Abstract
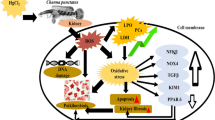
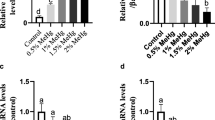
The current study was performed to examine the acute toxicity of mercuric chloride (HgCl2) on the silver carp (Hypophthalmichthys molitrix) larvae. Probit analysis was used to determine the median lethal concentration (LC50). The LC50 values of Hg2+ for the fish larvae at 24, 48, 72, and 96 h were 267.72, 252.97, 225.57, and 97.80 μg/L, respectively. The safe concentration of Hg was 9.78 μg/L for fish larvae. Based on the 96 h LC50, fish were exposed to four different groups including 0, 6.11, 12.23, and 24.45 μg/L for 96 h to assess the effects of different concentrations of Hg2+ on antioxidant capacity, energy metabolism parameters, and related gene expression. The findings revealed that there were no significant differences in the activities of superoxide dismutase (SOD) and total antioxidant capacity (T-AOC) in fish larvae among all the groups (P < 0.05). In the 12.23 μg/L group, fish larvae had a maximum in catalase (CAT) activity. The creatine kinase (CK) activities of fish larvae in control and 6.11 μg/L groups were significantly lower than those groups (P < 0.05). A high concentration of Hg2+ significantly upregulated the mRNA levels of heat shock protein 70 (HSP70) and metallothionein (MT) genes in fish larvae. Furthermore, the IBR index value showed the highest value in the 24.45 μg/L group. Overall, this study provides an increased understanding of the effects of Hg-acute toxicity on silver carp larvae.





Similar content being viewed by others
Data availability
The data and materials that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
References
Ahmad I, Pacheco M, Santos MA (2006) Anguilla L. oxidative stress biomarkers: an in situ study of freshwater wetland ecosystem (Pateira de Fermentelos, Portugal). Chemosphere 65:952–962
Almeida J, Diniz Y, Marques S, Faine L, Ribas B, Burneiko R, Novelli E (2002) The use of the oxidative stress responses as biomarkers in Nile tilapia (Oreochromis niloticus) exposed to in vivo cadmium contamination. Environ Int 27:673–679
Anatone K, Baumann Z, Mason RP, Hansen G, Chernoff B (2020) Century-old mercury pollution: evaluating the impacts on local fish from the eastern United States. Chemosphere 259:127484
Biswal A, Srivastava PP, Pal P, Gupta S, Varghese T, Jayant M (2021) A multi-biomarker approach to evaluate the effect of sodium chloride in alleviating the long-term transportation stress of Labeo rohita fingerlings. Aquaculture 531:735979
Cao L, Liu J, Dou S, Huang W (2020) Biomagnification of methylmercury in a marine food web in Laizhou Bay (North China) and associated potential risks to public health. Mar Pollut Bull 150:110762
Chen Q-L, Sun Y-L, Liu Z-H, Li Y-W (2017) Sex-dependent effects of subacute mercuric chloride exposure on histology, antioxidant status and immune-related gene expression in the liver of adult zebrafish (Danio rerio). Chemosphere 188:1–9
Chen C-Z, Li P, Wang W-B, Li Z-H (2022) Response of growth performance, serum biochemical parameters, antioxidant capacity, and digestive enzyme activity to different feeding strategies in common carp (Cyprinus carpio) under high-temperature stress. Aquaculture 548:737636
Chunfeng FZ, Zhanqiang F (2005) Evaluation on acute toxicity and safety concentration of mercury and selenium to swordtail Fish, Xiphophorus helleri. Environ Sci Technol 28:32–34
Cretì P, Trinchella F, Scudiero R (2010) Heavy metal bioaccumulation and metallothionein content in tissues of the sea bream Sparus aurata from three different fish farming systems. Environ Monit Assess 165:321–329
