Abstract
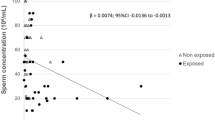
According to the United Nations’ Food and Agriculture Organization, the quantities of pesticide used around the world have increased regularly since the 1990s. Given that pesticides may be classified as carcinogenic, mutagenic, neurotoxic, or toxic for reproduction, some have endocrine-disrupting properties that might be associated with a decline in sperm parameters in general and sperm DNA integrity in particular. These days, a sperm analysis is not enough to determine the etiology of male infertility. Genome integrity analysis is a key step in clarifying a large proportion of cases of male infertility. The objective of the present retrospective study was to assess the impact of self-reported pesticide exposure on sperm parameters and sperm DNA integrity in men consulting for infertility. In a retrospective study, a population of 671 men living in the Picardy region of France were assessed in a conventional sperm parameter analysis, Shorr staining, a DNA fragmentation assay (terminal deoxynucleotidyl transferase-mediated dUDP nick-end labelling), and chromatin decondensation with aniline blue staining. The exposed and the non-exposed groups did not differ significantly in some of the conventional sperm parameters (including volume, sperm count, and percent typical forms). However, vitality, progressive motility, and non-progressive motility were significantly lower in the exposed group. Levels of DNA fragmentation and chromatin decondensation were moderately higher in the exposed group.

Similar content being viewed by others
Data availability
Not applicable.
References
Abell A, Ernst E, Bonde JP (2000) Semen quality and sexual hormones in greenhouse workers. Scand J Work Environ Health 26:492–500. https://doi.org/10.5271/sjweh.573
Alvarez Sedó C, Bilinski M, Lorenzi D, Uriondo H, Noblía F, Longobucco V, Lagar EV, Nodar F (2017) Effect of sperm DNA fragmentation on embryo development: clinical and biological aspects. JBRA Assist Reprod 21:343–350. https://doi.org/10.5935/1518-0557.20170061
Amjad S, Rahman MS, Pang WK, Ryu DY, Adegoke EO, Park YJ, Pand MG (2021) Effects of phthalates on the functions and fertility of mouse spermatozoa. Toxicology 454. https://doi.org/10.1016/j.tox.2021.152746
Åsenius F, Danson AF, Marzi SJ (2020) DNA methylation in human sperm: a systematic review. Hum Reprod Update 26:841–873. https://doi.org/10.1093/humupd/dmaa025
Baskaran S, Finelli R, Agarwal A, Henkel R (2021) Diagnostic value of routine semen analysis in clinical andrology. Andrologia 53:e13614. https://doi.org/10.1111/and.13614
Benatta M, Kettache R, Buchholz N, Trinchieri A (2020) The impact of nutrition and lifestyle on male fertility. Arch Ital Urol Androl 92. https://doi.org/10.4081/aiua.2020.2.121
Benchaib M, Ajina M, Lornage J, Niveleau A, Durand P, Guérin JF (2003) Quantitation by image analysis of global DNA methylation in human spermatozoa and its prognostic value in in vitro fertilization: a preliminary study. Fertil Steril 80:947–953. https://doi.org/10.1016/S0015-0282(03)01151-8
Boeri L, Fallara G, Pozzi E, Belladelli F, Corsini C, Raffo M, Schifano N, Capogrosso P, d’Arma A, Montorsi F, Salonia A (2022) The impact of different WHO reference criteria for semen analysis in clinical practice: who will benefit from the new 2021 thresholds for normal semen parameters? Andrology 10:1134–1142. https://doi.org/10.1111/andr.13213
Bretveld R, Brouwers M, Ebisch I, Roeleveld N (2007) Influence of pesticides on male fertility. Scand J Work Environ Health 33:13–28. https://doi.org/10.5271/sjweh.1060
Buck Louis GM, Sundaram R, Schisterman EF, Sweeney A, Lynch CD, Kim S, Maisog JM, Gore-Langton R, Eisenberg ML, Chen Z (2014) Semen quality and time to pregnancy: the Longitudinal Investigation of Fertility and the Environment Study. Fertil Steril 101:453–462. https://doi.org/10.1016/j.fertnstert.2013.10.022
Cabry R, Merviel P, Madkour A, Lefranc E, Scheffler F, Desailloud R, Bach V, Benkhalifa M (2020) The impact of endocrine disruptor chemicals on oocyte/embryo and clinical outcomes in IVF. Endocr Connect 9:R134–R142. https://doi.org/10.1530/EC-20-0135
Carlsen E, Giwercman A, Keiding N, Skakkebaek NE (1992) Evidence for decreasing quality of semen during past 50 years. BMJ 305:609–613. https://doi.org/10.1136/bmj.305.6854.609
