Abstract
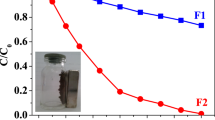
Two carbon dots (CD) with diameters of 4.9 ± 1.5 and 4.1 ± 1.2 nm were successfully synthesized through an acid ablation route with HNO3 or H2SO4, respectively, using Ilex paraguariensis as raw material. The CD were used to produce magnetite-containing nanocomposites through two different routes: hydrothermal and in situ. A thorough characterization of the particles by transmission electron microscopy (TEM), X-ray diffraction (XRD), thermogravimetric analysis (TGA), dynamic light scattering (DLS), Fourier transform infrared (FTIR), and X-ray photoelectron spectroscopy (XPS) indicates that all nanomaterials have spherical-like morphology with a core–shell structure. The composition of this structure depends on the route used: with the hydrothermal route, the shell is composed of the CD, but with the in situ process, the CD act as nucleation centers, and so the iron oxide domains are in the shell. Regarding the photocatalytic mechanism for the degradation of methyl orange, the interaction between the CD and the magnetite plays an important role in the photo-Fenton reaction at pH 6.2, in which ligand-to-metal charge transfer processes (LTMCT) allow Fe2+ regeneration. All materials (100 ppm) showed catalytic activity in the elimination of methyl orange (8.5 ppm), achieving discoloration of up to 98% under visible irradiation over 400 nm in 7 h. This opens very interesting possibilities for the use of agro-industrial residues for sustainable synthesis of catalytic nanomaterials, and the role of the interaction of iron-based catalysts with organic matter in heterogeneous Fenton-based processes.












Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Aggarwal R, Saini D, Singh B et al (2020) Bitter apple peel derived photoactive carbon dots for the sunlight induced photocatalytic degradation of crystal violet dye. Sol Energy 197:326–331. https://doi.org/10.1016/j.solener.2020.01.010
Ali I, Hasan N, Ahmad M, Khan Z (2018) Biosynthesis of iron nanoparticles using Trigonella foenum-graecum seed extract for photocatalytic methyl orange dye degradation and antibacterial applications. J Photochem Photobiol B Biol 183:154–163. https://doi.org/10.1016/j.jphotobiol.2018.04.014
Amaliyah S, Pangesti DP, Masruri M et al (2020) Green synthesis and characterization of copper nanoparticles using Piper retrofractum Vahl extract as bioreductor and capping agent. Heliyon 6:e04636. https://doi.org/10.1016/j.heliyon.2020.e04636
Andreozzi R, Caprio V, Insola A, Marotta R (1999) Advanced oxidation processes ( AOP ) for water purification and recovery. Catal Today 53:51–59
Arzate S, Pfister S, Oberschelp C, Sánchez-Pérez JA (2019) Environmental impacts of an advanced oxidation process as tertiary treatment in a wastewater treatment plant. Sci Total Environ 694https://doi.org/10.1016/j.scitotenv.2019.07.378
Balan V, Petrache IA, Popa MI, et al (2012) Biotinylated chitosan-based SPIONs with potential in blood-contacting applications. J Nanoparticle Res 14https://doi.org/10.1007/s11051-012-0730-y
Bandi R, Gangapuram BR, Dadigala R et al (2016) Facile and green synthesis of fluorescent carbon dots from onion waste and their potential applications as sensor and multicolour imaging agents. RSC Adv 6:28633–28639. https://doi.org/10.1039/c6ra01669c
Brillas E, Martínez-Huitle CA (2015) Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods. An updated review. Appl Catal B Environ 166–167:603–643. https://doi.org/10.1016/j.apcatb.2014.11.016
Bronstein LM, Huang X, Retrum J et al (2007) Influence of iron oleate complex structure on iron oxide nanoparticle formation. Chem Mater 19:3624–3632
