Abstract
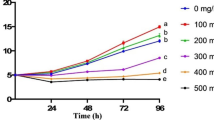
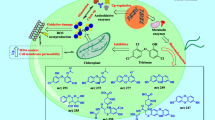
Although the addition of triclosan (TCS) in consumer products has been strictly restricted, its continuous applications in hospitals and other medical facilities and its numerous residues still pose a potential risk to aquatic organisms and aquatic ecosystems. In this study, we investigated the growth, biochemical alterations, and physiological responses of Chlorella vulgaris exposed to different concentrations of TCS. The potential toxicity mechanisms associated with excessive production of reactive oxygen species (ROS) and disruption of photosynthetic system II (PSII) were also analyzed. The results indicated that the growth, cellular ultrastructure, and physiology of C. vulgaris were severely affected by TCS in a dose-effect dependent manner. TCS inhibited the growth of C. vulgaris, leading to mitochondria enlargement, the disordering of the arrangement of thylakoids, cell wall rupture, organelles loss, and the cytoplasm lysis. TCS induced severe oxidative damage characterized by ROS accumulation, elevated malondialdehyde (MDA), and up-regulation of antioxidant enzyme activities. Moreover, in TCS-induced algal cells, the main sites of ROS accumulation were chloroplasts, mitochondria, and cell membranes, with ROS accumulating most in the mitochondria. In addition, TCS caused damage to the reaction center (RC inactivation), donor side (OEC damage), and accepted side (electron transport from QA to QB) of PSII in C. vulgaris, leading to inhibition of photosynthetic activity. These results could provide novel insights into the mechanisms of TCS-induced ROS accumulation and photosynthetic inhibition in C. vulgaris, which would contribute to a deep understanding of TCS toxicity on algae.






Similar content being viewed by others
Data availability
The data are available from the corresponding author on reasonable request.
References
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Asada K (2006) Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol 141:391–396. https://doi.org/10.1104/pp.106.082040
Asadi P, Rad HA, Qaderi F (2019) Comparison of Chlorella vulgaris and Chlorella sorokiniana pa.91 in post treatment of dairy wastewater treatment plant effluents. Environ Sci Pollut Res 26:29473–29489. https://doi.org/10.1007/s11356-019-06051-8
Bai X, Acharya K (2017) Algae-mediated removal of selected pharmaceutical and personal care products (PPCPs) from Lake Mead water. Sci Total Environ 581-582:734–740. https://doi.org/10.1016/j.scitotenv.2016.12.192
Bedoux G, Roig B, Thomas O, Dupont V, Le Bot B (2012) Occurrence and toxicity of antimicrobial triclosan and by-products in the environment. Environ Sci Pollut Res 19:1044–1065. https://doi.org/10.1007/s11356-011-0632-z
Bi R, Zeng X, Mu L, Hou L, Liu W, Li P, Chen H, Li D, Bouchez A, Tang J, Xie L (2018) Sensitivities of seven algal species to triclosan, fluoxetine and their mixtures. Sci Rep 8:15361. https://doi.org/10.1038/s41598-018-33785-1
Caverzan A, Casassola A, Brammer SP (2016) Antioxidant responses of wheat plants under stress. Genet Mol Biol 39:1–6. https://doi.org/10.1590/1678-4685-GMB-2015-0109
Chalifour A, Tam NFY (2016) Tolerance of cyanobacteria to the toxicity of BDE-47 and their removal ability. Chemosphere 164:451–461. https://doi.org/10.1016/j.chemosphere.2016.08.109
Che X, Ding R, Li Y, Zhang Z, Gao H, Wang W (2018) Mechanism of long-term toxicity of CuO NPs to microalgae. Nanotoxicology 12:923–939. https://doi.org/10.1080/17435390.2018.1498928
