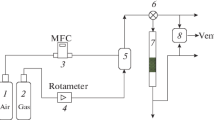
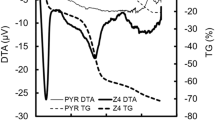
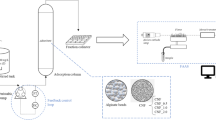
Abstract
A heavy metal ion adsorbent (HFPANF) with high surface area was obtained from polyacrylonitrile fibers with fibrillation and alkali hydrolysis, and an activated carbon fiber filter was prepared by using HFPANF as the binder. The surface area of polyacrylonitrile was 48.64 m2/g due to fibrillation, which also led to the carboxyl content of the HFPANF up to 3.4 mmol/g. Batch adsorption experiments on Cu2+ and Pb2+ showed that the adsorption capacities of HFPANF for Cu2+ and Pb2+ were 47.5 mg/g and 54.3 mg/g. The adsorption kinetics showed that the adsorption reached equilibrium at 90 min and that the adsorption followed the pseudo-second order model. It indicates that the adsorption process is chemisorption. HFPANF formed a single tooth chelate with Cu and a double tooth chelate with Pb. HFPANF-ACF filter was prepared by wet molding technique. When the HFPANF content was 30%, the filter reached a compressive strength of 15.37 MPa and its maximum flux was 180 L/h. 2.5 mg/L of Cu and Pb were used for dynamic adsorption experiments and the heavy metal removal rate was still above 95% after filtering 600 L. The pressure drop of HFPANF-ACF filter was much smaller compared with that of GAC filter due to the combined effect of fibrillated nanofibers and ACF, which can improve the filtration efficiency of the filter.
Graphical Abstract












Similar content being viewed by others
References
Arbab S, Mirbaha H, Zeinolebadi A, Nourpanah P (2014) Indicators for evaluation of progress in thermal stabilization reactions of polyacrylonitrile fibers. J Appl Polym Sci 131:40343. https://doi.org/10.1002/app.40343
Austria HFM, Lecaros R, Hung WS, Tayo LL, Hu CC, Tsai HA (2019) Investigation of salt penetration mechanism in hydrolyzed polyacrylonitrile asymmetric membranes for pervaporation desalination. Desalination 463:32–39. https://doi.org/10.1016/j.desal.2019.04.012
Bardestani R, Patience GS, Kaliaguine S (2019) Experimental methods in chemical engineering: specific surface area and pore size distribution measurements—BET, BJH, and DFT. Can J Chem Eng 97:2781–2791. https://doi.org/10.1002/cjce.23632
Bhunia P, Chatterjee S, Rudra P, De S (2018) Chelating polyacrylonitrile beads for removal of lead and cadmium from wastewater. Sep Purif Technol 193:202–212. https://doi.org/10.1016/j.seppur.2017.11.001
Biesinger MC, Lau LWM, Gerson AR, Smart RSC (2010) Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Sc, Ti, V, Cu and Zn. Appl Surf Sci 257:887–898. https://doi.org/10.1016/j.apsusc.2010.07.086
Bode-aluko CA, Pereao O, Ndayambaje G, Petrik L (2017) Adsorption of toxic metals on modified polyacrylonitrile nanofibres: a review. Water Air Soil Pollut 228(1):1–11. https://doi.org/10.1007/s11270-016-3222-3
Bommi G, Bommi KM (2009) Method for manufacturing carbon blocks. US20090274893
Brédas JL, Salaneck WR (1986) Electronic-structure evolution upon thermal treatment of polyacrylonitrile: a theoretical investigation. J Chem Phys 85(4):2219–2226. https://doi.org/10.1063/1.451116
Chai WS, Cheun JY, Kumar PS, Mubashir M, Show PL (2021) A review on conventional and novel materials towards heavy metal adsorption in wastewater treatment application. J Clean Prod 296:126589. https://doi.org/10.1016/j.jclepro.2021.126589
