Abstract
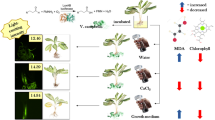
Glowing Episcia lilacina was generated through foliar application of the bioluminescent bacterium Vibrio campbellii RMT1. Firstly, different nutrient formulas were tested, incorporating yeast extract and various inorganic salts, such as CaCl2, MgCl2, MgSO4, KH2PO4, K2HPO4, and NaCl, in order to enhance bacterial growth and light emission. The combination of 0.15% of yeast extract and 0.3% of CaCl2 in a nutrient broth (NB) + 1% NaCl medium extended light emission to 24 h and resulted in higher light intensity compared to other combinations of yeast extract and inorganic salts. The peak intensity reached approximately 1.26 × 108 relative light units (RLU) at 7 h. The optimal presence of inorganic salt ions likely contributed to enhanced light emission, while the yeast extract acted as a nutrient source. Secondly, the effect of proline on salt-induced stress symptoms was investigated by applying 20 mM proline to the glowing plant. Additionally, a 0.5% agar nutrient was spread on the leaves prior to bacteria application to support bacterial growth and penetration. Exogenous proline application led to a significant accumulation of proline in plant cells, resulting in decreased malondialdehyde (MDA) levels. However, the proline accumulation also reduced the light intensity of the bioluminescent bacteria. This study demonstrates the potential for generating light on a living plant using bioluminescent bacteria. Further understanding of the interaction between plants and light-emitting bacteria could contribute to the development of sustainably light-emitting plants.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Bates LS, Waldren RA, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Bose JL, Kim U, Bartkowski W, Gunsalus RP, Overley AM, Lyell NL, Visick KL, Stabb EV (2007) Bioluminescence in Vibrio fischeri is controlled by the redox-responsive regulator ArcA. Mol Microbiol 65(2):538–553. https://doi.org/10.1111/j.1365-2958.2007.05809.x
Brodl E, Winkler A, Macheroux P (2018) Molecular mechanisms of bacterial bioluminescence. Comput Struct Biotechnol J 16:551–564. https://doi.org/10.1016/j.csbj.2018.11.003
Czyz A, Plata K, Wegrzyn G (2002) Induction of light emission by luminescent bacteria treated with UV light and chemical mutagens. J Appl Genet 43(3): 377–390. https://europepmc.org/article/med/12177528
Deluca M (1976) Firefly luciferase. Adv Enzymol Relat Areas Mol Biol 44:37–68. https://doi.org/10.1002/9780470122891.ch2
Deryabin DG, Aleshina ES (2008) Effect of salts on luminescence of natural and recombinant luminescent bacterial biosensors. Appl Biochem Microbiol 44:292–296. https://doi.org/10.1134/S0003683808030113
Dolan KM, Greenberg EP (1992) Evidence that GroEL, not sigma 32, is involved in transcriptional regulation of the Vibrio fischeri luminescence genes in Escherichia coli. J Bacteriol 174(15):5132–5135. https://doi.org/10.1128/jb.174.15.5132-5135.1992
Ellis EA, Oakley TH (2016) High rates of species accumulation in animals with bioluminescent courtship displays. Curr Biol 26(14):1916–1921. https://doi.org/10.1016/j.cub.2016.05.043
Fleiss A, Sarkisyan KS (2019) A brief review of bioluminescent systems (2019). Curr Genet 65(4):877–882. https://doi.org/10.1007/s00294-019-00951-5
Haddock SH, Moline MA, Case JF (2010) Bioluminescence in the sea. Ann Rev Mar Sci 2:443–493. https://doi.org/10.1146/annurev-marine-120308-081028
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Signal Behav 7(11):1456–1466. https://doi.org/10.4161/psb.21949
Henry JP, Michelson AM (1978) Bioluminescence: physiological control and regulation at the molecular level. Photochem Photobiol 28:293–310. https://doi.org/10.1111/j.1751-1097.1978.tb07711.x
