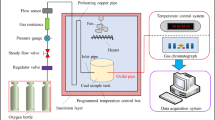
Abstract
The gases evolution during the low-temperature oxidation of coal is an essential parameter used to assess the state of coal oxidation and to estimate the gaseous pollutants. However, the current semi-quantitative method, which employs gas concentration as the measurement standard, is flawed. This paper presents a quantitative calculation method for gas products during coal oxidation. N2 is used as the tracer gas in the experiment, because nitrogen is an inert gas that will not participate in the reaction, and the amount of matter will not change in the reaction. According to the formula \({\textrm{M}}_i=\frac{X_i}{X_{N_2}}\times {V}_{N_2}\times {\rho}_i\), the corresponding mass flow rates of each gases component were calculated, and the gas yields during the reaction period were determined by comprehensive calculation. To this end, experiments were conducted on the low-temperature oxidation of coal using a flow reactor. After undergoing quantitative calculations, the main gas products’ mass flow rates, yields, and energies, including CO, CO2, CH4, C2H4, C2H6, C2H2, and C3H8 between 30 and 180 °C were obtained. The findings showed that CO2 > CO > CH was generated in all the coal samples. The amount of gases produced in the low-temperature oxidation of coal is proportional to the level of oxygen concentration. When the oxygen concentration ranges from 0 to 21%, the gaseous production of MTH coal ranges from 381.44 g/ton to 8562.80 g/ton. The results of gaseous energy calculations showed that the energy loss for low temperature oxidation of the four coal samples ranged from 4334.14~26,772.73 kJ/ton under air atmosphere. Energy loss is also significantly affected by the oxygen concentration, and the energy loss of MTH coal increases significantly from 520.52 kJ/ton at 0% oxygen concentration to 26,772.73 kJ/ton at 21% oxygen concentration, an increase of about 50 times. Significantly, this method not only reflects the real gas evolution during low-temperature oxidation of coal but also computes the gas emission and energy loss, which is crucial for studying the mechanism of coal spontaneous combustion and assessing gases pollutants.











Similar content being viewed by others
Data availability
The authors have the right to provide relevant data as they see fit.
Abbreviations
- MTH:
-
Mataihao
- XJ:
-
Xinji I
- ZJ:
-
Zhangjixi
- GH:
-
Gaohe
- M i :
-
Mass flow rate
- X i :
-
Molar fraction
- ρ i :
-
Respective gas density at standard temperature and pressure
- \({X}_{N_2}\) :
-
Molar fraction of N2
- \({V}_{N_2}\) :
-
Volume flow rate of N2
- Yi :
-
Yield of each gas
- t :
-
The reaction time
- T :
-
Reaction temperature
References
Bai Z, Deng J, Wang C, Hou Y, Zhang Y, Kang F, Ramakrishna S (2023) Study on the mechanism of lignite oxidation inhibition by antioxidant resveratrol. Energy 273:127235. https://doi.org/10.1016/j.energy.2023.127235
Beamish BB, Barakat MA, JDS G (2000) Adiabatic testing procedures for determining the self-heating propensity of coal and sample ageing effects. Thermochim Acta 362:79–87. https://doi.org/10.1016/S0040-6031(00)00588-8
Cao Q, Lu W, Li J, Burra KG, Gupta AK (2022) Energy recovery of expired pistachios from pyrolysis and CO2-assisted gasification. J Energy Resour Technol 145:12102. https://doi.org/10.1115/1.4054788
Cheng W, Hu X, Xie J, Zhao Y (2017) An intelligent gel designed to control the spontaneous combustion of coal: fire prevention and extinguishing properties. Fuel 210:826–835. https://doi.org/10.1016/j.fuel.2017.09.007
Deng J, Liang C, Chen W, Zhao J, Yao H (2020) Research on sensor correction method for monitoring in early warning system of coal spontaneous combustion IOP conference series. Earth Environ Sci 514:22058. https://doi.org/10.1088/1755-1315/514/2/022058
Deng J, Song JJ, Zhao JY, Zhang YN, Ren SJ, Shu CM (2021) Macrocharacteristics of gaseous indicator products and exothermicity during low-temperature oxidation of samples from different regions of the same coal seam from Huainan Anhui China. J Therm Anal Calorim 143:781–794. https://doi.org/10.1007/s10973-019-09239-9
