Abstract
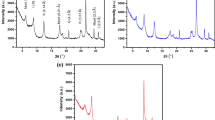
The present study demonstrates the synthesis of eco-friendly metal oxide-clay composites (MgO-clay and CaO-clay) with phytochemical functionalization. The physical and chemical properties of prepared composites were characterized using standard techniques viz. scanning electron microscopy, Fourier transform infrared spectroscopy, and X-ray diffraction. The effect of pH on the dye adsorption capability of the synthesized composites was studied. The adsorption of an anionic dye methyl orange (MO) and a cationic due methylene blue (MB) was favored in the acidic and basic regions, respectively. The Taguchi design approach was adopted for the removal of MO and MB from wastewater using the synthesized composites. The obtained results suggest that initial dye concentration and composite dosage were the most influential parameters in dye removal among all the studied parameters. The adsorption experiments were carried out using MgO-clay and CaO-clay composites with the optimum conditions obtained from Taguchi optimization to validate the predicted response. The experimental parameters viz. the effect of contact time, initial dye concentration, and solution temperature were studied for screened composite (CaO-clay) with optimized conditions. The obtained results were interpreted using standard isotherms and kinetic models. A maximum adsorption capacity of 571 ± 10 and 859 ± 14 mg g−1 was obtained from the Langmuir adsorption isotherm for MO and MB, respectively. Regeneration studies suggested that the CaO-clay composite can be utilized up to 3 cycles with reduced adsorption capacity of the dyes over cycles due to the solid binding nature of dyes on the CaO-clay composite. The fresh and utilized CaO-clay composite were tested for their environmental toxicity analysis using ecologically important soil microorganisms. The obtained results suggested no detrimental effects on soil microbe’s functionality, indicating their threat-free disposal in the soil environment.













Similar content being viewed by others
Data availability
The data will be available on request.
References
Abdallah Y, Ogunyemi SO, Abdelazez A, Zhang M, Hong X, Ibrahim E, Hossain A, Fouad H, Li B, Chen J (2019) The green synthesis of MgO nano-flowers using Rosmarinus officinalis L. (Rosemary) and the antibacterial activities against Xanthomonasoryzae pv. oryzae. Biomed Res Int 2019:1–8. https://doi.org/10.1155/2019/5620989
Adhikari S, Mandal S, Sarkar D, Kim DH, Madras G (2017) Kinetics and mechanism of dye adsorption on WO3 nanoparticles. Appl Surf Sci 420:472–482. https://doi.org/10.1016/j.apsusc.2017.05.191
Adomavičiūtė-Grabusovė S, Ramanavičius S, Popov A, Šablinskas V, Gogotsi O, Ramanavičius A (2021) Selective enhancement of SERS spectral bands of salicylic acid adsorbate on 2D Ti3C2Tx-based MXene film. Chemosensors 9:223. https://doi.org/10.3390/chemosensors9080223
Aldalbahi A, Thamer BM, Rahaman M, El-Newehy MH (2020) Self-nitrogen-doped nanoporous carbons derived from poly(1,5-diaminonaphthalene) for the removal of toxic dye pollutants from wastewater: non-linear isotherm and kinetic analysis. Polymers 12:2563. https://doi.org/10.3390/polym12112563
Amari A, Mohammed Alzahrani F, MohammedsalehKatubi K, Salem Alsaiari N, Tahoon MA, Ben Rebah F (2021) Clay-polymer nanocomposites: preparations and utilization for pollutants removal. Materials 14:1365. https://doi.org/10.3390/ma14061365
Ameen F, Alsamhary K, Alabdullatif JA, ALNadhari S (2021) A review on metal-based nanoparticles and their toxicity to beneficial soil bacteria and fungi. Ecotoxicol Environ Saf 213:112027. https://doi.org/10.1016/j.ecoenv.2021.112027
Amuda OS, Giwa AA, Bello IA (2007) Removal of heavy metal from industrial wastewater using modified activated coconut shell carbon. Biochem Eng J 36:174–181. https://doi.org/10.1016/j.bej.2007.02.013