Chen S, Xu X, Dai X, Yang C, Qiang S (2007) Identification of tenuazonic acid as a novel type of natural photosystem II inhibitor binding in QB-site of Chlamydomonas reinhardtii. Biochim Biophys Acta 1767:306–318. https://doi.org/10.1016/j.bbabio.2007.02.007
Cortez FS, Pereira CDS, Santos AR, Cesar A, Choueri RB, Martini GA, Bohrer-Morel MB (2012) Biological effects of environmentally relevant concentrations of the pharmaceutical Triclosan in the marine mussel Perna perna (Linnaeus, 1758). Environ Pollut 168:145–150. https://doi.org/10.1016/j.envpol.2012.04.024
Cronin MTD, Netzeva TI, Dearden JC, Edwards R, Worgan ADP (2004) Assessment and modeling of the toxicity of organic chemicals to Chlorella vulgaris: development of a novel database. Chem Res Toxicol 17:545–554. https://doi.org/10.1021/tx0342518
Dai Z, Luo X, Yang A, Wang J, Fu H, Wu Y (2021) The effects of triclosan on physiological and photosynthetic characteristics of Chlorella vulgaris. Water 13:1355. https://doi.org/10.3390/w13101355
Dann AB, Hontela A (2011) Triclosan: environmental exposure, toxicity and mechanisms of action. J Appl Toxicol 31:285–311. https://doi.org/10.1002/jat.1660
Deblois CP, Dufresne K, Juneau P (2013) Response to variable light intensity in photoacclimated algae and cyanobacteria exposed to atrazine. Aquat Toxicol 126:77–84. https://doi.org/10.1016/j.aquatox.2012.09.005
Dhillon GS, Kaur S, Pulicharla R, Brar SK, Cledón M, Verma M, Surampalli R (2015) Triclosan: current status, occurrence, environmental risks and bioaccumulation potential. Int J Environ Res Public Health 12:5657–5684. https://doi.org/10.3390/ijerph120505657
Ding T, Lin K, Bao L, Yang M, Li J, Yang B, Gan J (2018) Biouptake, toxicity and biotransformation of triclosan in diatom Cymbella sp. and the influence of humic acid. Environ Pollut 234:231–242. https://doi.org/10.1016/j.envpol.2017.11.051
European Comission (2016) Commission implementing decision (EU) 2016/110 of 27 January 2016 not approving triclosan as an existing active substance for use in biocidal products for product type 1. European Union OJ L 21:86–87
Food and Drug Administration (US-FDA), HHS (2017) Safety and effectiveness of health care antiseptics; topical antimicrobial drug products for over-the-counter human use. Final Rule Fed Regist 82(242):60474–60503
Gao K, Wu Y, Li G, Wu H, Villafañe VE, Helbling EW (2007) Solar UV radiation drives CO2 fixation in marine phytoplankton: a double-edged sword. Plant Physiol 144:54–59. https://doi.org/10.1104/pp.107.098491
Ghazizadeh V, Nazıroğlu M (2014) Electromagnetic radiation (Wi-Fi) and epilepsy induce calcium entry and apoptosis through activation of TRPV1 channel in hippocampus and dorsal root ganglion of rats. Metab Brain Dis 29:787–799. https://doi.org/10.1007/s11011-014-9549-9
González-Pleiter M, Rioboo C, Reguera M, Abreu I, Leganés F, Cid Á, Fernández-Piñas F (2017) Calcium mediates the cellular response of Chlamydomonas reinhardtii to the emerging aquatic pollutant triclosan. Aquat Toxicol 186:50–66. https://doi.org/10.1016/j.aquatox.2017.02.021
Hakala M, Tuominen I, Keränen M, Tyystjärvi T, Tyystjärvi E (2005) Evidence for the role of the oxygen-evolving manganese complex in photoinhibition of Photosystem II. Biochim Biophys Acta - Biogenergetics 1706:68–80. https://doi.org/10.1016/j.bbabio.2004.09.001
Ishibashi H, Matsumura N, Hirano M, Matsuoka M, Shiratsuchi H, Ishibashi Y, Takao Y, Arizono K (2004) Effects of triclosan on the early life stages and reproduction of medaka Oryzias latipes and induction of hepatic vitellogenin. Aquat Toxicol 67:167–179. https://doi.org/10.1016/j.aquatox.2003.12.005