Carvalho SSF, Carvalho NMF (2017) Dye degradation by green heterogeneous Fenton catalysts prepared in presence of Camellia sinensis. J Environ Manage 187:82–88. https://doi.org/10.1016/j.jenvman.2016.11.032
Chen WH, Kuo PC (2010) A study on torrefaction of various biomass materials and its impact on lignocellulosic structure simulated by a thermogravimetry. Energy 35:2580–2586. https://doi.org/10.1016/j.energy.2010.02.054
Chernyshova IV, Hochella MF, Madden AS (2007) Size-dependent structural transformations of hematite nanoparticles. 1. Phase transition. Phys Chem Chem Phys 9:1736–1750. https://doi.org/10.1039/b618790k
Das R, Bandyopadhyay R, Pramanik P (2018) Carbon quantum dots from natural resource: A review. Mater Today Chem 8:96–109. https://doi.org/10.1016/j.mtchem.2018.03.003
Dhand V, Soumya L, Bharadwaj S et al (2016) Green synthesis of silver nanoparticles using Coffea arabica seed extract and its antibacterial activity. Mater Sci Eng 58:36–43. https://doi.org/10.1016/j.msec.2015.08.018
Donadelli JA, Carlos L, Arques A, García Einschlag FS (2018) Kinetic and mechanistic analysis of azo dyes decolorization by ZVI-assisted Fenton systems: pH-dependent shift in the contributions of reductive and oxidative transformation pathways. Appl Catal B Environ 231:51–61. https://doi.org/10.1016/j.apcatb.2018.02.057
Fayazi M (2021) Preparation and characterization of carbon nanotubes/pyrite nanocomposite for degradation of methylene blue by a heterogeneous Fenton reaction. J Taiwan Inst Chem Eng 120:229–235. https://doi.org/10.1016/j.jtice.2021.03.033
Fayazi M, Ghanei-Motlagh M (2020) Electrochemical mineralization of methylene blue dye using electro-Fenton oxidation catalyzed by a novel sepiolite/pyrite nanocomposite. Int J Environ Sci Technol 17:4541–4548. https://doi.org/10.1007/s13762-020-02749-2
Fayazi M, Taher MA, Afzali D, Mostafavi A (2016) Enhanced Fenton-like degradation of methylene blue by magnetically activated carbon/hydrogen peroxide with hydroxylamine as Fenton enhancer. J Mol Liq 216:781–787. https://doi.org/10.1016/j.molliq.2016.01.093
García FE, Senn AM, Meichtry JM et al (2019) Iron-based nanoparticles prepared from yerba mate extract. Synthesis, characterization and use on chromium removal. J Environ Manage 235:1–8. https://doi.org/10.1016/j.jenvman.2019.01.002
Gholinejad M, Zareh F, Nájera C (2018) Nitro group reduction and Suzuki reaction catalysed by palladium supported on magnetic nanoparticles modified with carbon quantum dots generated from glycerol and urea. Appl Organomet Chem 32:1–14. https://doi.org/10.1002/aoc.3984
Haji S, Benstaali B, Al-Bastaki N (2011) Degradation of methyl orange by UV/H2O2 advanced oxidation process. Chem Eng J 168:134–139. https://doi.org/10.1016/j.cej.2010.12.050
He D, Wu X, Chen Y et al (2018) In-situ growth of lepidocrocite on Bi2O3 rod: a perfect cycle coupling photocatalysis and heterogeneous fenton-like process by potential-level matching with advanced oxidation. Chemosphere 210:334–340. https://doi.org/10.1016/j.chemosphere.2018.06.142
Hou L, Zhang Q, Jérôme F et al (2014) Shape-controlled nanostructured magnetite-type materials as highly efficient Fenton catalysts. Appl Catal B Environ 144:739–749. https://doi.org/10.1016/j.apcatb.2013.07.072
Hou X, Huang X, Jia F et al (2017) Hydroxylamine promoted goethite surface Fenton degradation of organic pollutants. Environ Sci Technol 51:5118–5126. https://doi.org/10.1021/acs.est.6b05906
Hu C, Li M, Qiu J, Sun Y (2019) Design and fabrication of carbon dots for energy conversion and storage. Chem Soc Rev 48:2315–2337. https://doi.org/10.1039/c8cs00750k