Baspinar MS, Demir I, Orhan M (2010) Utilization potential of silica fume in fired clay bricks. Waste Manag Res J Sustain Circ Econ 28:149–157. https://doi.org/10.1177/0734242X09104385
Bauer AW (1959) Single-disk antibiotic-sensitivity testing of staphylococci. AMA Arch Internal Med 104:208. https://doi.org/10.1001/archinte.1959.00270080034004
Chikri R, Elhadiri N, Benchanaa M, El maguana Y (2020) Efficiency of sawdust as low-cost adsorbent for dyes removal. J Chem 2020:1–17. https://doi.org/10.1155/2020/8813420
CLSI (Clinical and Laboratory Standard Institute) (2012) Methods for dilution antimicrobial susceptibility test for bacteria that grow aerobically; approved standard, 9th edition, CLSI document MO7-A9, Clinical and Laboratory Standards Institute, Wayne
Crini G (2005) Recent developments in polysaccharide-based materials used as adsorbents in wastewater treatment. Prog Polym Sci 30:38–70. https://doi.org/10.1016/j.progpolymsci.2004.11.002
Dada AO (2012) Langmuir, Freundlich, Temkin and Dubinin-Radushkevich isotherms studies of equilibrium sorption of Zn2+ unto phosphoric acid modified rice husk. IOSR J Appl Chem 3:38–45. https://doi.org/10.9790/5736-0313845
Daneshvar E, Vazirzadeh A, Niazi A, Kousha M, Naushad M, Bhatnagar A (2017) Desorption of methylene blue dye from brown macroalga: effects of operating parameters, isotherm study and kinetic modeling. J Clean Prod 152:443–453. https://doi.org/10.1016/j.jclepro.2017.03.119
Deng H, Lu J, Li G, Zhang G, Wang X (2011) Adsorption of methylene blue on adsorbent materials produced from cotton stalk. Chem Eng J 172:326–334. https://doi.org/10.1016/j.cej.2011.06.013
Dogdu OG, Tunacan T, Dikmen E (2019) Removal of indigo dye by photocatalysis process using Taguchi experimental design. Environ Res Technol 2:19–25. https://doi.org/10.35208/ert.457739
Đolić M, Karanac M, Radovanović D, Umićević A, Kapidžić A, Veličković Z, Marinković A, Kamberović Ž (2021) Closing the loop: As(V) adsorption onto goethite impregnated coal-combustion fly ash as integral building materials. J Clean Prod 303:126924. https://doi.org/10.1016/j.jclepro.2021.126924
Dubey RS, Rajesh YBRD, More MA (2015) Synthesis and characterization of SiO2 nanoparticles via sol-gel method for industrial applications. Mater Today Proc 2:3575–3579. https://doi.org/10.1016/j.matpr.2015.07.098
Fatiqin A, Amrulloh H, Simanjuntak W (2021) Green synthesis of MgO nanoparticles using Moringa oleifera leaf aqueous extract for antibacterial activity. Bull Chem Soc Ethiop 35:161–170. https://doi.org/10.4314/bcse.v35i1.14
Femi-Adepoju AG, Dada AO, Otun KO, Adepoju AO, Fatoba OP (2019) Green synthesis of silver nanoparticles using terrestrial fern (Gleichenia Pectinata (Willd.) C. Presl.): characterization and antimicrobial studies. Heliyon 5:e01543. https://doi.org/10.1016/j.heliyon.2019.e01543
Ferro-García MA, Rivera-Utrilla J, Rodríguez-Gordillo J, Bautista-Toledo I (1988) Adsorption of zinc, cadmium, and copper on activated carbons obtained from agricultural by-products. Carbon 26:363–373. https://doi.org/10.1016/0008-6223(88)90228-X
Gong R, Ye J, Dai W, Yan X, Hu J, Hu X, Li S, Huang H (2013) Adsorptive removal of methyl orange and methylene blue from aqueous solution with finger-citron-residue-based activated carbon. Ind Eng Chem Res 52:14297–14303. https://doi.org/10.1021/ie402138w
Gu S, Kang X, Wang L, Lichtfouse E, Wang C (2019) Clay mineral adsorbents for heavy metal removal from wastewater: a review. Environ Chem Lett 17:629–654. https://doi.org/10.1007/s10311-018-0813-9
Guo H, Ke Y, Wang D, Lin K, Shen R, Chen J, Weng W (2013) Efficient adsorption and photocatalytic degradation of Congo red onto hydrothermally synthesized NiS nanoparticles. J Nanopart Res 15:1475. https://doi.org/10.1007/s11051-013-1475-y
