Abstract
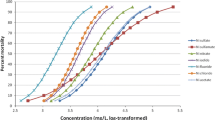
Caenorhabditis elegans is used for assessing the toxicity of chemicals in aqueous medium. However, chemicals can absorb to the bacterial food, which reduces the freely dissolved concentrations of the tested compounds. Thus, based on total or nominal concentrations, toxicity is underestimated, resulting in misleading assumptions on toxicity mechanisms or comparisons to other test organisms. As the verification of freely dissolved exposure concentrations (Cfree) is challenging in small test systems, simple partitioning models might by a good option for estimating Cfree. Therefore, C. elegans was exposed to seven differently acting organic chemicals with varying hydrophobicities, thus also different affinities to bind to the food of C. elegans. Measured concentrations of the dissolved aqueous and the bacterial-bound fraction allowed the calculation of binding constants (Kb). Experimental Kb were comparable to literature data of hydrophobic chemicals and correlated well with their hydrophobicity, expressed as log KOW. The chronic toxicity of the various compounds on C. elegans’ reproduction, based on their aqueous concentration, was weakly related to their log KOW. Toxicity expressed based on chemical activity and comparisons with a baseline toxicity model, nevertheless, suggested a narcotic mode of action for most hydrophobic compounds (except methylisothiazolinone and trichlorocarbanilide). Although revealing a similar toxicity ranking than Daphnia magna, C. elegans was less sensitive, probably due to its ability to reduce its internal concentrations by means of its very impermeable cuticle or by efficient detoxification mechanisms. It could be shown that measured aqueous concentrations in the nematode test system corresponded well with freely dissolved concentrations that were modeled using simple mass-balance models from nominal concentrations. This offers the possibility to estimate freely dissolved concentrations of chemicals from nominal concentrations, making routine testing of chemicals and their comparison to other species more accurate.


Similar content being viewed by others
Data availability
Raw data are available on request (hoess@ecossa.de).
References
Armitage JM, Wania F, Arnot JA (2014) Application of mass balance models and the chemical activity concept to facilitate the use of in vitro toxicity data for risk assessment. Environ Sci Technol 48:9770–9779. https://doi.org/10.1021/es501955g
ASTM (2022) Standard guide for conducting laboratory soil toxicity tests with the nematode Caenorhabditis elegans. American Society for Testing and Materials, Philadelphia, PA, USA
Bauer FJ, Thomas PC, Fouchard SY, Neunlist SJM (2018) High-accuracy prediction of mechanisms of action using structural alerts. Comput Toxicol 7:36–45. https://doi.org/10.1016/j.comtox.2018.06.004
Baughman GL, Paris DF (1981) Microbial bioconcentration of organic pollutants from aquatic systems - a critical review. Crit Rev Microbiol 8:205–227. https://doi.org/10.3109/10408418109085079
Bielská L, Kah M, Sigmund G et al (2017) Bioavailability and toxicity of pyrene in soils upon biochar and compost addition. Sci Total Environ 595:132–140. https://doi.org/10.1016/j.scitotenv.2017.03.230
Boyd WA, Smith MV, Kissling GE, Freedman JH (2010) Medium- and high-throughput screening of neurotoxicants using C. elegans. Neurotoxicol Teratol 32:68–73. https://doi.org/10.1016/j.ntt.2008.12.004
Brockmeier EK, Basili D, Herbert J et al (2022) Data-driven learning of narcosis mode of action identifies a CNS transcriptional signature shared between whole organism Caenorhabditis elegans and a fish gill cell line. Sci Total Environ 849:157666. https://doi.org/10.1016/j.scitotenv.2022.157666
Broeks A, Janssen HWRM, Calafat J, Plasterk RHA (1995) A P-glycoprotein protects Caenorhabditis elegans against natural toxins. EMBO J 14:1858–1866. https://doi.org/10.1002/j.1460-2075.1995.tb07178.x