Hu S, Tian R, Dong Y et al (2013) Modulation and effects of surface groups on photoluminescence and photocatalytic activity of carbon dots. Nanoscale 5:11665–11671. https://doi.org/10.1039/c3nr03893a
Huang X, Hou X, Jia F et al (2017) Ascorbate-promoted surface iron cycle for efficient heterogeneous Fenton alachlor degradation with hematite nanocrystals. ACS Appl Mater Interfaces 9:8751–8758. https://doi.org/10.1021/acsami.6b16600
Jovanović SP, Syrgiannis Z, Budimir MD et al (2020) Graphene quantum dots as singlet oxygen producer or radical quencher - the matter of functionalization with urea/thiourea. Mater Sci Eng C 109:110539. https://doi.org/10.1016/j.msec.2019.110539
Khanna L, Gupta G, Tripathi SK (2019) Effect of size and silica coating on structural, magnetic as well as cytotoxicity properties of copper ferrite nanoparticles. Mater Sci Eng C 97:552–566. https://doi.org/10.1016/j.msec.2018.12.051
Kumar S, Ojha AK, Bhorolua D et al (2019) Facile synthesis of CuO nanowires and Cu 2 O nanospheres grown on rGO surface and exploiting its photocatalytic, antibacterial and supercapacitive properties. Phys B Condens Matter 558:74–81. https://doi.org/10.1016/j.physb.2019.01.040
Lenders JJM, Mirabello G, Sommerdijk NAJM (2016) Bioinspired magnetite synthesis via solid precursor phases. Chem Sci 7:5624–5634. https://doi.org/10.1039/c6sc00523c
Litter MI (2005) Introduction to photochemical advanced oxidation processes for water treatment. Environmental Photochemistry Part II. Springer-Verlag, Berlin/Heidelberg, pp 325–366
Liu ML, Bin CB, Li CM, Huang CZ (2019) Carbon dots: Synthesis, formation mechanism, fluorescence origin and sensing applications. Green Chem 21:449–471. https://doi.org/10.1039/c8gc02736f
Lorenzo D, Dominguez CM, Romero A, Santos A (2019) Wet peroxide oxidation of chlorobenzenes catalyzed by goethite and promoted by hydroxylamine. Catalysts 9https://doi.org/10.3390/catal9060553
Lu W, Gong X, Nan M et al (2015) Comparative study for N and S doped carbon dots: Synthesis, characterization and applications for Fe3+ probe and cellular imaging. Anal Chim Acta 898:116–127. https://doi.org/10.1016/j.aca.2015.09.050
Magnacca G, Allera A, Montoneri E et al (2014) Novel magnetite nanoparticles coated with waste sourced bio- based substances as sustainable and renewable adsorbing materials. ACS Sustain Chem Eng 2:1518–1524. https://doi.org/10.1021/sc500213j
Martínez-Pachón D, Espinosa-Barrera P, Rincón-Ortíz J, Moncayo-Lasso A (2019) Advanced oxidation of antihypertensives losartan and valsartan by photo-electro-Fenton at near-neutral pH using natural organic acids and a dimensional stable anode-gas diffusion electrode (DSA-GDE) system under light emission diode (LED) lighting. Environ Sci Pollut Res 26:4426–4437. https://doi.org/10.1007/s11356-018-2645-3
Mercado DF, Bracco LLB, Arques A et al (2018a) Reaction kinetics and mechanisms of organosilicon fungicide flusilazole with sulfate and hydroxyl radicals. Chemosphere 190:327–336. https://doi.org/10.1016/j.chemosphere.2017.09.134
Mercado DF, Caregnato P, Villata LS, Gonzalez MC (2018b) Ilex paraguariensis extract-coated magnetite nanoparticles: a sustainable nano-adsorbent and antioxidant. J Inorg Organomet Polym Mater 28:519–527. https://doi.org/10.1007/s10904-017-0757-8
Mercado DF, Cipollone M, González MC, Sánchez FH (2018c) Yerba mate applications: magnetic response of powders and colloids of iron oxide nanoparticles coated with Ilex paraguariensis derivatives. J Magn Magn Mater 462:13–21. https://doi.org/10.1016/j.jmmm.2018.04.048
Mercado DF, Magnacca G, Malandrino M et al (2014) Paramagnetic iron-doped hydroxyapatite nanoparticles with improved metal sorption properties. A bioorganic substrates- mediated synthesis. ACS Appl Mater Interfaces 6:3937–3946. https://doi.org/10.1021/am405217j