Carrell DT, Emery BR, Hammoud S (2008) The aetiology of sperm protamine abnormalities and their potential impact on the sperm epigenome. Int J Androl 31:537–545. https://doi.org/10.1111/j.1365-2605.2008.00872.x
Clair E, Mesnage R, Travert C, Séralini G-É (2012) A glyphosate-based herbicide induces necrosis and apoptosis in mature rat testicular cells in vitro, and testosterone decrease at lower levels. Toxicol in Vitro 26:269–279. https://doi.org/10.1016/j.tiv.2011.12.009
Dialog FS (2014) Semen quality and time to pregnancy the Longitudinal Investigation of Fertility and the Environment Study [WWW Document]. Fertility and Sterility Dialog. URL https://www.fertstertdialog.com/posts/8919-semen-quality-and-time-to-pregnancy-the-longitudinal-investigation-of-fertility-and-the-environment-study (accessed 8.9.22)
Djekkoun N, Lalau J-D, Bach V, Depeint F, Khorsi-Cauet H (2021) Chronic oral exposure to pesticides and their consequences on metabolic regulation: role of the microbiota. Eur J Nutr 60:4131–4149. https://doi.org/10.1007/s00394-021-02548-6
El-Dakdoky MH, Abd El-Wahab HMF (2013) Impact of boric acid exposure at different concentrations on testicular DNA and male rats fertility. Toxicol Mech Methods 23:360–367. https://doi.org/10.3109/15376516.2013.764951
Jaeger A (2009) La toxicologie médicale : état des lieux et perspectives. Réanimation 18:545–546. https://doi.org/10.1016/j.reaurg.2009.08.009
Jensen TK, Bonde JP, Joffe M (2006) The influence of occupational exposure on male reproductive function. Occup Med 56:544–553. https://doi.org/10.1093/occmed/kql116
Jouzel J-N, Prete G (2022) Pesticides et santé humaine. Études Octobre, 45–56. https://doi.org/10.3917/etu.4297.0045
Khalafalla K, Majzoub A, Elbardisi H, Bhathella A, Chaudhari A, Agarwal A, Henkel R, AlMarzooki T, Burjaq H, Arafa M (2021) The effect of sperm DNA fragmentation on intracytoplasmic sperm injection outcome. Andrologia 53:e14180. https://doi.org/10.1111/and.14180
Kim K-H, Kabir E, Jahan SA (2017) Exposure to pesticides and the associated human health effects. Sci Total Environ 575:525–535. https://doi.org/10.1016/j.scitotenv.2016.09.009
Knapke ET, de P Magalhaes D, Dalvie MA, Mandrioli D, Perry MJ (2022) Environmental and occupational pesticide exposure and human sperm parameters: a navigation guide review. Toxicology 465:153017. https://doi.org/10.1016/j.tox.2021.153017
Li H, Deng H, Zhao J, Zhong Y-C, Zhu L, Wu Y-Q, Ma W-M (2020) Impact of sperm DNA fragmentation on the outcome of frozen-thawed blastocyst transfer in in vitro fertilization and embryo transfer. Zhonghua Nan Ke Xue 26:422–426
Lopes-Ferreira M, Maleski ALA, Balan-Lima L, Bernardo JTG, Hipolito LM, Seni-Silva AC, Batista-Filho J, Falcao MAP, Lima C (2022) Impact of pesticides on human health in the last six years in Brazil. Int J Environ Res Public Health 19:3198. https://doi.org/10.3390/ijerph19063198
Lushchak VI, Matviishyn TM, Husak VV, Storey JM, Storey KB (2018) Pesticide toxicity: a mechanistic approach. EXCLI J 17:1101–1136. https://doi.org/10.17179/excli2018-1710
Mahna D, Puri S, Sharma S (2021) DNA methylation modifications: mediation to stipulate pesticide toxicity. Int J Environ Sci Technol 18:531–544. https://doi.org/10.1007/s13762-020-02807-9
Menezo Y, Clement P, Amar E (2017) Evaluation of sperm DNA structure, fragmentation and decondensation: an essential tool in the assessment of male infertility. Transl Androl Urol 6:S553–S556. https://doi.org/10.21037/tau.2017.03.11
Merviel P, Cabry R, Chardon K, Haraux E, Scheffler F, Mansouri NB, Devaux A, Chahine H, Bach V, Copin H, Benkhalifa M (2017) Impact of oocytes with CLCG on ICSI outcomes and their potential relation to pesticide exposure. J Ovarian Res 10:42. https://doi.org/10.1186/s13048-017-0335-2
Montjean D, Ravel C, Benkhalifa M, Cohen-Bacrie P, Berthaut I, Bashamboo A, McElreavey K (2013) Methylation changes in mature sperm deoxyribonucleic acid from oligozoospermic men: assessment of genetic variants and assisted reproductive technology outcome. Fertil Steril 100:1241–1247. https://doi.org/10.1016/j.fertnstert.2013.06.047
Montjean D, Zini A, Ravel C, Belloc S, Dalleac A, Copin H, Boyer P, McElreavey K, Benkhalifa M (2015) Sperm global DNA methylation level: association with semen parameters and genome integrity. Andrology 3:235–240. https://doi.org/10.1111/andr.12001