Clavijo A, Kronberg MF, Rossen A et al (2016) The nematode Caenorhabditis elegans as an integrated toxicological tool to assess water quality and pollution. Sci Total Environ 569–570:252–261. https://doi.org/10.1016/j.scitotenv.2016.06.057
Fein JB, Daughney CJ, Yee N, Davis TA (1997) A chemical equilibrium model for metal adsorption onto bacterial surfaces. Geochim Cosmochim Acta 61:3319–3328. https://doi.org/10.1016/S0016-7037(97)00166-X
Ferguson J (1939) The use of chemical potentials as indices of toxicity. Proc R Soc Lond Ser B Biol Sci 127:387–404. https://doi.org/10.1098/rspb.1939.0030
Fischer F, Böhm L, Höss S et al (2016) Passive dosing in chronic toxicity tests with the nematode Caenorhabditis elegans. Environ Sci Technol 50:9708–9716. https://doi.org/10.1021/acs.est.6b02956
Fukushige T, Smith HE, Miwa J et al (2017) A genetic analysis of the Caenorhabditis elegans detoxification response. Genetics 206:939–952. https://doi.org/10.1534/genetics.117.202515
Gao S, Chen W, Zeng Y et al (2018) Classification and prediction of toxicity of chemicals using an automated phenotypic profiling of Caenorhabditis elegans. BMC Pharmacol Toxicol 19:1–11. https://doi.org/10.1186/s40360-018-0208-3
Haegerbaeumer A, Höss S, Heininger P, Traunspurger W (2018) Is Caenorhabditis elegans representative of freshwater nematode species in toxicity testing? Environ Sci Pollut Res 25:2879–2888. https://doi.org/10.1007/s11356-017-0714-7
Helmcke KJ, Avila DS, Aschner M (2010) Utility of Caenorhabditis elegans in high throughput neurotoxicological research. Neurotoxicol Teratol 32:62–67. https://doi.org/10.1016/j.ntt.2008.11.005
Höss S, Ahlf W, Fahnenstich C et al (2010) Variability of freshwater sediment contact tests in sediments with low-level anthropogenic contamination – determination of toxicity thresholds. Environ Pollut 158:2999–3010. https://doi.org/10.1016/j.envpol.2010.05.013
Höss S, Bergtold M, Haitzer M et al (2001a) Refractory dissolved organic matter can influence the reproduction of Caenorhabditis elegans (Nematoda). Freshw Biol 46:1–10. https://doi.org/10.1046/j.1365-2427.2001.00639.x
Höss S, Henschel T, Haitzer M et al (2001b) Toxicity of cadmium to Caenorhabditis elegans (Nematoda) in whole sediment and porewater - the ambiguous role of organic matter. Environ Toxicol Chem 20:2794–2801. https://doi.org/10.1002/etc.5620201219
Höss S, Jänsch S, Junker T et al (2009) Assessing the toxicity of contaminated soils using the nematode Caenorhabditis elegans as test organism. Ecotoxicol Environ Saf 72:1811–1818. https://doi.org/10.1016/j.ecoenv.2009.07.003
Höss S, Römbke J (2019) Effects of waste materials on Caenorhabditis elegans (Nematoda) using the ISO standard soil toxicity test. Environ Sci Pollut Res 26:26304–26312. https://doi.org/10.1007/s11356-019-05891-8
Höss S, Schlottmann K, Traunspurger W (2011) Toxicity of ingested cadmium to the nematode Caenorhabditis elegans. Environ Sci Technol 45:10219–10225. https://doi.org/10.1021/es2027136
Huffman DL, Abrami L, Sasik R et al (2004a) Mitogen-activated protein kinase pathways defend against bacterial pore-forming toxins. Proc Natl Acad Sci USA 101:10995–11000. https://doi.org/10.1073/pnas.0404073101
Huffman DL, Bishof LJ, Griffitts JS, Aroian RV (2004b) Pore worms: using Caenorhabditis elegans to study how bacterial toxins interact with their target host. IntJMedMicrobiol 293:1–9. https://doi.org/10.1078/1438-4221-00303
Hunt PR (2017) The C. elegans model in toxicity testing. J Appl Toxicol 37:50–59. https://doi.org/10.1002/jat.3357
ISO (2010) Water quality - determination of the toxic effect of sediment and soil samples on growth, fertility and reproduction of Caenorhabditis elegans (Nematoda); ISO 10872:2010, International Organization for Standardization, Geneva, Switzerland
Jackson BP, Williams PL, Lanzirotti A, Bertsch PM (2005) Evidence for biogenic pyromorphite formation by the nematode Caenorhabditis elegans. Environ Sci Technol 39:5620–5625. https://doi.org/10.1021/es050154k
Jos A, Repetto G, Ríos JC et al (2005) Ecotoxicological evaluation of the additive butylated hydroxyanisole using a battery with six model systems and eighteen endpoints. Aquat Toxicol 71:183–192. https://doi.org/10.1016/j.aquatox.2004.11.004