Deng J, Zhao J, Zhang Y, Huang A, Liu X, Zhai X, Wang C (2016) Thermal analysis of spontaneous combustion behavior of partially oxidized coal. Process Saf Environ 104:218–224. https://doi.org/10.1016/j.psep.2016.09.007
Déparrois N, Singh P, Burra KG, Gupta AK (2019) Syngas production from co-pyrolysis and co-gasification of polystyrene and paper with CO2. Appl Energ 246:1–10. https://doi.org/10.1016/j.apenergy.2019.04.013
Dong X, Wen Z, Wang F, Meng Y (2019) Law of gas production during coal heating oxidation. Int J Min Sci Techno 29:617–620. https://doi.org/10.1016/j.ijmst.2019.06.011
Jia X, Wu J, Lian C, Rao J (2022) Assessment of coal spontaneous combustion index gas under different oxygen concentration environment: an experimental study. Environ Sci Pollut R 29:87257–87267. https://doi.org/10.1007/s11356-022-21920-5
Kim J, Lee Y, Ryu C, Park HY, Lim H (2015) Low-temperature reactivity of coals for evaluation of spontaneous combustion propensity. Korean J Chem Eng 32:1297–1304. https://doi.org/10.1007/s11814-014-0331-9
Li J, Burra KG, Wang Z, Liu X, Gupta AK (2021a) Acid and alkali pretreatment effects on CO2-assisted gasification of pinewood. J Energy Resour Technol 144:22306–22309. https://doi.org/10.1115/1.4051145
Li J, Burra KG, Wang Z, Liu X, Gupta AK (2021b) Effect of alkali and alkaline metals on gas formation behavior and kinetics during pyrolysis of pine wood. Fuel 290:120081. https://doi.org/10.1016/j.fuel.2020.120081
Li J, Burra KRG, Wang Z, Liu X, Gupta AK (2021c) Co-gasification of high-density polyethylene and pretreated pine wood. Appl Energ 285:116472. https://doi.org/10.1016/j.apenergy.2021.116472
Li S, Xu K, Xue G, Liu J, Xu Z (2022a) Prediction of coal spontaneous combustion temperature based on improved grey wolf optimizer algorithm and support vector regression. Fuel 324:124670. https://doi.org/10.1016/j.fuel.2022.124670
Li J, Burra KG, Wang Z, Liu X, Gupta AK (2022b) Syngas evolution and energy efficiency in CO2 assisted gasification of ion-exchanged pine wood. Fuel 317:123549. https://doi.org/10.1016/j.fuel.2022.123549
Li J, Li Z, Yang Y, Wang C (2018) Study on oxidation and gas release of active sites after low-temperature pyrolysis of coal. Fuel 233:237–246. https://doi.org/10.1016/j.fuel.2018.06.039
Li J, Lu W, Li J, Yang Y, Li Z (2023) Towards understanding of internal mechanism of coal reactivity enhancement after thermal decomposition at low temperature. Fuel 337:127118. https://doi.org/10.1016/j.fuel.2022.127118
Liu X, Burra KRG, Wang Z, Li J, Che D, Gupta AK (2021) Towards enhanced understanding of synergistic effects in co-pyrolysis of pinewood and polycarbonate. Appl Energ 289:116662. https://doi.org/10.1016/j.apenergy.2021.116662
Lu H, Li J, Lu W, Xu Z, Li J, He Q (2023) Variation laws of CO2/CO and influence of key active groups on it during low-temperature oxidation of coal. Fuel 339:127415. https://doi.org/10.1016/j.fuel.2023.127415
Lu W, Sun X, Gao L, Hu X, Song H, Kong B (2022) Study on the characteristics and mechanism of DL-malic acid in inhibiting spontaneous combustion of lignite and bituminous coal. Fuel 308:122012. https://doi.org/10.1016/j.fuel.2021.122012
Ma L, Zou L, Ren L, Chung Y, Zhang P, Shu C (2020) Prediction indices and limiting parameters of coal spontaneous combustion in the Huainan mining area in China. Fuel 264:116883. https://doi.org/10.1016/j.fuel.2019.116883
Qi X, Xin H, Wang D, Qi G (2013) A rapid method for determining the R70 self-heating rate of coal. Thermochim Acta 571:21–27. https://doi.org/10.1016/j.tca.2013.08.008
Raymond CJ, Farmer J, Dockery CR (2016) Thermogravimetric analysis of target inhibitors for the spontaneous self-heating of coal. Combust Sci Technol 188:1249–1261. https://doi.org/10.1080/00102202.2016.1177034
Shang S, Huang L, Ma S, Liu W (2022) Experimental study on the effect of air volume on coal oxidation gas at low temperature. Inner Mongolia Coal Econ:73–75. https://doi.org/10.13487/j.cnki.imce.021781
Song Z, Kuenzer C (2014) Coal fires in China over the last decade: a comprehensive review. Int J Coal Geol 133:72–99. https://doi.org/10.1016/j.coal.2014.09.004