Gupta S, Babu BV (2009a) Utilization of waste product (tamarind seeds) for the removal of Cr(VI) from aqueous solutions: equilibrium, kinetics, and regeneration studies. J Environ Manage 90:3013–3022. https://doi.org/10.1016/j.jenvman.2009.04.006
Gupta S, Babu BV (2009b) Removal of toxic metal Cr(VI) from aqueous solutions using sawdust as adsorbent: equilibrium, kinetics and regeneration studies. Chem Eng J 150:352–365. https://doi.org/10.1016/j.cej.2009.01.013
Heidarizad M, Şengör SS (2016) Synthesis of graphene oxide/magnesium oxide nanocomposites with high-rate adsorption of methylene blue. J Mol Liq 224:607–617. https://doi.org/10.1016/j.molliq.2016.09.049
Hsieh L-L, Kang H-J, Shyu H-L, Chang C-Y (2009) Optimal degradation of dye wastewater by ultrasound/Fenton method in the presence of nanoscale iron. Water Sci Technol A J Int Assoc Water Pollut Res 60:1295–1301. https://doi.org/10.2166/wst.2009.366
Jain N, Alam P, Laskar IR, Panwar J (2015) ‘Aggregation induced phosphorescence’ active iridium(iii) complexes for integrated sensing and inhibition of bacterial growth in aqueous solution. RSC Adv 5:61983–61988. https://doi.org/10.1039/C5RA10161A
Jitjamnong J, Luengnaruemitchai A, Samanwonga N, Chuaykarn N (2019) Biodiesel production from canola oil and methanol using ba impregnated calcium oxide with microwave irradiation-assistance. Chiang Mai J Sci 46:987–1000
Karri RR, Tanzifi M, Tavakkoli Yaraki M, Sahu JN (2018) Optimization and modeling of methyl orange adsorption onto polyaniline nano-adsorbent through response surface methodology and differential evolution embedded neural network. J Environ Manage 223:517–529. https://doi.org/10.1016/j.jenvman.2018.06.027
Kumar A, Yeshwanth M, Kumar K, Panwar J, Gupta S (2022) Functionalized Cu-based metal oxide nanoparticles with enhanced Cd+2 adsorption capacity and their ecotoxicity assessment by molecular docking. J Environ Manage 307:114523. https://doi.org/10.1016/j.jenvman.2022.114523
Kumar K A, Verma G, Raghuvanshi S, Gupta S (2021) Defluoridation studies using graphene oxidebased nanoadsorbents. In: Green chemistry and water remediation: Research and Applications. Elsevier, pp 35–57. https://doi.org/10.1016/B978-0-12-817742-6.00002-5
Mahmoud HR, Ibrahim SM, El-Molla SA (2016) Textile dye removal from aqueous solutions using cheap MgO nanomaterials: adsorption kinetics, isotherm studies and thermodynamics. Adv Powder Technol 27:223–231. https://doi.org/10.1016/j.apt.2015.12.006
Meshkatalsadat MH, Zahedifar M, Pouramiri B (2022) Facile green synthesis of CaO NPs using the Crataegus pontica C.Koch extract for photo-degradation of MB dye. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-022-19671-4
Moodley JS, Krishna SB, Pillay K, Govender P (2018) Green synthesis of silver nanoparticles from Moringa oleifera leaf extracts and its antimicrobial potential. Adv Nat Sci Nanosci Nanotechnol 9:015011. https://doi.org/10.1088/2043-6254/aaabb2
Mouni L, Belkhiri L, Bollinger JC, Bouzaza A, Assadi A, Tirri A, Dahmoune F, Madani K, Remini H (2018) Removal of Methylene Blue from aqueous solutions by adsorption on Kaolin: kinetic and equilibrium studies. Appl Clay Sci 153:38–45. https://doi.org/10.1016/j.clay.2017.11.034
Mylarappa M, Raghavendra N, Surendra BS, Kumar KS, Kantharjau S (2022) Electrochemical, photocatalytic and sensor studies of clay/MgO nanoparticles. Appl Surf Sci Adv 10:100268. https://doi.org/10.1016/j.apsadv.2022.100268
Pradhan SK, Panwar J, Gupta S (2017) Enhanced heavy metal removal using silver-yttrium oxide nanocomposites as novel adsorbent system. J Environ Chem Eng 5:5801–5814. https://doi.org/10.1016/j.jece.2017.11.007
Pradhan SK, Pareek V, Panwar J, Gupta S (2019) Synthesis and characterization of ecofriendly silver nanoparticles combined with yttrium oxide (Ag-Y2O3) nanocomposite with assorted adsorption capacity for Cu(II) and Cr(VI) removal: a mechanism perspective. J Water Process Eng 32:100917. https://doi.org/10.1016/j.jwpe.2019.100917