Chaturvedi A, Bhattacharjee S, Kumar V (2021) A study on the interdependence of heavy metals while contributing to groundwater pollution index. Environ Sci Pollut Res 28:25798–25807. https://doi.org/10.1007/s11356-021-12352-8
Chowdhury S, Mazumder MAJ, Al-Attas O (2016) Heavy metals in drinking water: Occurrences, implications, and future needs in developing countries. Sci Total Environ 569–570:476–488. https://doi.org/10.1016/j.scitotenv.2016.06.166
Dai J, Yan H, Yang H, Cheng RS (2010) Simple method for preparation of chitosan/poly(acrylic acid) blending hydrogel beads and adsorption of copper(II) from aqueous solutions. Chem Eng J 165:240–249. https://doi.org/10.1016/j.cej.2010.09.024
Feng JJ, Cao J, Abbas R, Tao MN (2021) ADMAP-functionalized polyacrylonitrile fiber applied for the efficient identification and removal of metal ions from water. J Chem Educ 98(8):2655–2660. https://doi.org/10.1021/acs.jchemed.0c01418
Geller BE (2002) Status and prospects for development of polyacrylonitrile fibre production. Rev Fiber Chem 34(3):151–161. https://doi.org/10.1023/A:1020525628197
Gopinath A, Kadirvelu K (2018) Strategies to design modified activated carbon fibers for the decontamination of water and air. Environ Chem Lett 16:1137–1168. https://doi.org/10.1007/s10311-018-0740-9
Hussain S, Habib-Ur-Rehman M, Khanam T, Sheer A, Kebin Z, Jianjun Y (2019) Health risk assessment of different heavy metals dissolved in drinking water. Int J Environ Res Public Health 16(10):1737. https://doi.org/10.3390/ijerph16101737
Jiang L, Jia Z, Xu X (2022) Preparation of antimicrobial activated carbon fiber for adsorption. J Porous Mater 29:1071–1081. https://doi.org/10.1007/s10934-022-01225-1
Jin SY, Kim MH, Jeong YG, Yoon YI, Park WH (2017) Effect of alkaline hydrolysis on cyclization reaction of pan nanofibers. Mater Des 124:69–77. https://doi.org/10.1016/j.matdes.2017.03.066
Justi M, de Freita MP, Silla JM (2021) Molecular structure features and fast identification of chemical properties of metal carboxylate complexes by FTIR and partial least square regression. J Mol Struct 1237:130405. https://doi.org/10.1016/j.molstruc.2021.130405
Karas LJ, Wu CH, Das R, Wu JC (2020) Hydrogen bond design principles. WIREs Comput Mol Sci 10:e1477
Khamizov RK (2020) A pseudo-second order kinetic equation for sorption processes. Russ J Phys Chem A 94(1):171–176. https://doi.org/10.1134/S0036024420010148
Kim IC, Yun HG, Lee KH (2002) Preparation of asymmetric polyacrylonitrile membrane with small pore size by phase inversion and post-treatment process. J Membr Sci 199(1–2):75–84. https://doi.org/10.1016/S0376-7388(01)00680-9
Kudryavtsev YV, Krentsel LB, Bondarenko GN, Litmanovich AD, Platé NA, Schapowalow S (2000) Alkaline hydrolysis of polyacrylonitrile,2.On the products welling. Macromol Chem Phys 201:1419–1425. https://doi.org/10.1002/1521-3935(20000801)201:13%3c1419::AID-MACP1419%3e3.0.CO;2-3
Kurniawan TA, Chan GYS, Lo WH, Babel S (2006) Physico–chemical treatment techniques for wastewater laden with heavy metals. Chem Eng J 118(1):83–98. https://doi.org/10.1016/j.cej.2006.01.015
Lai CL, Chao WC, Hung WS, An QF (2015) Physicochemical effects of hydrolyzed asymmetric polyacrylonitrile membrane microstructure on dehydrating butanol. J Membr Sci 490:275–281. https://doi.org/10.1016/j.memsci.2015.05.008