Mercado DF, Weiss RG (2018) Polydimethylsiloxane as a matrix for the stabilization and immobilization of zero-valent iron nanoparticles. Applications to dehalogenation of environmentally deleterious molecules. J Braz Chem Soc 29:1427–1439. https://doi.org/10.21577/0103-5053.20180006
Molaei MJ (2020) The optical properties and solar energy conversion applications of carbon quantum dots: a review. Sol Energy 196:549–566. https://doi.org/10.1016/j.solener.2019.12.036
Mondal P, Anweshan A, Purkait MK (2020) Green synthesis and environmental application of iron-based nanomaterials and nanocomposite: a review. Chemosphere 259:127509. https://doi.org/10.1016/j.chemosphere.2020.127509
Monje DS, Chacon KM, Galindo IC et al (2021) Carbon dots from agroindustrial residues: a critical comparison of the effect of physicochemical properties on their performance as photocatalyst and emulsion stabilizer. Mater Today Chem 20:100445. https://doi.org/10.1016/j.mtchem.2021.100445
Monje DS, Ruiz OS, Valencia GC, Mercado DF (2022) Iron oxide nanoparticles embedded in organic microparticles from Yerba Mate useful for remediation of textile wastewater through a photo - Fenton treatment : Ilex paraguariensis as a platform of environmental interest – Part 1. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-022-19744-4
Munoz M, de Pedro ZM, Casas JA, Rodriguez JJ (2015) Preparation of magnetite-based catalysts and their application in heterogeneous Fenton oxidation - a review. Appl Catal B Environ 176–177:249–265
Muthukumar H, Manickam M (2015) Amaranthus spinosus leaf extract mediated FeO nanoparticles : physicochemical traits, photocatalytic and antioxidant activity. ACS Sustain Chem Eng 3:3149–3156. https://doi.org/10.1021/acssuschemeng.5b00722
Nalbandian L, Patrikiadou E, Zaspalis V et al (2015) Magnetic nanoparticles in medical diagnostic applications: synthesis, characterization and proteins conjugation. Curr Nanosci 12:455–468. https://doi.org/10.2174/1573413712666151210230002
Neyens E, Baeyens J (2003) A review of classic Fenton’s peroxidation as an advanced oxidation technique. J Hazard Mater 98:33–50
Önal ES, Yatkin T, Ergüt M, Özer A (2017) Green synthesis of iron nanoparticles by aqueous extract of Eriobotrya japonica leaves as a heterogenous Fenton-like catalyst: degradation of Basic Red 46. Int J Chem Eng Appl 8:328–333. https://doi.org/10.18178/ijcea.2017.8.5.678
Ozbey Unal B, Bilici Z, Ugur N et al (2019) Adsorption and Fenton oxidation of azo dyes by magnetite nanoparticles deposited on a glass substrate. J Water Process Eng 32:100897. https://doi.org/10.1016/j.jwpe.2019.100897
Qin M, Lu B, Feng S et al (2019) Role of exposed facets and surface OH groups in the Fenton-like reactivity of lepidocrocite catalyst. Chemosphere 230:286–293. https://doi.org/10.1016/j.chemosphere.2019.05.071
Rahimi S, Moattari RMM, Rajabi L et al (2015) Iron oxide/hydroxide (α, γ-FeOOH) nanoparticles as high potential adsorbents for lead removal from polluted aquatic media. J Ind Eng Chem 23:33–43. https://doi.org/10.1016/j.jiec.2014.07.039
Sachdev A, Matai I, Gopinath P (2014) Implications of surface passivation on physicochemical and bioimaging properties of carbon dots. RSC Adv 4:20915–20921. https://doi.org/10.1039/c4ra02017k
Salgado P, Márquez K, Rubilar O et al (2019) The effect of phenolic compounds on the green synthesis of iron nanoparticles (Fe x O y -NPs) with photocatalytic activity. Appl Nanosci 9:371–385. https://doi.org/10.1007/s13204-018-0931-5
Sánchez Pérez JA, Arzate S, Soriano-Molina P et al (2020) Neutral or acidic pH for the removal of contaminants of emerging concern in wastewater by solar photo-Fenton? A techno-economic assessment of continuous raceway pond reactors. Sci Total Environ 736:139681. https://doi.org/10.1016/j.scitotenv.2020.139681