Crump KL, Trudeau VL (2009) Mercury-induced reproductive impairment in fish. Environ Toxicol Chem 28:895–907
da Silva ETL, Pedreira MM, Dias MLF, Gomes MVT, Soares MA, Pedreira RSF, Schorer M (2021) Mercury chloride toxicity in juveniles Prochilodus argenteus a species from southeastern Brazil. Environ Sci Pollut Res:1–8
Ebrahimpour M, Mosavisefat M, Mohabbati R (2010) Acute toxicity bioassay of mercuric chloride: an alien fish from a river. Toxicol Environ Chem 92:169–173
Hamed HS, Ismal SM, Faggio C (2021) Effect of allicin on antioxidant defense system, and immune response after carbofuran exposure in Nile tilapia, Oreochromis niloticus. Comp Biochem Physiol C: Toxicol Pharmacol 240:108919
Harley J, Lieske C, Bhojwani S, Castellini JM, López JA, O’Hara TM (2015) Mercury and methylmercury distribution in tissues of sculpins from the Bering Sea. Polar Biol 38:1535–1543
Hedayati A, Ghaffari Z (2013) Effect of mercuric chloride on some hematological, biochemical parameters in silver carp ( Hypophthalmichthys molitrix ). Int J Vet Med: Research & Reports 2013:1–11
Hoffman DJ, Rattner BA, Burton GA Jr, Cairns J Jr (2002) Handbook of ecotoxicology. CRC Press
Huang W, Cao L, Ye Z, Yin X, Dou S (2010) Antioxidative responses and bioaccumulation in Japanese flounder larvae and juveniles under chronic mercury exposure. Comp Biochem Physiol C: Toxicol Pharmacol 152:99–106
Kim J-H, Kang J-C (2016) Oxidative stress, neurotoxicity, and metallothionein (MT) gene expression in juvenile rock fish Sebastes schlegelii under the different levels of dietary chromium (Cr6+) exposure. Ecotoxicol Environ Saf 125:78–84
Kim W-K, Lee S-K, Park J-W, Choi K, Cargo J, Schlenk D, Jung J (2014) Integration of multi-level biomarker responses to cadmium and benzo [k] fluoranthene in the pale chub (Zacco platypus). Ecotoxicol Environ Saf 110:121–128
Kong Y, Li M, Shan X, Wang G, Han G (2021) Effects of deltamethrin subacute exposure in snakehead fish, Channa argus: biochemicals, antioxidants and immune responses. Ecotoxicol Environ Saf 209:111821
Li Z-H, Velisek J, Zlabek V, Grabic R, Machova J, Kolarova J, Li P, Randak T (2011) Chronic toxicity of verapamil on juvenile rainbow trout (Oncorhynchus mykiss): effects on morphological indices, hematological parameters and antioxidant responses. J Hazard Mater 185:870–880
Liu H, Zhang H, Zhang S, Wang A, Fu S (2021) Lead exposure induces structural damage, digestive stress, immune response and microbiota dysbiosis in intestine of silver carp (Hypophthalmichthys Molitrix)
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408
Mao L, Liu X, Wang Z, Wang B, Lin C, Xin M, Zhang B-T, Wu T, He M, Ouyang W (2021) Trophic transfer and dietary exposure risk of mercury in aquatic organisms from urbanized coastal ecosystems. Chemosphere 281:130836
Moniruzzaman M, Kumar S, Das D, Sarbajna A, Chakraborty SB (2020) Enzymatic, non enzymatic antioxidants and glucose metabolism enzymes response differently against metal stress in muscles of three fish species depending on different feeding niche. Ecotoxicol Environ Saf 202:110954
Ni H, Peng L, Gao X, Ji H, Ma J, Li Y, Jiang S (2019) Effects of maduramicin on adult zebrafish (Danio rerio): acute toxicity, tissue damage and oxidative stress. Ecotoxicol Environ Saf 168:249–259
O’Brien K, Mueller I, Orczewska J, Dullen K, Ortego M (2014) Hearts of some Antarctic fishes lack mitochondrial creatine kinase. Comp Biochem Physiol A: Mol Integr Physiol 178:30–36
Paul T, Kumar S, Shukla S, Pal P, Kumar K, Poojary N, Biswal A, Mishra A (2020) A multi-biomarker approach using integrated biomarker response to assess the effect of pH on triclosan toxicity in Pangasianodon hypophthalmus (Sauvage, 1878). Environ Pollut 260:114001