Jachero L, Ahumada I, Fuentes E, Richter P (2016) Decreases in the bioconcentration of triclosan in wheat plants according to increasing amounts of biosolids added to soil. Geoderma 276:19–25. https://doi.org/10.1016/j.geoderma.2016.04.014100173
Ji X, Cheng J, Gong D, Zhao X, Qi Y, Su Y, Ma W (2018) The effect of NaCl stress on photosynthetic efficiency and lipid production in freshwater microalga-Scenedesmus obliquus XJ002. Sci Total Environ 633:593–599. https://doi.org/10.1016/j.scitotenv.2018.03.240
Kolpin DW, Furlong ET, Meyer MT, Thurman EM, Zaugg SD, Barber LB, Buxton HT (2002) Pharmaceuticals, hormones, and other organic wastewater contaminants in U.S. streams, 1999-2000: a national reconnaissance. Environ Sci Technol 36:1202–1211. https://doi.org/10.1021/es011055j
Larsen C, Zhu HY, Flick R, Passeport E (2019) Mechanisms of pharmaceutical and personal care product removal in algae-based wastewater treatment systems. Sci Total Environ 695:133772. https://doi.org/10.1016/j.scitotenv.2019.133772
Latifi A, Ruiz M, Zhang C (2009) Oxidative stress in cyanobacteria. FEMS Microbiol Rev 33:258–278. https://doi.org/10.1111/j.1574-6976.2008.00134.x
Li F, Liang Z, Zheng X, Zhao W, Wu M, Wang Z (2015) Toxicity of nano-TiO2 on algae and the site of reactive oxygen species production. Aquat Toxicol 158:1–13. https://doi.org/10.1016/j.aquatox.2014.10.014
Li S, Wang P, Zhang C, Zhou X, Yin Z, Hu T, Hu D, Liu C, Zhu L (2020) Influence of polystyrene microplastics on the growth, photosynthetic efficiency and aggregation of freshwater microalgae Chlamydomonas reinhardtii. Sci Total Environ 714:136767. https://doi.org/10.1016/j.scitotenv.2020.136767
Lindström A, Buerge IJ, Poiger T, Bergqvist PA, Müller MD, Buser HR (2002) Occurrence and environmental behavior of the bactericide triclosan and its methyl derivative in surface waters and in wastewater. Environ Sci Technol 36:2322–2329. https://doi.org/10.1021/es0114254
Liu H, Xia Y, Cai W, Zhang Y, Zhang X, Du S (2017) Enantioselective oxidative stress and oxidative damage caused by Rac- and S-metolachlor to Scenedesmus obliquus. Chemosphere 173:22–30. https://doi.org/10.1016/j.chemosphere.2017.01.028
Liu Q, Tang X, Zhang X, Yang Y, Sun Z, Jian X, Zhao Y, Zhang X (2020) Evaluation of the toxic response induced by BDE-47 in a marine alga, Phaeodactylum tricornutum, based on photosynthesis-related parameters. Aquat Toxicol 227:105588. https://doi.org/10.1016/j.aquatox.2020.105588
Lu Z, Zhang Y, Gao Y, Liu B, Sun X, He F, Zhou Q, Wu Z (2016) Effects of pyrogallic acid on Microcystis aeruginosa: oxidative stress related toxicity. Ecotoxicol Environ Saf 132:413–419. https://doi.org/10.1016/j.ecoenv.2016.06.039
Machado MD, Soares EV (2021) Toxicological effects induced by the biocide triclosan on Pseudokirchneriella subcapitata. Aquat Toxicol 230:105706. https://doi.org/10.1016/j.aquatox.2020.105706
Macri D (2017) Worldwide use of triclosan: can dentistry do without this antimicrobial? Contemp Clin Dent 8:7–8. https://doi.org/10.4103/ccd.ccd_225_17
Mihaich E, Capdevielle M, Urbach-Ross D, Slezak B (2017) Hypothesis-driven weight-of-evidence analysis of endocrine disruption potential: a case study with triclosan. Crit Rev Toxicol 47:1–26. https://doi.org/10.1080/10408444.2016.1269722
Montaseri H, Forbes P (2016) A review of monitoring methods for triclosan and its occurrence in aquatic environments. Trac Trends. Anal Chem 85:221–231. https://doi.org/10.1016/j.trac.2016.09.010
