Abstract
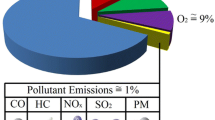
This paper addresses a preliminary study on the emission control of pollutants from an industrial furnace. The stack sampling technique was implemented in this study, and the quantity of pollutants released from the furnace smoke was monitored. The transverse points required to collect the data were selected based on the upstream and downstream disturbances. At first, the investigation was made with the furnace oil. A 15.28% and 5.1% of CO2 and O2 were observed in the flue gas from of regular furnace oil. Hence, by considering the emission level observed, the analysis was repeated with the low sulphur-low stack and the coal tar fuel. The emission of SO2 and N2 was reduced with coal-tar fuel compared with the regular furnace fuel. Thus, the proposed operational adjustment attenuates the pollutant emission, which also helps to reduce the unfavorable impact on the environment. In the future, the authors intend to include a design change in the furnace burner, which would help to increase fuel efficiency and reduce pollution.

Similar content being viewed by others
Data availability
All data associated with this article is provided in the manuscript.
References
Beer JM (2000) Combustion technology developments in power generation in response to environmental challenges. Prog Energy Combust Sci 26(4–6):301–327. https://doi.org/10.1016/S0360-1285(00)00007-1
Bojic M, Mourdoukoutas P (2000) Energy saving does not yield CO2 emissions reductions: the case of waste fuel use in a steel mill. Appl Therm Eng 20(11):963–975. https://doi.org/10.1016/S1359-4311(99)00073-3
Environment and Pollution Control Law Series (2021) Seventh edition, CPCB, MOEFCC. https://cpcb.nic.in/7thEditionPollutionControlLawSeries2021.pdf. Accessed 04 Apr 2022
Fang J, Li G, Aunan K, Vennemo H, Seip HM, Oye KA, Beér JM (2002) A proposed industrial-boiler efficiency program in Shanxi: potential CO2-mitigation, health benefits and associated costs. Appl Energy 71(4):275–285. https://doi.org/10.1016/S0306-2619(02)00013-2
Filipponi M, Rossi F, Presciutti A, De Ciantis S, Castellani B, Carpinelli A (2016) Thermal analysis of an industrial furnace. Energies 9(10):833. https://doi.org/10.3390/en9100833
Hainsworth D, Pourkashanian M, Richardson AP, Rupp JL, Williams A (1996) The influence of carbon dioxide on smoke formation and stability in methane-oxygen-carbon dioxide flames. Fuel 75(3):393–396. https://doi.org/10.1016/0016-2361(95)00265-0
Holmgren K, Henning D (2004) Comparison between material and energy recovery of municipal waste from an energy perspective: a study of two Swedish municipalities. Resour Conserv Recycl 43(1):51–73. https://doi.org/10.1016/j.resconrec.2004.05.001
Hsu CK, Lee CL, Wang CH, Jou CJG (2014) Reduction of energy consumption and pollution emissions for industrial furnace using hydrogen-rich tail gas. Int J Hydrogen Energy 39(18):9675–9680. https://doi.org/10.1016/j.ijhydene.2014.03.195
Hu JL, Kao CH (2007) Efficient energy-saving targets for APEC economies. Energy Policy 35(1):373–382. https://doi.org/10.1016/j.enpol.2005.11.032
Kuprianov VI (2005) Applications of a cost-based method of excess air optimization for the improvement of thermal efficiency and environmental performance of steam boilers. Renew Sustain Energy Rev 9(5):474–498. https://doi.org/10.1016/j.rser.2004.05.006
Lee CL, Jou CJG, Tsai CH, Wang HP (2007) Improvement of a medium-pressure-boiler performance through adjustments on the hydrogen-rich fuel for refinery plant. Fuel 86(5–6):625–631. https://doi.org/10.1016/j.fuel.2006.09.001
