Abstract
Endocrine disrupting chemicals (EDCs) may affect many biological processes like growth and stress response. Bisphenol A (BPA) is a plasticizer that is used to harden plastics and polycarbonates. Phthalates are used to add flexibility to polyvinyl chloride containing plastics. The main metabolite of di(2-ethylhexyl) phthalate (DEHP) is mono(2-ethylhexyl) phthalate (MEHP) and it is even more toxic than the parent compound. Humans are usually exposed to these chemicals in mixtures by different routes starting from fetal period. However, there are not many studies in literature that investigate the combined effects of these chemicals. The aim of this study is to investigate toxic effects of BPA and/or MEHP on HepG2 cell line. We have evaluated cytotoxicity, cytomorphological, apoptotic changes, oxidative stress, oxidant/antioxidant status alterations, and endoplasmic reticulum (ER) stress. Combined exposure to BPA and MEHP caused alterations in oxidant/antioxidant status and ER stress marker proteins in both cytoplasmic and nuclear cellular fractions. We can suggest that combined exposure to EDCs may cause serious toxicological outcomes and more mechanistic studies are needed to determine the combined toxic effects.
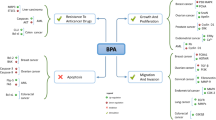
Graphical abstract










Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this manuscript.
References
Akerboom TP, Sies H (1981) Assay of glutathione, glutathione disulfide, and glutathione mixed disulfides in biological samples. Methods Enzymol 771:373–382
Arbuckle TE, Davis K, Boylan K et al (2016) Bisphenol A, phthalates and lead and learning and behavioral problems in Canadian children 6–11 years of age: CHMS 2007–2009. Neurotoxicology 54:89–98
Asahi J, Kamo H, Baba R, Doi Y et al (2010) Bisphenol A induces endoplasmic reticulum stress-associated apoptosis in mouse non-parenchymal hepatocytes. Life Sci 87:431–438. https://doi.org/10.1016/j.lfs.2010.08.007
Ateş N, Yildirim Ö, Tamer L et al (2004) Plasma catalase activity and malondialdehyde level in patients with cataract. Eye 18:785–788. https://doi.org/10.1038/sj.eye.6700718
Axelsson J, Rylander L, Rignell-Hydbom A, Lindh CH et al (2015) Prenatal phthalate exposure and reproductive function in young men. Environ Res 138:264–270. https://doi.org/10.1016/j.envres.2015.02.024
Bai J, He Z, Li Y et al (2019) Mono-2-ethylhexyl phthalate induces the expression of genes involved in fatty acid synthesis in HepG2 cells. Environ Toxicol Pharmacol 69:104–111. https://doi.org/10.1016/j.etap.2019.04.004
Balci A, Ozkemahli G, Erkekoglu P et al (2020) Histopathologic, apoptotic and autophagic, effects of prenatal bisphenol A and/or di(2-ethylhexyl) phthalate exposure on prepubertal rat testis. Environ Sci Pollut Res Int 27:20104–20116. https://doi.org/10.1007/s11356-020-08274-6
Bekö G, Callesen M, Weschler CJ et al (2015) Phthalate exposure through different pathways and allergic sensitization in preschool children with asthma, allergic rhinoconjunctivitis and atopic dermatitis. Environmental Res 137:432–439. https://doi.org/10.1016/j.envres.2015.01.012
Berlett BS, Stadtman ER (1997) Protein oxidation in aging, disease, and oxidative stress. J Biol Chem 272:20313–20316. https://doi.org/10.1074/jbc.272.33.20313
Biswas SK, Rahman I (2009) Environmental toxicity, redox signaling and lung inflammation: the role of glutathione. Mos Asp Med 30:60–76. https://doi.org/10.1016/j.mam.2008.07.001
Cajaraville MP, Ortiz-Zarragoitia M (2006) Specificity of the peroxisome proliferation response in mussels exposed to environmental pollutants. Aquat Tox 78:S117–S123. https://doi.org/10.1016/j.aquatox.2006.02.016
Chao MW, Erkekoglu P, Tseng CY et al (2014) Intracellular generation of ROS by 3,5-dimethylaminophenol: persistence, cellular response, and impact of molecular toxicity. Toxicol Sci 141:300–313. https://doi.org/10.1093/toxsci/kfu127
