, Zhao YJ, Shi Z, Sun XY, Bu T, Zhang CQ, Mao ZX, Li XH, Wang L (2021b) Magnetic amino-functionalized-MOF(M = Fe, Ti, Zr)@COFs with superior biocompatibility: performance and mechanism on adsorption of azo dyes in soft drinks. Chem Eng J 420:10. https://doi.org/10.1016/j.cej.2021.129955
Wang X, Ying JW, Mai YL, Zhan JJ, Chen JZ, Wen MT, Yu L (2019) MOF-derived metal oxide composite Mn2Co1Ox/CN for efficient formaldehyde oxidation at low temperature. Catal Sci Technol 9:5845–5854. https://doi.org/10.1039/c9cy01104h
Weber W, Morris J (1963) Kinetics of adsorption on carbon from solution. J SanitEng Div 89
Wu B, Lin XC, Ge L, Wu L, Xu TW (2013) A novel route for preparing highly proton conductive membrane materials with metal-organic frameworks. Chem Commun 49(143):145. https://doi.org/10.1039/c2cc37045j
Xie J, Chen W, Wu Y, Wu X, Zhao Q (2017) Highly efficient adsorption capacity of MIL-53 (Fe) metal organic framework material for Congo red. Ind Water Treatment 37:27–30
Xiong ZK, Zheng HL, Hu YD, Hu XB, Ding W, Ma JY, Li YS (2021) Selective adsorption of Congo red and Cu(II) from complex wastewater by core-shell structured magnetic carbon@zeolitic imidazolate frameworks-8 nanocomposites. Sep Purif Technol 277:18. https://doi.org/10.1016/j.seppur.2021.119053
Xu C, Yu WH, Zheng KX, Ling C, Yu ST, Jiang L (2020) Novel composite oxygen-containing resins with effective adsorption towards anilines: physical & chemical adsorption. J Chem Technol Biotechnol 95:2187–2194. https://doi.org/10.1002/jctb.6405
Yang DQ, Hennequin B, Sacher E (2006) XPS demonstration of pi-pi interaction between benzyl mercaptan and multiwalled carbon nanotubes and their use in the adhesion of Pt nanoparticles. Chem Mater 18:5033–5038. https://doi.org/10.1021/cm061256s
You LJ, Huang C, Lu FF, Wang A, Liu XC, Zhang QQ (2018) Facile synthesis of high performance porous magnetic chitosan - polyethylenimine polymer composite for Congo red removal. Int J Biol Macromol 107:1620–1628. https://doi.org/10.1016/j.ijbiomac.2017.10.025
Yu L, Xue WH, Cui L, Xing W, Cao XL, Li HY (2014) Use of hydroxypropyl-beta-cyclodextrin/polyethylene glycol 400, modified Fe3O4 nanoparticles for congo red removal. Int J Biol Macromol 64:233–239. https://doi.org/10.1016/j.ijbiomac.2013.12.009
Yuan Y, Shu J, Liu P, Zhang Y, Duan Y, Zhang J (2015) Study on π–π interaction in H-and J-aggregates of poly (3-hexylthiophene) nanowires by multiple techniques. J Phys Chem B 119:8446–8456. https://doi.org/10.1021/acs.jpcb.5b02805
Zeng SY, Duan SX, Tang RF, Li L, Liu CH, Sun DZ (2014) Magnetically separable Ni0.6Fe2.4O4 nanoparticles as an effective adsorbent for dye removal: synthesis and study on the kinetic and thermodynamic behaviors for dye adsorption. Chem Eng J 258:218–228. https://doi.org/10.1016/j.cej.2014.07.093
Zhang Y, Yin Y, Tong M, Sun M, Yang J (2020) Solvent synthesis of micro nano coordination polymer MIL-101 and performance research of cuperior adsorption of Congo red. Henan Chem Ind 37:18–21. https://doi.org/10.14173/j.cnki.hnhg.2020.01.005.
Zhao SQ, Chen D, Wei FH, Chen NN, Liang Z, Luo Y (2017) Removal of Congo red dye from aqueous solution with nickel-based metal-organic framework/graphene oxide composites prepared by ultrasonic wave-assisted ball milling. Ultrason Sonochem 39:845–852. https://doi.org/10.1016/j.ultsonch.2017.06.013
Zhao YZ, Zhu PZ, Pan LW, Xie YQ, Ng SW, Zhang KL (2022) Preparation and characterization of a newly constructed multifunctional Co(II)-organic framework: proton conduction and adsorption of Congo red in aqueous medium. CrystEngComm 24:3380–3393. https://doi.org/10.1039/d2ce00330a
Zheng X, Wang JL, Xue XL, Liu WX, Kong YD, Cheng R, Yuan DH (2018) Facile synthesis of Fe3O4@MOF-100(Fe) magnetic microspheres for the adsorption of diclofenac sodium in aqueous solution. Environ Sci Pollut Res 25:31705–31717. https://doi.org/10.1007/s11356-018-3134-4
Zheng ZJ, Xu K, Lu FF, Zhong BH, You LJ, Xiong WJ, Tang T, Wang SY (2023) Magnetic covalent organic framework for the adsorption of silver nanoparticles and recycled as surface-enhanced Raman substrate and high-efficiency catalysts for 4-nitrophenol degradation. Environ Sci Pollut Res 30:34636–34648. https://doi.org/10.1007/s11356-022-24720-z
Acknowledgements
The authors are grateful for constructive feedback from reviewers that helped improve an earlier version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (grant number: U1830127).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Conceptualization and investigation were completed by Zhenhong Zhang and Yuye Zhong. Material preparation, data collection, and analysis were performed by Zhenhong Zhang, Yuye Zhong, and Peng Sun. Review and editing were performed by Pingping Zhao, Houbin Li, and Xinghai Liu. The first draft of the manuscript was written by Zhenhong Zhang. Zhenhong Zhang and Yuye Zhong contributed equally to this work and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
There are no ethical issues involved in this study.
Consent for publication
Written informed consent for publication of this paper was obtained from the Wuhan University of Arts and Science and all authors.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• The magnetic adsorbent with embedded structure is compounded by in-situ growth.
• The Langmuir isotherm shows the high adsorption capacity of Co0.6Fe2.4O4/MIL-101-NH2 for Congo red (up to 1756.19 mg•g−1) at 298.15 K.
• The excellent separability and reusability of the adsorbent show its practicability and industrialization feasibility.
• Co0.6Fe2.4O4/MIL-101-NH2 has strong adsorption performance in complicated environments due to its selectivity for Congo red.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, Z., Zhong, Y., Sun, P. et al. Magnetically separable Co0.6Fe2.4O4/MIL-101-NH2 adsorbent for Congo red efficient removal. Environ Sci Pollut Res 31, 9764–9783 (2024). https://doi.org/10.1007/s11356-023-31796-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-31796-8