Hodges DM, DeLong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611. https://doi.org/10.1007/s004250050524
Homaei AA, Mymandi AB, Sariri R, Kamrani E, Stevanato R, Etezad SM, Khajeh K (2013) Purification and characterization of a novel thermostable luciferase from Benthosema pterotum. J Photochem Photobiol b: Biol 125:131–136. https://doi.org/10.1016/j.jphotobiol.2013.05.015
Hurtado-Gallego J, Martín-Betancor K, Rodea-Palomares I, Leganés F, Rosal R, Fernández-Piñas F (2018) Two novel cyanobacterial bioluminescent whole-cell bioreporters based on superoxide dismutases MnSod and FeSod to detect superoxide anion. Chemosphere 201:772–779. https://doi.org/10.1016/j.chemosphere.2018.03.012
Kotlobay AA, Sarkisyan KS, Mokrushina YA, Marcet-Houben M, Serebrovskaya EO, Markina NM, Gonzalez Somermeyer L, Gorokhovatsky AY, Vvedensky A, Purtov KV, Petushkov VN (2018) Genetically encodable bioluminescent system from fungi. Proc Natl Acad Sci 115(50):12728–12732. https://doi.org/10.1073/pnas.1803615115
Khakhar A, Starker CG, Chamness JC, Lee N, Stokke S, Wang C, Swanson R, Rizvi F, Imaizumi T, Voytas DF (2020) Building customizable auto-luminescent luciferase-based reporters in plants. Elife 9:e52786. https://doi.org/10.7554/eLife.52786
Kotagiri D, Kolluru VC (2017) Effect of salinity stress on the morphology and physiology of five different Coleus species. Biomed Pharmacol J 10(4). https://doi.org/10.13005/bpj/1275
Kotlobay AA, Kaskova ZM, Yampolsky IV (2020) Palette of luciferases: natural biotools for new applications in biomedicine. Acta Naturae 12(2):15–27. https://doi.org/10.32607/actanaturae.10967
Krichevsky A, Meyers B, Vainstein A, Maliga P, Citovsky V (2010) Autoluminescent plants. PloS One 5(11):e15461. https://doi.org/10.1371/journal.pone.0015461
Kwak SY, Giraldo JP, Wong MH, Koman VB, Lew TT, Ell J, Weidman MC, Sinclair RM, Landry MP, Tisdale WA, Strano MS (2017) A nanobionic light-emitting plant. Nano Lett 17(12):7951–7961. https://doi.org/10.1021/acs.nanolett.7b04369
Lange CR, Lange SR (1997) Biomonitoring. Water Environ Res 69(4):900–15. https://www.jstor.org/stable/25044936
Majdura J, Jankiewicz U, Gałązka A, Orzechowski S (2023) The role of quorum sensing molecules in bacterial-plant interaction. Metabolites 13(1):114. https://doi.org/10.3390/metabo13010114
Mitiouchkina T, Mishin AS, Somermeyer LG, Markina NM, Chepurnyh TV, Guglya EB, Karataeva TA, Palkina KA, Shakhova ES, Fakhranurova LI, Chekova SV (2020) Plants with genetically encoded autoluminescence. Nat Biotechnol 38(8):944–946. https://doi.org/10.1038/s41587-020-0500-9
Nijvipakul S, Wongratana J, Suadee C, Entsch B, Ballou DP, Chaiyen P (2008) LuxG is a functioning flavin reductase for bacterial luminescence. J Bacteriol 190(5):1531–1538. https://doi.org/10.1128/JB.01660-07
Ohmiya Y, Hirano T (1996) Shining the light: the mechanism of the bioluminescence reaction of calcium-binding photoproteins. Chem Biol 3(5):337–347. https://doi.org/10.1016/S1074-5521(96)90116-7
Okuma E, Soeda K, Tada M, Murata Y (2000) Exogenous proline mitigates the inhibition of growth of Nicotiana tabacum cultured cells under saline conditions. Soil Sci Plant Nutr 46(1):257–263. https://doi.org/10.1080/00380768.2000.10408781
Ramesh C, Mohanraju R, Murthy KN, Karthick P, Narayana S (2014) Impact of light, temperature, salinity and glycerol on the intensity of luminescence and growth of marine bioluminescent bacteria Vibrio campbelli (strain STF1). Curr Sci 106(4):511–3. https://doi.org/10.18520/CS/V106/I4/511-513
Ramesh C, Meyer-Rochow VB (2021) Bioluminescence in aquatic and terrestrial organisms elicited through various kinds of stimulation. Aquat Ecol 55(3):737–764. https://doi.org/10.1007/s10452-021-09875-0
Saxena SC, Kaur H, Verma P, Petla BP, Andugula VR, Majee M (2013) Osmoprotectants: potential for crop improvement under adverse conditions. In Plant Acclimation to Environmental Stress; Springer: New York. NY, USA 2013:197–232