Chen X, Wang J, Qin Q et al (2012) Mono-2-ethylhexyl phthalate induced loss of mitochondrial membrane potential and activation of Caspase3 in HepG2 cells. Environ Tox Pharmacol 33:421–430. https://doi.org/10.1016/j.etap.2012.02.001
Choi S, Park SY, Jeong J, Cho E, Phark S, Lee M, Kwak D, Lim JY, Jung WW, Sul D (2010) Identification of toxicological biomarkers of di(2-ethylhexyl) phthalate in proteins secreted by HepG2 cells using proteomic analysis. Proteomics 10(9):1831–1846. https://doi.org/10.1002/pmic.200900674
Colborn T (2004) Neurodevelopment and endocrine disruption. Environ Health Perspect 112:944–949. https://doi.org/10.1289/ehp.6601
Davies MJ (2016) Protein oxidation and peroxidation. Biochem J 473:805–825. https://doi.org/10.1042/BJ20151227
Environmental Protection Agency (EPA) (2021) What is endocrine disruption? https://www.epa.gov/endocrine-disruption/what-endocrine-disruption#main-content. Accessed 2 March 2021
Erkekoglu P, Rachidi W, De Rosa V et al (2010a) Protective effect of selenium supplementation on the genotoxicity of di(2-ethylhexyl)phthalate and mono(2-ethylhexyl)phthalate treatment in LNCaP cells. Free Rad Biol Med 49:559–566. https://doi.org/10.1016/j.freeradbiomed.2010.04.038
Erkekoglu P, Rachidi W, Yuzugullu OG et al (2010b) Evaluation of cytotoxicity and oxidative DNA damaging effects of di(2-ethylhexyl)-phthalate (DEHP) and mono(2-ethylhexyl)-phthalate (MEHP) on MA-10 Leydig cells and protection by selenium. Toxicol Appl Pharmacol 248:52–62. https://doi.org/10.1016/j.taap.2010.07.016
Erkekoglu P, Rachidi W, Yuzugullu OG, Giray B, Favier A, Ozturk M, Hincal F (2010c) Evaluation of cytotoxicity and oxidative DNA damaging effects of di(2-ethylhexyl) phthalate (DEHP) and mono(2-ethylhexyl)-phthalate (MEHP) on MA-10 Leydig cells and protection by selenium. Toxicol Appl Pharmacol 248(1):52–62. https://doi.org/10.1016/j.taap.2010.07.016
Erkekoglu P, Rachidi W, Yüzügüllü OG et al (2011) Induction of ROS, p53, p21 in DEHP- and MEHP-exposed LNCaP cells-protection by selenium compounds. Food Chem Tox 49(7):1565–1571. https://doi.org/10.1016/j.fct.2011.04.001
Erkekoglu P, Zeybek ND, Giray B et al (2012) The effects of di(2-ethylhexyl)phthalate exposure and selenium nutrition on sertoli cell vimentin structure and germ-cell apoptosis in rat testis. Arch Environ Contam Toxicol 62(3):539–547
Fic A, Žegura B, SollnerDolenc M et al (2013) Mutagenicity and DNA damage of bisphenol A and its structural analogues in HepG2 cells. Arch Ind Hyg Eviron Toxicol 64:189–200. https://doi.org/10.2478/10004-1254-64-2013-2319
Flohé L, Gunzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–121. https://doi.org/10.1016/S0076-6879(84)05015-1
Forcic D, Branovic-Cakanic K, Ivancic J, Jug R, Barut M, Strancar A (2005) Purification of genomic DNA by short monolithic columns. J Chromatogr A 1065:115–120. https://doi.org/10.1016/j.chroma.2004.10.030
Gascon M, Casas M, Morales E et al (2015) Prenatal exposure to bisphenol A and phthalates and childhood respiratory tract infections and allergy. J Allergy Clin Immunol 135:370–378. https://doi.org/10.1016/j.jaci.2014.09.030
Gassman NR (2017) Induction of oxidative stress by bisphenol A and its pleiotropic effects. Environ Mol Mutagen 58:60–71. https://doi.org/10.1002/em.22072
Geens T, Aerts D, Berthot C et al (2012) A review of dietary and non-dietary exposure to bisphenol-A. Food Chem Toxicol 50:3725–3740. https://doi.org/10.1016/j.fct.2012.07.059
Gualtieri AF, Iwachow MA, Venara M et al (2011) Bisphenol A effect on glutathione synthesis and recycling in testicular Sertoli cells. J Endocrinol Invest 34:e102–e109. https://doi.org/10.1007/BF03347468
Guo Y, Wu Q, Kannan K (2011) Phthalate metabolites in urine from China, and implications for human exposures. Environ Int 37:893–898. https://doi.org/10.1016/j.envint.2011.03.005
Harley KG, Gunier RB, Kogut K et al (2013) Prenatal and early childhood bisphenol A concentrations and behavior in school-aged children. Environ Res 126:43–50