Wang C, Yuan J, Bai Z, Hou Y, Deng J, Xiao Y (2023) Experimental investigation on using antioxidants to control spontaneous combustion of lignite coal. Fuel 334:126679. https://doi.org/10.1016/j.fuel.2022.126679
Wang H, Dlugogorski BZ, Kennedy EM (2003) Analysis of the mechanism of the low-temperature oxidation of coal. Combust Flame 134:107–117. https://doi.org/10.1016/S0010-2180(03)00086-5
Wang X, Hu X, Liang Y, Wu M, Xue D, Yu X, Liu J, Dong H, Luo C, Yang Z (2022) Early warning of coal spontaneous combustion: a study of CO response mechanism based on PANI/Ti3AlC2 composite gas sensing film. Chemistryselect 7:e202201563. https://doi.org/10.1002/slct.202201563
Wang Z, Burra KG, Zhang M, Li X, He X, Lei T, Gupta AK (2020a) Syngas evolution and energy efficiency in CO2-assisted gasification of pine bark. Appl Energ 269:114996. https://doi.org/10.1016/j.apenergy.2020.114996
Wang Z, Burra KG, Zhang M, Li X, Policella M, Lei T, Gupta AK (2020b) Co-pyrolysis of waste tire and pine bark for syngas and char production. Fuel 274:117878. https://doi.org/10.1016/j.fuel.2020.117878
Wen H, Yu Z, Fan S, Zhai X, Liu W (2017) Prediction of spontaneous combustion potential of coal in the gob area using CO extreme concentration: a case study. Combust Sci Technol 189:1713–1727. https://doi.org/10.1080/00102202.2017.1327430
Xin C, Liu Y (2015) Research on indexity gas of spontaneous combustion of coal prediction coal. Technology 34:145–148. https://doi.org/10.13301/j.cnki.ct.2015.08.057
Zhang J, Ren T, Liang Y, Wang Z (2016) A review on numerical solutions to self-heating of coal stockpile: Mechanism, theoretical basis, and variable study. Fuel 182:80–109. https://doi.org/10.1016/j.fuel.2016.05.087
Zhang M, Gong Z, Tan Z, Liu H, Yang M (2021) Study on heat adsorption based on the experiment of coal adsorption of methane by using the weight method. J Natural Gas Geosci 6:245–253. https://doi.org/10.1016/j.jnggs.2021.08.002
Zhang Y, Wu J, Chang L, Wang J, Li Z (2013) Changes in the reaction regime during low-temperature oxidation of coal in confined spaces. J Loss Prevent Proc 26:1221–1229. https://doi.org/10.1016/j.jlp.2013.05.008
Zhao J, Song J, Lu S, Ming H, Chang J, Deng J, Zhang Y, Shu C (2023a) Spontaneous oxidation kinetics of weathered coal based upon thermogravimetric characteristics. Energy 275:127489. https://doi.org/10.1016/j.energy.2023.127489
Zhao J, Zhang Y, Song J, Guo T, Deng J, Shu C (2023b) Oxygen distribution and gaseous products change of coal fire based upon the semi-enclosed experimental system. Energy 263:125721. https://doi.org/10.1016/j.energy.2022.125721
Zhou B, Yang S, Jiang X, Cai J, Xu Q, Song W, Zhou Q (2021) The reaction of free radicals and functional groups during coal oxidation at low temperature under different oxygen concentrations. Process Saf Environ 150:148–156. https://doi.org/10.1016/j.psep.2021.04.007
Funding
This work was supported by the “Outstanding Youth Research Project of Anhui Province (2022AH030084)”, and the “National Natural Science Foundation of China (No.52104178)”, and their supports are gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Jinhu Li: investigation, visualization, writing—original draft, getting funding; Qin Cao: investigation, visualization, writing—original draft; Wei Lu: methodology, project administration, writing—review and editing; Jingjuan Geng: investigation, review & writing; Jinliang Li: investigation, visualization, writing—original draft; Hui Zhuo: investigation, review & writing.
Corresponding author
Ethics declarations
Ethical approval
Not applicable. This study did not involve any ethical issues.
Consent to participate
All authors obtained the consent of the responsible authorities of the institution/organization where the work was carried out before submitting the work.
Consent to publish
All authors agreed to submit the manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Shimin Liu
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, ., Cao, Q., Lu, W. et al. Quantitative calculation of gases generation during low-temperature oxidation of coal. Environ Sci Pollut Res 30, 113774–113789 (2023). https://doi.org/10.1007/s11356-023-30219-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-30219-y