Kienzler A, Barron MG, Belanger SE et al (2017) Mode of action (MOA) assignment classifications for ecotoxicology: an evaluation of approaches. Environ Sci Technol 51:10203–10211. https://doi.org/10.1021/acs.est.7b02337
Kim SW, Kim D, Jeong S-W, An Y-J (2020) Size-dependent effects of polystyrene plastic particles on the nematode Caenorhabditis elegans as related to soil physicochemical properties. Environ Pollut 258:113740. https://doi.org/10.1016/j.envpol.2019.113740
Kwon H, Roh JY, Lim D et al (2011) Maintaining the constant exposure condition for an acute Caenorhabditis elegans mortality test using passive dosing. Environ Health Toxicol 26:1–7. https://doi.org/10.5620/eht.2011.26.e2011015
Lanzerstorfer P, Sandner G, Pitsch J et al (2021) Acute, reproductive, and developmental toxicity of essential oils assessed with alternative in vitro and in vivo systems. Arch Toxicol 95:673–691. https://doi.org/10.1007/s00204-020-02945-6
Lenz KA, Pattison C, Ma H (2017) Triclosan (TCS) and triclocarban (TCC) induce systemic toxic effects in a model organism the nematode Caenorhabditis elegans. Environ Pollut 231:462–470. https://doi.org/10.1016/j.envpol.2017.08.036
Lima TRR, Martins AC, Pereira LC, Aschner M (2022) Toxic effects induced by diuron and its metabolites in Caenorhabditis elegans. Neurotox Res. https://doi.org/10.1007/s12640-022-00596-2
Mackay D, Arnot JA, Petkova EP et al (2009) The physicochemical basis of QSARs for baseline toxicity. SAR QSAR Environ Res 20:393–414. https://doi.org/10.1080/10629360902949153
Mackay D, Arnot JA, Celsie A et al (2014) QSARs for aquatic toxicity: celebrating, extending and displaying the pioneering contributions of Ferguson, Konemann and Veith. SAR QSAR Environ Res 25:343–355. https://doi.org/10.1080/1062936X.2014.900521
Mayer P, Reichenberg F (2006) Can highly hydrophobic organic substances cause aquatic baseline toxicity and can they contribute to mixture toxicity? Environ Toxicol Chem 25:2639–2644. https://doi.org/10.1897/06-142r.1
Mueller M-T, Fueser H, Trac LN et al (2020) Surface-related toxicity of polystyrene beads to nematodes and the role of food availability. Environ Sci Technol 54:1790–1798. https://doi.org/10.1021/acs.est.9b06583
Offermann K, Matthäi A, Ahlf W (2009) Assessing the importance of dietborne cadmium and particle characteristics on bioavailability and bioaccumulation in the nematode Caenorhabditis elegans. Environ Toxicol Chem 28:1149–1158. https://doi.org/10.1897/08-272.1
Partridge FA, Tearle AW, Gravato-nobre MJ et al (2008) The C. elegans glycosyltransferase BUS-8 has two distinct and essential roles in epidermal morphogenesis. Dev Biol 317:549–559. https://doi.org/10.1016/j.ydbio.2008.02.060
Ristau K, Akgül Y, Bartel AS et al (2015) Toxicity in relation to mode of action for the nematode Caenorhabditis elegans: acute to chronic ratios and quantitative structure-activity relationships. Environ Toxicol Chem 34:2347–2353. https://doi.org/10.1002/etc.3069
Roh J-Y, Lee H, Kwon J-H (2014) Changes in the expression of cyp35a family genes in the soil nematode Caenorhabditis elegans under controlled exposure to chlorpyrifos using passive dosing. Environ Sci Technol 48:10475–10481. https://doi.org/10.1021/es5027773
Saleem S, Böhme A, Schüürmann G (2023) Baseline narcosis for the glass-vial 96-h growth inhibition of the nematode C. elegans and its use for identifying electrophilic and pro-electrophilic toxicity. Environ Sci Technol 57:1692–1700. https://doi.org/10.1021/acs.est.2c05217
Schertzinger G, Zimmermann S, Grabner D, Sures B (2017) Assessment of sublethal endpoints after chronic exposure of the nematode Caenorhabditis elegans to palladium, platinum and rhodium. Environ Pollut 230:31–39. https://doi.org/10.1016/j.envpol.2017.06.040
Schertzinger G, Zimmermann S, Sures B (2019) Predicted sediment toxicity downstream of combined sewer overflows corresponds with effects measured in two sediment contact bioassays. Environ Pollut 248:782–791. https://doi.org/10.1016/j.envpol.2019.02.079