Hempel SL, Buettner GR, O’Malley YQ et al (1999) Dihydrofluorescein diacetate is superior for detecting intracellular oxidants: comparison with 2′,7′-dichlorodihydrofluorescein diacetate, 5(and 6)-carboxy-2′,7′-dichlorodihydrofluorescein diacetate, and dihydrorhodamine 123. Free Radic Biol Med 27:146–159
Hines CJ, Hopf NB, Deddens JA et al (2011) Estimated daily intake of phthalates in occupationally exposed groups. J Exp Sci Environ Epimiol 21:133–141. https://doi.org/10.1038/jes.2009.62
Huc L, Lemarié A, Guéraud F et al (2012) Low concentrations of bisphenol A induce lipid accumulation mediated by the production of reactive oxygen species in the mitochondria of HepG2 cells. Toxicol in Vitro 26:709–717. https://doi.org/10.1016/j.tiv.2012.03.017
Ipapo KN, Factor-Litvak P, Whyatt RM et al (2017) Maternal prenatal urinary phthalate metabolite concentrations and visual recognition memory among infants at 27 weeks. Environ Res 155:7–14
Karihtala P, Soini Y (2007) Reactive oxygen species and antioxidant mechanisms in human tissues and their relation to malignancies. APMIS 115:81–103. https://doi.org/10.1111/j.1600-0463.2007.apm_514.x
Kim S, Mun GI, Choi E et al (2018) Submicromolar bisphenol A induces proliferation and DNA damage in human hepatocyte cell lines in vitro and in juvenile rats in vivo. Food Chem Tox 111:125–132. https://doi.org/10.1016/j.fct.2017.11.010
Krieg RC, Dong Y, Schwamborn K et al (2005) Protein quantification and its tolerance for different interfering reagents using the BCA-method with regard to 2D SDS PAGE. J Biochem Biophys Methods 65:13–19. https://doi.org/10.1016/j.jbbm.2005.08.005
Kumar P, Nagarajan A, Uchil PD (2018) Analysis of cell viability by the MTT assay. Cold Spring Harb Protoc 2018(6). https://doi.org/10.1101/pdb.prot095505
Li J, Zhang W, Zhou P et al (2021) Selenium deficiency induced apoptosis via mitochondrial pathway caused by oxidative stress in porcine gastric tissues. Res Vet Sci 144:142–148. https://doi.org/10.1016/j.rvsc.2021.10.017
Li X, Yin P, Zhao L (2017) Effects of individual and combined toxicity of bisphenol A, dibutyl phthalate and cadmium on oxidative stress and genotoxicity in HepG2 cells. Food Chem Tox 105:73–81. https://doi.org/10.1016/j.fct.2017.03.054
Lyu ZQ, Xie X, Ke YB (2016) Effects of long term and low dose Di- (2-ethylhexyl) phthalate exposure on global genome DNA methylation in HePG2 cells. Zhonghua Lao Dong Wei Sheng Zhi Ye Bing Za Zhi 34(5):346–51
Maćczak A, Cyrkler M, Bukowska B et al (2017) Bisphenol A, bisphenol S, bisphenol F and bisphenol AF induce different oxidative stress and damage in human red blood cells (in vitro study). Toxicol in Vitro 41:143–149. https://doi.org/10.1016/j.tiv.2017.02.018
Marnett LJ (2000) Oxyradicals and DNA damage. Carcinogenesis 21:361–370. https://doi.org/10.1093/carcin/21.3.361
Meli R, Monnolo A, Annunziata C et al (2020) Oxidative stress and BPA toxicity: an antioxidant approach for male and female reproductive dysfunction. Antioxidants 9(5):405. https://doi.org/10.3390/antiox9050405
Melnick RL (2001) Is peroxisome proliferation an obligatory precursor step in the carcinogenicity of di(2-ethylhexyl)phthalate (DEHP)? Environ Health Perspect 109:437–442. https://doi.org/10.1289/ehp.01109437
Nakagawa T, Zhu H, Morishima N et al (2000) Caspase-12 mediates endoplasmic-reticulum-specific apoptosis and cytotoxicity by amyloid-beta. Nature 403:98–103. https://doi.org/10.1038/47513
Ooe H, Taira T, Iguchi-Ariga SM et al (2005) Induction of reactive oxygen species by bisphenol A and abrogation of bisphenol A-induced cell injury by DJ-1. Toxicol Sci 88:114–126. https://doi.org/10.1093/toxsci/kfi278
Ozkemahli G, Ozyurt AB, Erkekoglu P et al (2022) The effects of prenatal and lactational bisphenol A and/or di(2-ethylhexyl) phthalate exposure on female reproductive system. Toxicol Mech Methods 23:1–15. https://doi.org/10.1080/15376516.2022.2057265