Leal D, Matsuhiro B, Rossi M, Caruso F (2008) FT-IR spectra of alginic acid block fractions in three species of brown seaweeds. Carbohyd Res 343(2):308–316. https://doi.org/10.1016/j.carres.2007.10.016
Li N, Bai RB (2005) Copper adsorption on chitosan–cellulose hydrogel beads: behaviors and mechanisms. Sep Purif Technol 42:237–247. https://doi.org/10.1016/j.seppur.2004.08.002
Li C, Ma HY, Venkateswaran S, Hsiao BS (2019) Highly efficient and sustainable carboxylated cellulose filters for removal of cationic dyes/heavy metals ions. Chem Eng J 389:123458. https://doi.org/10.1016/j.cej.2019.123458
Li XT, Cao J, Zhao YL, Ma N, Tao MN, Zhang WQ (2021) Carbohydrazide modified polyacrylonitrile fiber as efficient and recyclable furfural adsorbent. React Funct Polym 169:105071. https://doi.org/10.1016/j.reactfunctpolym.2021.105071
Li BZ, Zhao J, Lin XX, Tu DG (2022) Highly efficient sunlight-driven self-cleaning electrospun nanofiber membrane NM88B@HPAN for water treatment. J Clean Prod 355:131812. https://doi.org/10.1016/j.jclepro.2022.131812
Liao ZP, Wu YW, Cao SC (2022) Facile in situ decorating polyacrylonitrile membranes using polyoxometalates for enhanced separation performance. J Membr Sci 653:120493. https://doi.org/10.1016/j.memsci.2022.120493
Liu HJ, Yang F, Zheng YM, Kang J, Qu JH (2011) Improvement of metal adsorption onto chitosan/Sargassum sp. composite sorbent by an innovative ion-imprint technology. Water Res 45:145–154. https://doi.org/10.1016/j.watres.2010.08.017
Ma XG et al (2012) Research on heavy metal pollution sudden emergency processing method in water environment. Adv Mater Res 610–613:1682–1685. https://doi.org/10.4028/www.scientific.net/AMR.610-613.1682
Nakamoto K (2006) Infrared and Raman spectra of inorganic and coordination compounds. In Handbook of vibrational spectroscopy (eds J.M. Chalmers and P.R. Griffiths). https://doi.org/10.1002/0470027320.s4104
Naushad M, Vasudevan S, Sharma G, Kumar A, Alothman ZA (2015) Adsorption kinetics, isotherms, and thermodynamic studies for hg2+ adsorption from aqueous medium using alizarin red-s-loaded amberlite ira-400 resin. Desalin Water Treat 57(39):18551–18559. https://doi.org/10.1080/19443994.2015.1090914
Papageorgiou SK, Kouvelos EP, Romanos GE, Sapalidis AA (2010) Metal–carboxylate interactions in metal–alginate complexes studied with FTIR spectroscopy. Carbohyd Res 345:469–473. https://doi.org/10.1016/j.carres.2009.12.010
Patil AS, Boyd GP (1996) Fiber blend for low cost, asbestos free friction material. US, US5508109 A
Philibert M, Zacchi CM, Pottier C, Sacareau D, Baudin I (2020) Mechanical resistance of granular activated carbon media in drinking water treatment. Water Environ J 34:381–389. https://doi.org/10.1111/wej.12535
Qiao XL, Zhang ZJ, Ping ZH (2007) Hydrophilic modification of ultrafiltration membranes and their application in salvia miltiorrhiza decoction. Sep Purif Technol 56(3):265–269. https://doi.org/10.1016/j.seppur.2007.02.007
Rosenzweig S, Sorial GA, Sahle-Demessie E, Luxton T (2015) Study of Cu(ii) chemisorption mechanisms on modified carbon nanotubes based on isotherms, column experiments, and FTIR first derivative analysis. Water Air Soil Pollut 226(7):1–10. https://doi.org/10.1007/s11270-015-2482-7
Sruthi PR, Anas S (2020) An overview of synthetic modification of nitrile group in polymersand applications. Jpolymsc Sci 58:1039–1061. https://doi.org/10.1002/pol.20190190