Lunghi P, Burzacca R (2004) Energy recovery from industrial waste of a confectionery plant by means of BIGFC plant. Energy 29(12–15):2601–2617. https://doi.org/10.1016/j.energy.2004.05.016
Mayrhofer M, Koller M, Seemann P, Prieler R, Hochenauer C (2021) Assessment of natural gas/hydrogen blends as an alternative fuel for industrial heat treatment furnaces. Int J Hydrogen Energy 46(41):21672–21686. https://doi.org/10.1016/j.ijhydene.2021.03.228
Ptasinski KJ, Koymans MN, Verspagen HHG (2006) Performance of the Dutch energy sector based on energy, exergy and extended exergy accounting. Energy 31(15):3135–3144. https://doi.org/10.1016/j.energy.2006.03.010
Wilk M, Magdziarz A, Kuźnia M, Jerzak W (2010) The reduction of the emission of NOx in the heat-treating furnaces. Metall Foundry Eng 36(1):47–55. https://doi.org/10.7494/mafe.2010.36.1.47
Worrell E, Price L, Martin N (2001) Energy efficiency and carbon dioxide emissions reduction opportunities in the US iron and steel sector. Energy 26(5):513–536. https://doi.org/10.1016/S0360-5442(01)00017-2
Zhang Y, Song X, Xu Y, Shen H, Kong X, Xu H (2019) Utilization of wheat bran for producing activated carbon with high specific surface area via NaOH activation using industrial furnace. J Clean Prod 210:366–375. https://doi.org/10.1016/j.jclepro.2018.11.041
Zhou H, Margenot AJ, Li Y, Si B, Wang T, Zhang Y, Li S, Bhattarai R (2022) Phosphorus pollution control using waste-based adsorbents: Material synthesis, modification, and sustainability. Crit Rev Environ Sci Technol 52(12):2023–2059. https://doi.org/10.1080/10643389.2020.1866414
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. The first draft of the manuscript was written by Varatharajan Prasannavenkadesan. Material preparation, data collection, and analysis were performed by Chockalingam Singaravelu. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Philippe Garrigues
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Model calculation for coal tar fuel
1 kg (carbon) + 8/3 kg = 11/3 kg (carbon dioxide).
1 kg (sulphur) + 1 kg (oxygen) = 2 kg (sulphur dioxide).
Minimum weight of air required for combustion = weight of oxygen required for combustion × 23/100.
Weight of nitrogen present in the fuel = weight of air required for combustion × 77/100.
Air fuel ratio = mass of air flow/mass of fuel flow.
Element | O2 required kg | Dry products |
|---|---|---|
C = 0.9 | 0.9 × [8/3] = 2.4 | 0.9 × [11/3] = 3.3 |
H = 0.06 | 0.06 × 8 = 0.48 | _ |
O2 = 0.025 | _ | _ |
S = 0.005 | 0.005 × [1 × 1] = 0.005 | 0.005 × 2 = 0.01 |
N = 0.01 | _ | 0.01 |
Weight O2 required – weight O2 already present in air = 2.885–0.025 = 2.60 kg.
Minimum weight of air required for combustion = 2.86 × [100/23] = 12.434 kg.
20% of excess air supplies = 12.434 × 1.2 = 14.92.
Weight of N2 present in the fuel = 0.01 kg.
Weight of N2 present in the air = [14.92 × 77]/100 = 11.488 kg.
Total weight of N2 = 11.488 + 0.01 = 11.5 kg.
Weight of free O2 due to excess air = [14.92–12.434] × [23/100] = 0.571 kg.
Calculation for determining % of flue gases
Name | Weight [X] | Molecular weight [Y] | Relative volume Z = (X/Y) m3 | % Volume = [Z/ΣZ]*100 |
|---|---|---|---|---|
CO2 | 3.3 | 44 | 0.075 | 14.91 |
N2 | 11.5 | 28 | 0.410 | 81.51 |
O2 | 0.571 | 32 | 0.0178 | 3.53 |
SO2 | 0.01 | 64 | 0.000156 | 0.00031 |
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Prasannavenkadesan, V., Singaravelu, C. Reduction of pollutant emissions from a heat treatment furnace. Environ Sci Pollut Res (2023). https://doi.org/10.1007/s11356-022-25025-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11356-022-25025-x