Penna A, Cahalan M (2007) Western blotting using the Invitrogen NuPage Novex Bis Tris minigels. J vis Exp 7:264. https://doi.org/10.3791/264
Perera F, Roen Nolte EL, Wang Y et al (2016) Bisphenol A exposure and symptoms of anxiety and depression among inner city children at 10–12 years of age. Environ Res 151:195–202
Peropadre A, Fernández Freire P, Pérez Martín JM et al (2015) Endoplasmic reticulum stress as a novel cellular response to di (2-ethylhexyl) phthalate exposure. Toxicol in Vitro 30:281–287. https://doi.org/10.1016/j.tiv.2015.10.009
Pompella A, Visvikis A, Paolicchi A et al (2003) The changing faces of glutathione, a cellular protagonist. Biochem Pharmacol 66:1499–1503. https://doi.org/10.1016/s0006-2952(03)00504-5
Richard MJ, Portal B, Meo J et al (1992) Malondialdehyde kit evaluated for determining plasma and lipoprotein fractions that react with thiobarbituric acid. Clin Chem 38:704–709
Rudel RA, Camann DE, Spengler JD et al (2003) Phthalates, alkylphenols, pesticides, polybrominated diphenyl ethers, and other endocrine-disrupting compounds in indoor air and dust. Environ Sci Tech 37(20):4543–4553. https://doi.org/10.1021/es0264596
Rusyn I, Kadiiska MB, Dikalova A et al (2001) Phthalates rapidly increase production of reactive oxygen species in vivo: role of Kupffer cells. Mol Pharmacol 59:744–750. https://doi.org/10.1124/mol.59.4.744
Rutkowski DT, Kaufman RJ (2004) A trip to the ER: coping with stress. Trend Cell Biol 14(1):20–28. https://doi.org/10.1016/j.tcb.2003.11.001
Shacter E (2000) Quantification and significance of protein oxidation in biological samples. Drug Met Rev 32:307–326. https://doi.org/10.1081/dmr-100102336
Smit LA, Lenters V, Høyer BB et al (2015) Prenatal exposure to environmental chemical contaminants and asthma and eczema in school-age children. Allergy 70:653–660. https://doi.org/10.1111/all.12605
Sobarzo CM, Rosana N, Livia L et al (2015) Mono-(2-ethylhexyl) phthalate (MEHP) affects intercellular junctions of Sertoli cell: a potential role of oxidative stress. Rep Toxicol 58:203–212. https://doi.org/10.1016/j.reprotox.2015.10.010
Stojanoska MM, Milosevic N, Milic N et al (2017) The influence of phthalates and bisphenol A on the obesity development and glucose metabolism disorders. Endocrine 55:666–681. https://doi.org/10.1007/s12020-016-1158-4
Sun X, Lin Y, Huang Q et al (2015) Di(2-ethylhexyl) phthalate-induced apoptosis in rat INS-1 cells is dependent on activation of endoplasmic reticulum stress and suppression of antioxidant protection. J Cell Mol Med 19(3):581–594. https://doi.org/10.1111/jcmm.12409
Tan AS, Berridge MV (2000) Superoxide produced by activated neutrophils efficiently reduces the tetrazolium salt, WST-1 to produce a soluble formazan: a simple colorimetric assay for measuring respiratory burst activation and for screening anti-inflammatory agents. J Immunol Methods 238(1–2):59–68. https://doi.org/10.1016/s0022-1759(00)00156-3
Tetz LM, Cheng AA, Korte CS et al (2013) Mono-2-ethylhexyl phthalate induces oxidative stress responses in human placental cells in vitro. Toxicol Appl Pharmacol 268:47–54. https://doi.org/10.1016/j.taap.2013.01.020
Trasande L, Spanier AJ, Sathyanarayana S et al (2013) Urinary phthalates and increased insulin resistance in adolescents. Pediatrics 132(3):e646–e655. https://doi.org/10.1542/peds.2012-4022
Vrachnis N, Loukas N, Vrachnis D et al (2021) A systematic review of bisphenol a from dietary and non-dietary sources during pregnancy and its possible connection with fetal growth restriction: investigating its potential effects and the window of fetal vulnerability. Nutrients 13:2426. https://doi.org/10.3390/nu13072426
Wang W, Craig ZR, Basavarajappa MS et al (2012a) Di (2-ethylhexyl) phthalate inhibits growth of mouse ovarian antral follicles through an oxidative stress pathway. Toxicol Appl Pharmacol 258:288–295. https://doi.org/10.1016/j.taap.2011.11.008