Sun MK, Gottlieb E, Yuan R, Ghosh S (2020) Polyene-free photoluminescent polymers via hydrothermal hydrolysis of polyacrylonitrile in neutral water. ACS Macro Letters 9(9):1403–1408. https://doi.org/10.1021/acsmacrolett.0c00410
Vedenyapina MD, Kurmysheva AY, Kulaishin SA (2021) Adsorption of heavy metals on activated carbons (a review). Solid Fuel Chem 55:83–104. https://doi.org/10.3103/S0361521921020099
Wang F, Wang X, Jiang Y (2019) Study of adsorption performance and adsorption mechanism for U(VI) ion on modified polyacrylonitrile fibers. J Radioanal Nucl Chem 323:365–377. https://doi.org/10.1007/s10967-019-06928-5
Wee JH, Bae Y, Ahn H (2022) Fibrous and granular activated carbon mixed media for effective gas removal as a cabin air filter. Carbon Lett 32:1111–1118. https://doi.org/10.1007/s42823-022-00345-7
Wei BX, Li HY, Tian YQ, Xu XM, Jin ZG (2015) Thermal degradation behavior of hypochlorite-oxidized starch nanocrystals under different oxidized levels. Carbohyd Polym 124:124–130. https://doi.org/10.1016/j.carbpol.2015.01.081
Xu GM, Guan WJ, Sun XN (2020) Filtration performance and application of activated carbon fiber enhanced microfibrous entrapped sorbent (ACF-MFES). Russ J Phys Chem 94:182–188. https://doi.org/10.1134/S0036024420010070
Xu K, Deng J, Lin R, Zhang H (2020) Surface fibrillation of para-aramid nonwoven as a multi-functional air filter with ultralow pressure drop. J Mater Chem A 8(42):22269–22279
Yue ZR, Mangun C, Economy J, Kemme P, Cropek D, Maloney S (2001) Removal of chemical contaminants from water to below USEPA MCL using fiber glass supported activated carbon filters. Environ Sci Technol 35(13):2844–2848. https://doi.org/10.1021/es001858r
Yue YY, Hou KY, Chen JY (2022) Ag/AgBr/AgVO3 Photocatalyst-embedded polyacrylonitrile/polyamide/chitosan nanofiltration membrane for integrated filtration and degradation of RhB. ACS Appl Mater Interfaces 14(21):24708–24719. https://doi.org/10.1021/acsami.2c04988
Zhao R, Li X, Sun BL, Shen MQ, Tan XC, Ding Y (2015) Preparation of phosphorylated polyacrylonitrile-based nanofiber mat and its application for heavy metal ion removal. Chem Eng J 268:290–299. https://doi.org/10.1016/j.cej.2015.01.061
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design, material preparation, data collection and analysis, writing & editing of the manuscript were performed by Yuanhuo Ji; review and editing of the manuscript were performed by Xiwen Wang. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This work is original and has not been published elsewhere in any form or language. The manuscript has been submitted to this journal solely for consideration.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Guilherme L. Dotto.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Fibrillation increases the surface area and degree of alkaline hydrolysis of PANF.
• Micronanofibers and carboxyl improve the compressive strength of filter.
• High carboxyl content of the filter can effectively remove heavy metal ions.
• 3D structure of micronanofibers reduce the pressure drop of the filter.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ji, Y., Wang, X. Purification performance of modified polyacrylonitrile fiber–activated carbon fiber filter for heavy metal ions. Environ Sci Pollut Res 30, 23372–23385 (2023). https://doi.org/10.1007/s11356-022-23833-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23833-9