Shama G, Malik DJ (2013) The uses and abuses of rapid bioluminescence-based ATP assays. Int J Hyg Environ Health 216(2):115–125. https://doi.org/10.1016/j.ijheh.2012.03.009
Sharifian S, Homaei A, Hemmati R, Khajeh K (2017) Light emission miracle in the sea and preeminent applications of bioluminescence in recent new biotechnology. J Photochem Photobiol b: Biol 172:115–128. https://doi.org/10.1016/j.jphotobiol.2017.05.021
Sharifian S, Homaei A, Hemmati R, Luwor RB, Khajeh K (2018) The emerging use of bioluminescence in medical research. Biomed Pharmacother 101:74–86. https://doi.org/10.1016/j.biopha.2018.02.065
Shimomura O (2006) Bioluminescence: chemical principles and methods. World Scientific Publishing, Singapore
Stabb EV, Butler MS, Adin DM (2004) Correlation between osmolarity and luminescence of symbiotic Vibrio fischeri strain ES114. J Bacteriol 186(9):2906–2908. https://doi.org/10.1128/JB.186.9.2906-2908.2004
Susanto A, Winardi W, Hidayat M, Wirawan A (2021) The use of indoor plant as an alternative strategy to improve indoor air quality in Indonesia. Rev Environ Health 36(1):95–99. https://doi.org/10.1515/reveh-2020-0062
Tabei Y, Era M, Ogawa A, Morita H (2011) Effects of magnesium sulfate on the luminescence of Vibrio fischeri under nutrient-starved conditions. Biosci Biotechnol Biochem 75(6):1073–1078. https://doi.org/10.1271/bbb.100880
Ulitzur S, Yashphe J (1975) An adenosine 3′, 5′-monophosphate-requiring mutant of the luminous bacteria Beneckea harveyi. Biochim Biophys Acta Gen Subj 404(2):321–328. https://doi.org/10.1016/0304-4165(75)90339-6
Vázquez CI, Iglesias RA (2018) Engineered nanomaterials in energy production industry, Chapter-38. In: Hussain CM, editor. Handbook of nanomaterials for industrial applications. Elsevier 713e23. https://doi.org/10.1016/B978-0-12-813351-4.00039-0
Wainwright PC, Longo SJ (2017) Functional innovations and the conquest of the oceans by acanthomorph fishes. Curr Biol 27(11):R550–R557. https://doi.org/10.1016/j.cub.2017.03.044
Watanabe H, Inaba H, Hastings JW (1991) Effects of aldehyde and internal ions on bioluminescence expression of Photobacterium phosphoreum. Arch Microbiol 156:1–4. https://doi.org/10.1007/BF00418179
Widder EA (2010) Bioluminescence in the ocean: origins of biological, chemical, and ecological diversity. Science 328(5979):704–708. https://doi.org/10.1126/science.1174269
Acknowledgements
The authors thank Research and Innovation for Sustainability Centre (RISC) and Magnolia Quality Development Corporation Limited (MQDC) for supporting our project.
Funding
This project is funded by the School of Bioresources and Technology, King Mongkut’s University of Technology Thonburi.
Author information
Authors and Affiliations
Contributions
Hsu Myat Thar: methodology, investigation, data curation, formal analysis, validation, writing—original draft; Chairat Treesubsuntorn: review and editing; Paitip Thiravetyan: resources, supervision, review, and editing; Rujira Dolphen: conceptualization, data curation, formal analysis, validation, writing—review and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Disclaimer
We declare that we have no financial and personal relationships with other people or organizations that can inappropriately influence our work.
Additional information
Responsible Editor: Gangrong Shi
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thar, H.M., Treesubsuntorn, C., Thiravetyan, P. et al. Development of light-emitting Episcia lilacina leaf by applying Vibrio campbellii RMT1 and extending the glowing by CaCl2 and yeast extract. Environ Sci Pollut Res (2023). https://doi.org/10.1007/s11356-023-28657-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-023-28657-9