Ramanavicius S, Ramanavicius A (2020) Progress and insights in the application of MXenes as new 2D nano-materials suitable for biosensors and biofuel cell design. Int J Mol Sci 21:9224. https://doi.org/10.3390/ijms21239224
Ratnam MV, Karthikeyan C, Rao KN, Meena V (2020) Magnesium oxide nanoparticles for effective photocatalytic degradation of methyl red dye in aqueous solutions: optimization studies using response surface methodology. Mater Today Proc 26:2308–2313. https://doi.org/10.1016/j.matpr.2020.02.498
Senthil Kumar P, Fernando PS, Ahmed RT, Srinath R, Priyadharshini M, Vignesh AM, Thanjiappan A (2014) Effect of temperature on the adsorption of methylene blue dye onto sulfuric acid–treated orange peel. Chem Eng Commun 201:1526–1547. https://doi.org/10.1080/00986445.2013.819352
Schlich K, Beule L, Hund-Rinke K (2016) Single versus repeated applications of CuO and Ag nanomaterials and their effect on soil microflora. Environ Pollut 215:322–330. https://doi.org/10.1016/j.envpol.2016.05.028
Sharma BK, Mehta BR, Chaudhari VP, Shah EV, Mondal Roy S, Roy DR (2022) Green synthesis of dense rock MgO nanoparticles using carica papaya leaf extract and its shape dependent antimicrobial activity: Joint Experimental and DFT Investigation. J Cluster Sci 33:1667–1675. https://doi.org/10.1007/s10876-021-02090-9
Sharma J, Sharma M, Basu S (2017) Synthesis of mesoporous MgO nanostructures using mixed surfactants template for enhanced adsorption and antimicrobial activity. J Environ Chem Eng 5:3429–3438. https://doi.org/10.1016/j.jece.2017.07.015
Tsibranska I, Hristova E (2011) Comparison of different kinetic models for adsorption of heavy metals onto activated carbon from apricot stones. Bul Chem Commun 43:370–377
Unlü N, Ersoz M (2006) Adsorption characteristics of heavy metal ions onto a low cost biopolymeric sorbent from aqueous solutions. J Hazard Mater 136:272–280. https://doi.org/10.1016/j.jhazmat.2005.12.013
Voelker D, Schlich K, Hohndorf L, Koch W, Kuehnen U, Polleichtner C, Kussatz C, Hund-Rinke K (2015) Approach on environmental risk assessment of nanosilver released from textiles. Environ Res 140:661–672. https://doi.org/10.1016/j.envres.2015.05.011
Wong S, Ghafar NA, Ngadi N, Razmi FA, Inuwa IM, Mat R, Amin NA (2020) Effective removal of anionic textile dyes using adsorbent synthesized from coffee waste. Sci Rep 10:2928. https://doi.org/10.1038/s41598-020-60021-6
Yusuff AS, Ajayi OA, Popoola LT (2021) Application of Taguchi design approach to parametric optimization of adsorption of crystal violet dye by activated carbon from poultry litter. Scientific African 13:e00850. https://doi.org/10.1016/j.sciaf.2021.e00850
Zhang L, Hu P, Wang J, Liu Q, Huang R (2015) Adsorption of methyl orange (MO) by Zr (IV)-immobilized cross-linked chitosan/bentonite composite. Int J Biol Macromol 81:818–827. https://doi.org/10.1016/j.ijbiomac.2015.09.017
Acknowledgements
The authors thank the Birla Institute of Technology and Science, Pilani, for providing experimental facilities.
Author information
Authors and Affiliations
Contributions
Anil Kumar K: conceptualization, synthesis, characterization, batch studies, data presentation and interpretation, methodology, writing—original document. Shobham: antimicrobial studies, writing. Jitendra Panwar: conceptualization, supervision, reviewing, and editing. Suresh Gupta: conceptualization, supervision, reviewing, and editing.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: George Z. Kyzas
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
K, A.K., Shobham, Panwar, J. et al. One-pot synthesis of metal oxide-clay composite for the evaluation of dye removal studies: Taguchi optimization of parameters and environmental toxicity studies. Environ Sci Pollut Res 30, 61541–61561 (2023). https://doi.org/10.1007/s11356-022-23752-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23752-9