Nassef M, Sang GK, Seki M, Kang IJ, Hano T, Shimasaki Y, Oshima Y (2010) In ovo nanoinjection of triclosan, diclofenac and carbamazepine affects embryonic development of medaka fish (Oryzias latipes). Chemosphere 79:966–973. https://doi.org/10.1016/j.chemosphere.2010.02.002
Oukarroum A, Bussotti F, Goltsev V, Kalaji HM (2015) Correlation between reactive oxygen species production and photochemistry of photosystems I and II in Lemna gibba L. plants under salt stress. Environ Exp Bot 109:80–88. https://doi.org/10.1016/j.envexpbot.2014.08.005
Ozaki K, Ito E, Tanabe S, Natsume K, Tsuji K, Harada K (2009) Electron microscopic study on lysis of a cyanobacterium Microcystis. J Health Sci 55:578–585. https://doi.org/10.1248/jhs.55.578
Pan C, Peng F, Shi W, Hu L, Wei X, Ying G (2018) Triclosan-induced transcriptional and biochemical alterations in the freshwater green algae Chlamydomonas reinhardtii. Ecotoxicol Environ Saf 148:393–401. https://doi.org/10.1016/j.ecoenv.2017.10.011
Peng X, Yu Y, Tang C, Tan J, Huang Q, Wang Z (2008) Occurrence of steroid estrogens, endocrine-disrupting phenols, and acid pharmaceutical residues in urban riverine water of the Pearl River Delta, South China. Sci Total Environ 397:158–166. https://doi.org/10.1016/j.scitotenv.2008.02.059
Poole AC, Pischel L, Ley C, Suh G, Goodrich JK, Haggerty TD, Ley RE, Parsonnet J (2016) Crossover control study of the effect of personal care products containing triclosan on the microbiome. mSphere 1:e00056–e00015. https://doi.org/10.1128/mSphere.00056-15
Rodricks JV, Swenberg JA, Borzelleca JF, Maronpot RR, Shipp AM (2010) Triclosan: a critical review of the experimental data and development of margins of safety for consumer products. Crit Rev Toxicol 40:422–484. https://doi.org/10.3109/10408441003667514
Scientific Committee on Consumer Safety (2010) Opinion on triclosan antimicrobial resistance. European Union, Brussels. https://doi.org/10.2772/11162
Singh A, Kumar A, Yadav S, Singh KI (2019) Reactive oxygen species-mediated signaling during abiotic stress. Plant Gene 18:100173. https://doi.org/10.1016/j.plgene.2019.100173
Strasser BJ, Strasser RJ (1995) Measuring fast fluorescence transients to address environmental questions: the JIP-Test. In: Mathis P (ed) Photosynthesis: from light to biosphere. KAP Press, Dordrecht, pp 977–980. https://doi.org/10.1007/978-94-009-0173-5_1142
Sun C, Xu Y, Hu N, Ma J, Sun S, Cao W, Klobučar G, Hu C, Zhao Y (2020) To evaluate the toxicity of atrazine on the freshwater microalgae Chlorella sp. using sensitive indices indicated by photosynthetic parameters. Chemosphere 244:125514. https://doi.org/10.1016/j.chemosphere.2019.125514
Taştan BE, Tekinay T, Çelik HS, Özdemir C, Cakir DN (2017) Toxicity assessment of pesticide triclosan by aquatic organisms and degradation studies. Regul Toxicol Pharmacol 91:208–215. https://doi.org/10.1016/j.yrtph.2017.10.030
Tato T, Salgueiro-González N, León VM, González S, Beiras R (2018) Ecotoxicological evaluation of the risk posed by bisphenol A, triclosan, and 4-nonylphenol in coastal waters using early life stages of marine organisms (Isochrysis galbana, Mytilus galloprovincialis, Paracentrotus lividus, and Acartia clausi). Environ Pollut 232:173–182. https://doi.org/10.1016/j.envpol.2017.09.031
Wang J, Zhu H, Liu X, Liu Z (2014) Oxidative stress and Ca2+ signals involved on cadmium-Induced apoptosis in rat hepatocyte. Biol Trace Elem Res 161:180–189. https://doi.org/10.1007/s12011-014-0105-6
Wang S, Poon K, Cai Z (2018) Removal and metabolism of triclosan by three different microalgal species in aquatic environment. J Hazard Mater 342:643–650. https://doi.org/10.1016/j.jhazmat.2017.09.004