Wang W, Craig ZR, Basavarajappa MS et al (2012b) Mono-(2-ethylhexyl) phthalate induces oxidative stress and inhibits growth of mouse ovarian antral follicles. Biol Rep 87:152. https://doi.org/10.1095/biolreprod.112.102467
Wittassek M, Angerer J (2008) Phthalates: metabolism and exposure. Int J Androl 31(2):131–138. https://doi.org/10.1111/j.1365-2605.2007.00837.x
Xin F, Jiang LX et al (2014) Bisphenol A induces oxidative stress-associated DNA damage in INS-1 cells. Mutat Res Gen Toxicol Environ Mutagen 769:29–33. https://doi.org/10.1016/j.mrgentox.2014.04.019
Yang G, Zhou X, Wang J et al (2012) MEHP-induced oxidative DNA damage and apoptosis in HepG2 cells correlates with p53-mediated mitochondria-dependent signaling pathway. Food Chem Tox 50:2424–2431. https://doi.org/10.1016/j.fct.2012.04.023
Yin L, Dai Y, Cui Z et al (2017) The regulation of cellular apoptosis by the ROS-triggered PERK/EIF2α/chop pathway plays a vital role in bisphenol A-induced male reproductive toxicity. Toxicol Appl Pharmacol 314:98–108. https://doi.org/10.1016/j.taap.2016.11.013
Zeybek ND, Sur Ü, Oflaz O et al (2020) Renal changes and apoptosis caused by subacute exposure to Aroclor 1254 in selenium-deficient and selenium-supplemented rats. Arh Hig Rada Toksikol 71:110–120. https://doi.org/10.2478/aiht-2020-71-3360
Zhang G, Ling X, Liu K et al (2016a) The p-eIF2α/ATF4 pathway links endoplasmic reticulum stress to autophagy following the production of reactive oxygen species in mouse spermatocyte-derived cells exposed to dibutyl phthalate. Free Rad Res 50:698–707. https://doi.org/10.3109/10715762.2016.1169403
Zhang G, Liu K, Ling X et al (2016b) DBP-induced endoplasmic reticulum stress in male germ cells causes autophagy, which has a cytoprotective role against apoptosis in vitro and in vivo. Toxicol Lett 245:86–98. https://doi.org/10.1016/j.toxlet.2016.01.016
Zhang Y, Han L, Yang H et al (2017) Bisphenol A affects cell viability involved in autophagy and apoptosis in goat testis Sertoli cell. Environ Toxicol Pharmacol 55:137–147. https://doi.org/10.1016/j.etap.2017.07.014
Zhao Y, Chen L, Li LX et al (2014) Gender-specific relationship between prenatal exposure to phthalates and intrauterine growth restriction. Pediatr Res 76:401–408. https://doi.org/10.1038/pr.2014.103
Funding
This study was supported by Hacettepe Research Fund Project No. TSA-2017–12882.
Author information
Authors and Affiliations
Contributions
GO: conceptualization, formal analysis, investigation, methodology, writing, original draft preparation, visualization, validation. PE: conceptualization, formal analysis, methodology, writing—review and editing, project co-administration. AE: formal analysis, visualization. NDZ: formal analysis, visualization. NY: formal analysis, visualization. BKG: conceptualization, methodology, writing—review and editing, project administration, data curation, supervision.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests. This study is the PhD thesis (Evaluation of the effects of combined exposure to endocrine disruptor chemical substances in HEPG2 cell line) of Gizem Özkemahlı (Thesis No. 499175).
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlight
• We determined cytotoxic, oxidant and ER-stress causing effects of BPA and/or MEHP.
• BPA and/or MEHP exposure caused oxidative stress in HepG2 cells.
• Oxidative stress in by the combined exposure was higher compared to other groups.
• Combined exposure to BPA and MEHP may lead to cellular membrane damage.
• Endoplasmic reticulum stress was mainly induced in the combined exposure group.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ozkemahli, G., Erkekoglu, P., Ercan, A. et al. Effects of single or combined exposure to bisphenol A and mono(2-ethylhexyl)phthalate on oxidant/antioxidant status, endoplasmic reticulum stress, and apoptosis in HepG2 cell line. Environ Sci Pollut Res 30, 12189–12206 (2023). https://doi.org/10.1007/s11356-022-22937-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22937-6