Schmidt SN, Armitage JM, Arnot JA et al (2018) Linking algal growth inhibition to chemical activity: excess toxicity below 0.1 % of saturation. Chemosphere 208:880–886. https://doi.org/10.1016/j.chemosphere.2018.05.168
Smith KEC, Schmidt SN, Dom N et al (2013) Baseline toxic mixtures of non-toxic chemicals: “solubility addition” increases exposure for solid hydrophobic chemicals. Environ Sci Technol 47:2026–2033. https://doi.org/10.1021/es3040472
Sochova I, Hofman J, Holoubek I (2007) Effects of seven organic pollutants on soil nematode Caenorhabditis elegans. Environ Int 33:798–804. https://doi.org/10.1016/j.envint.2007.03.001
Spann N, Goedkoop W, Traunspurger W (2015) Phenanthrene bioaccumulation in the nematode Caenorhabditis elegans. Environ Sci Technol 49:1842–1850. https://doi.org/10.1021/es504553t
Sreevidya VS, Lenz KA, Svoboda KR, Ma H (2018) Benzalkonium chloride, benzethonium chloride, and chloroxylenol - three replacement antimicrobials are more toxic than triclosan and triclocarban in two model organisms. Environ Pollut 235:814–824. https://doi.org/10.1016/j.envpol.2017.12.108
Stupp GS, von Reuss SH, Izrayelit Y et al (2013) Chemical detoxification of small molecules by Caenorhabditis elegans. ACS Chem Biol 8:309–313. https://doi.org/10.1021/cb300520u
Thomas PC, Velthoven K, Geurts M, van Wijk D (2009) Bioavailability and detoxification of cationics: II. Relationship between toxicity and CEC of cationic surfactants on Caenorhabditis elegans (Nematoda) in artificial and natural substrates. Chemosphere 75:310–318. https://doi.org/10.1016/j.chemosphere.2009.02.003
Traunspurger W, Haitzer M, Höss S et al (1997) Ecotoxicological assessment of aquatic sediments with Caenorhabditis elegans (Nematoda) - a method for testing in liquid medium and whole sediment samples. Environ Toxicol Chem 16:245–250. https://doi.org/10.1002/etc.5620160221
Tuikka A, Schmitt C, Höss S et al (2011) Toxicity assessment of sediments from three European river basins using a sediment contact test battery. Ecotoxicol Environ Saf 74:123–131. https://doi.org/10.1016/j.ecoenv.2010.08.038
US EPA (2012) Estimation programs interface SuiteTM for Microsoft® Windows, v 4.11; United States Environmental Protection Agency, Washington, DC, USA
Verhaar HJM, van Leeuwen CJ, Hermens JLM (1992) Classifying environmental pollutants. Chemosphere 25:471–491. https://doi.org/10.1016/0045-6535(92)90280-5
Williams PL, Dusenbery DB (1990) Aquatic toxicity testing using the nematode Caenorhabditis elegans. Environ Toxicol Chem 9:1285–1290. https://doi.org/10.1002/etc.5620091007
Xiong H, Pears C, Woollard A (2017) An enhanced C. elegans based platform for toxicity assessment. Sci Rep 1–11. https://doi.org/10.1038/s41598-017-10454-3
Acknowledgements
This study was funded by Unilever (Project: CH-2011-0953). The gifts of Caenorhabditis elegans (strains N2) and Escherichia coli (strain OP50) from the Caenorhabditis Genetic Center, which is supported by the National Institutes of Health, are gratefully acknowledged. We want to thank Jayne Roberts and Geoff Hodges for helpful comments on an earlier version of the manuscript and for their help with MechoA profiling. Moreover, we want to thank two anonymous reviewers for their valuable comments on an earlier version of the manuscript.
Funding
This study was funded by Unilever (Project: CH-2011-0953)
Author information
Authors and Affiliations
Contributions
Sebastian Höss and Roger van Egmont contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Sebastian Höss and David Sanders. The first draft of the manuscript was written by Sebastian Höss, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ludek Blaha
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 212 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Höss, ., Sanders, D. & van Egmond, R. Determining the toxicity of organic compounds to the nematode Caenorhabditis elegans based on aqueous concentrations. Environ Sci Pollut Res 30, 96290–96300 (2023). https://doi.org/10.1007/s11356-023-29193-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-29193-2