Padungtod C, Savitz DA, Overstreet JW, Christiani DC, Ryan LM, Xu X (2000) Occupational pesticide exposure and semen quality among Chinese workers. J Occup Environ Med 42:982–992. https://doi.org/10.1097/00043764-200010000-00004
Perry MJ (2008) Effects of environmental and occupational pesticide exposure on human sperm: a systematic review. Hum Reprod Update 14:233–242. https://doi.org/10.1093/humupd/dmm039
Pesticides et santé – Nouvelles données (2021) ⋅ Inserm, La science pour la santé [WWW Document], n.d. URL https://www.inserm.fr/expertise-collective/pesticides-et-sante-nouvelles-donnees-2021/?fbclid=IwAR1080UTeLYvsQi8x1LWpGgenfjkeLC47eT26t7adrB9jjDp4PAFgdheQi4 (accessed 10.12.21)
Pesticides, agriculture et environnement : Réduire l’utilisation des pesticides et en limiter les impacts environnementaux. [WWW Document], n.d. . INRAE Institutionnel. URL https://www.inrae.fr/actualites/pesticides-agriculture-environnement-reduire-lutilisation-pesticides-limiter-impacts-environnementaux (accessed 10.13.22)
Rudneva SA, Bragina EE, Arifulin EA, Sorokina TM, Shileyko LV, Ermolaeva SA, Kurilo LF, Chernykh VB (2015) DNA fragmentation in spermatozoa and its relationship with impaired spermatogenesis. Androl Genital Surg 15:26–33. https://doi.org/10.17650/2070-9781-2014-4-26-33
Sengupta, P., Banerjee, R., 2013. Environmental toxins: alarming impacts of pesticides on male fertility. Human Exp Toxicol 33. https://doi.org/10.1177/0960327113515504
Sifakis S, Androutsopoulos VP, Tsatsakis AM, Spandidos DA (2017) Human exposure to endocrine disrupting chemicals: effects on the male and female reproductive systems. Environ Toxicol Pharmacol 51:56–70. https://doi.org/10.1016/j.etap.2017.02.024
Song B, Chen Y, Wang C, Li G, Wei Z, He X, Cao Y (2022) Poor semen parameters are associated with abnormal methylation of imprinted genes in sperm DNA. Reprod Biol Endocrinol 20:155. https://doi.org/10.1186/s12958-022-01028-8
Tudi M, Daniel Ruan H, Wang L, Lyu J, Sadler R, Connell D, Chu C, Phung DT (2021) Agriculture development, pesticide application and its impact on the environment. Int J Environ Res Public Health 18:1112. https://doi.org/10.3390/ijerph18031112
Wang S, Tan W, Huang Y, Mao X, Li Z, Zhang X, Wei P, Xue L (2022) Sperm DNA fragmentation measured by sperm chromatin dispersion impacts morphokinetic parameters, fertilization rate and blastocyst quality in ICSI treatments. Zygote 30:72–79. https://doi.org/10.1017/S0967199421000332
Zhang X, Wallace AD, Du P, Kibbe WA, Jafari N, Xie H, Lin S, Baccarelli A, Soares MB, Hou L (2012) DNA methylation alterations in response to pesticide exposure in vitro. Environ Mol Mutagen 53:542–549. https://doi.org/10.1002/em.21718
Acknowledgements
We thank the technicians and medical biologists at Amiens University Medical Center’s reproductive medicine unit for their assistance. And a special thank for Mr. David Fraser for the English correction.
Author information
Authors and Affiliations
Contributions
Marwa Lahimer was in charge of the experimental work, Severine Capelle, Hafida Khorsi-Cauet, and Moncef Benkhalifa wrote the protocol and designed the study. Mounir Ajina and Habib Ben Ali were in charge of the statistical analysis. Elodie Lefranc, Rosalie Cabry, Veronique Bach, Debbie Montjean, and Hafida Khorsi-Cauet provided additional scientific advice. Moncef Benkhalifa provided the research funds. The manuscript was written by Marwa Lahimer, Habib Ben Ali, Moncef Benkhalifa, and Hafida Khorsi-Cauet.
Corresponding author
Ethics declarations
Ethics approval
In line with the French legislation on retrospective studies of laboratory waste and de-identified personal data, approval by institutional review board was neither required nor sought. After treatment, the sperm sample was considered to be laboratory waste.
Consent to participate
In line with the French legislation on studies of laboratory waste, study participants did not have to consent to use of de-identified personal information for research purposes.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Moncef Benkhalifa and Hafida Khorsi-Cauet are co-authors.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lahimer, M., Capelle, S., Lefranc, E. et al. Effect of pesticide exposure on human sperm characteristics, genome integrity, and methylation profile analysis. Environ Sci Pollut Res 30, 77560–77567 (2023). https://doi.org/10.1007/s11356-023-27695-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-27695-7