Rua-Ibarz A, Bolea-Fernandez E, Maage A, Frantzen S, Sanden M, Vanhaecke F (2019) Tracing mercury pollution along the Norwegian coast via elemental, speciation, and isotopic analysis of liver and muscle tissue of deep-water marine fish (Brosme brosme). Environ Sci Technol 53:1776–1785
Serafini S, de Freitas Souza C, Baldissera MD, Baldisserotto B, Picoli F, Segat JC, Baretta D, da Silva AS (2019) Fish exposed to eprinomectin show hepatic oxidative stress and impairment in enzymes of the phosphotransfer network. Aquaculture 508:199–205
Sfakianakis D, Renieri E, Kentouri M, Tsatsakis A (2015) Effect of heavy metals on fish larvae deformities: a review. Environ Res 137:246–255
Wang M, Wang Y, Zhang L, Wang J, Hong H, Wang D (2013) Quantitative proteomic analysis reveals the mode-of-action for chronic mercury hepatotoxicity to marine medaka (Oryzias melastigma). Aquat Toxicol 130:123–131
Wang Y, Yin D, Xiang Y, Xu Q, Zhang C, Xie Q, Wang D (2019) A review of studies on the biogeochemical behaviors of mercury in the Three Gorges Reservoir, China. Bull Environ Contam Toxicol 102:686–694
Wen B, Jin S-R, Chen Z-Z, Gao J-Z, Liu Y-N, Liu J-H, Feng X-S (2018) Single and combined effects of microplastics and cadmium on the cadmium accumulation, antioxidant defence and innate immunity of the discus fish (Symphysodon aequifasciatus). Environ Pollut 243:462–471
Yearbook CFS (2020) China fishery statistical yearbook. China Agriculture Press, Beijing, China 2020:24–34
Yu X, Khan S, Khan A, Tang Y, Nunes LM, Yan J, Ye X, Li G (2020) Methyl mercury concentrations in seafood collected from Zhoushan Islands, Zhejiang, China, and their potential health risk for the fishing community: capsule: methyl mercury in seafood causes potential health risk. Environ Int 137:105420
Zhang J, Shen H, Wang X, Wu J, Xue Y (2004) Effects of chronic exposure of 2, 4-dichlorophenol on the antioxidant system in liver of freshwater fish Carassius auratus. Chemosphere 55:167–174
Zheng N, Wang S, Dong W, Hua X, Li Y, Song X, Chu Q, Hou S, Li Y (2019) The toxicological effects of mercury exposure in marine fish. Bull Environ Contam Toxicol 102:714–720
Zupo V, Graber G, Kamel S, Plichta V, Granitzer S, Gundacker C, Wittmann KJ (2019) Mercury accumulation in freshwater and marine fish from the wild and from aquaculture ponds. Environ Pollut 255:112975
Funding
This work was financially supported by the National Key Research and Development Program of China (2018YFD0900902, 2018YFD0900905) and the Engineering Research Center of Ecology and Agricultural Use of Wetland, Ministry of Education (Yangtze University) (KFT202006).
Author information
Authors and Affiliations
Contributions
Cheng-Zhuang Chen: writing—original draft preparation; Ya-Jun Wang: sampling and index measurement; Ping Li: writing—reviewing and editing; Ling Liu: data analysis; Yi Chai: culture and sampling; Zhi-Hua Li: conceptualization and overall guidance.
Corresponding author
Ethics declarations
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. Approval of Animal Ethics Committee of Yangtze University was taken.
Consent to participate
Not applicable.
Consent for publication
All the authors are in agreement with the publishment.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Bruno Nunes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, CZ., Chai, Y., Wang, YJ. et al. Physiological and molecular responses in the silver carp (Hypophthalmichthys molitrix) larvae after acute mercury exposure. Environ Sci Pollut Res 30, 49760–49770 (2023). https://doi.org/10.1007/s11356-023-25842-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-25842-8