Wang J, Zhou W, Chen H, Zhan J, He C, Wang Q (2019) Ammonium nitrogen tolerant Chlorella strain screening and its damaging effects on photosynthesis. Front Microbiol 9:3250. https://doi.org/10.3389/fmicb.2018.03250
Wang XD, Lu YC, Xiong XH, Yuan Y, Lu LX, Liu YJ, Mao JH, Xiao WW (2020) Toxicological responses, bioaccumulation, and metabolic fate of triclosan in Chlamydomonas reinhardtii. Environ Sci Pollut Res 27:11246–11259. https://doi.org/10.1007/s11356-020-07704-9
Xin X, Huang G, An C, Raina-Fulton R, Weger H (2019) Insights into long-term toxicity of triclosan to freshwater green algae in Lake Erie. Environ Sci Technol 53:2189–2198. https://doi.org/10.1021/acs.est.9b00259
Xu J, Li M, Mak NK, Chen F, Jiang Y (2011) Triphenyltin induced growth inhibition and antioxidative responses in the green microalga Scenedesmus quadricauda. Ecotoxicology 20:73–80. https://doi.org/10.1007/s10646-010-0557-1
Xu D, Su C, Song X, Shi Q, Fu J, Hu L, Xia X, Song E, Song Y (2015) Polychlorinated biphenyl quinone induces endoplasmic reticulum stress, unfolded protein response, and calcium release. Chem Res Toxicol 28:1326–1337. https://doi.org/10.1021/acs.chemrestox.5b00124
Zhang S, Benoit G (2019) Comparative physiological tolerance of unicellular and colonial Microcystis aeruginosa to extract from Acorus calamus rhizome. Aquat Toxicol 215:105271. https://doi.org/10.1016/j.aquatox.2019.105271
Zhang X, Ma F, Zhu X, Zhu J, Rong J, Zhan J, Chen H, He C, Wang Q (2017) The acceptor side of photosystem II is the initial target of nitrite stress in Synechocystis sp. PCC 6803. Appl Environ Microbiol 83:e02952–e02916. https://doi.org/10.1128/AEM.02952-16
Zhang X, Chen H, Wang H, Wang Q (2021) Time-course effects of Tris(1,3-dichloro-2-propyl) phosphate (TDCPP) on Chlorella pyrenoidosa: Growth inhibition and adaptability mechanisms. J Hazard Mater 402:123784. https://doi.org/10.1016/j.jhazmat.2020.123784
Zorrilla LM, Gibson EK, Jeffay SC, Crofton KM, Setzer WR, Cooper RL, Stoker TE (2009) The effects of triclosan on puberty and thyroid hormones in male Wistar rats. Toxicol Sci 107:56–64. https://doi.org/10.1093/toxsci/kfn225
Funding
This work was supported jointly by grants from the National Natural Science Fund of China (51468066), the project of Key Laboratory of Resources Conversion and Pollution Control of the State Ethnic Affairs Commission (PTZD22006), and the Fundamental Research Funds for the Central Universities, South-Central University for Nationalities (CZY20030).
Author information
Authors and Affiliations
Contributions
Jin Yan: investigation, methodology. Ying Zou: investigation, methodology. Fengrui Zhang: methodology, data curation. Xinyue Huang: formal analysis, writing — revising and editing. Shenghua Zhang: conceptualization, supervision, funding acquisition, writing — original draft preparation. Gaboury Benoit: writing — revising and editing.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent to participate
Not applicable
Consent for publication
All authors have given consent for publication
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Bruno Nunes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 387 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yan, J., Zou, Y., Zhang, F. et al. Growth, ROS accumulation site, and photosynthesis inhibition mechanism of Chlorella vulgaris by triclosan. Environ Sci Pollut Res 30, 12125–12137 (2023). https://doi.org/10.1007/s11356-022-23009-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-23009-5