Şenel B, Demir N, Büyükköroğlu G, Yıldız M (2019) Graphene quantum dots: Synthesis, characterization, cell viability, genotoxicity for biomedical applications. Saudi Pharm J 27:846–858. https://doi.org/10.1016/j.jsps.2019.05.006
Shebanova ON, Lazor P (2003) Raman spectroscopic study of magnetite (FeFe2O4): a new assignment for the vibrational spectrum. J Solid State Chem 174:424–430. https://doi.org/10.1016/S0022-4596(03)00294-9
Sun D, Ban R, Zhang PH et al (2013) Hair fiber as a precursor for synthesizing of sulfur- and nitrogen-co-doped carbon dots with tunable luminescence properties. Carbon N Y 64:424–434. https://doi.org/10.1016/j.carbon.2013.07.095
Sun H, Xie G, He D, Zhang L (2020) Ascorbic acid promoted magnetite Fenton degradation of alachlor: mechanistic insights and kinetic modeling. Appl Catal B Environ 267:118383. https://doi.org/10.1016/j.apcatb.2019.118383
Tabbì G, Giuffrida A, Bonomo RP (2013) Determination of formal redox potentials in aqueous solution of copper(II) complexes with ligands having nitrogen and oxygen donor atoms and comparison with their EPR and UV-Vis spectral features. J Inorg Biochem 128:137–145. https://doi.org/10.1016/j.jinorgbio.2013.07.035
Tadesse A, Ramadevi D, Hagos M et al (2018) Synthesis of nitrogen doped carbon quantum dots/magnetite nanocomposites for efficient removal of methyl blue dye pollutant from contaminated water. RSC Adv 8:8528–8536
Vasimalai N, Vilas-Boas V, Gallo J et al (2018) Green synthesis of fluorescent carbon dots from spices for in vitro imaging and tumour cell growth inhibition. Beilstein J Nanotechnol 9:530–544. https://doi.org/10.3762/bjnano.9.51
Villegas-Guzman P, Giannakis S, Torres-Palma RA, Pulgarin C (2017) Remarkable enhancement of bacterial inactivation in wastewater through promotion of solar photo-Fenton at near-neutral pH by natural organic acids. Appl Catal B Environ 205:219–227. https://doi.org/10.1016/j.apcatb.2016.12.021
Wang T, Zhai Y, Zhu Y et al (2018) A review of the hydrothermal carbonization of biomass waste for hydrochar formation: process conditions, fundamentals, and physicochemical properties. Renew Sustain Energy Rev 90:223–247. https://doi.org/10.1016/j.rser.2018.03.071
Wang Y, Gao Y, Chen L, Zhang H (2015) Goethite as an efficient heterogeneous Fenton catalyst for the degradation of methyl orange. Catal Today 252:107–112. https://doi.org/10.1016/j.cattod.2015.01.012
Wu J, Wang P, Wang F, Fang Y (2018) Investigation of the microstructures of graphene quantum dots (GQDs) by surface-enhanced Raman spectroscopy. Nanomaterials 8:864. https://doi.org/10.3390/nano8100864
Yeh CKJ, Hsu CY, Chiu CH, Huang KL (2008) Reaction efficiencies and rate constants for the goethite-catalyzed Fenton-like reaction of NAPL-form aromatic hydrocarbons and chloroethylenes. J Hazard Mater 151:562–569. https://doi.org/10.1016/j.jhazmat.2007.06.014
Zang H, Miao C, Shang J et al (2018) Structural effects on the catalytic activity of carbon-supported magnetite nanocomposites in heterogeneous Fenton-like reactions. RSC Adv 8:16193–16201
Zhou Y, Meng J, Zhang M, et al (2019a) Which type of pollutants need to be controlled with priority in wastewater treatment plants: traditional or emerging pollutants? Environ Int 131. https://doi.org/10.1016/j.envint.2019.104982
Zhou Y, Zahran EM, Quiroga BA et al (2019b) Size-dependent photocatalytic activity of carbon dots with surface-state determined photoluminescence. Appl Catal B Environ 248:157–166. https://doi.org/10.1016/j.apcatb.2019.02.019
Zhu B, Xia P, Ho W, Yu J (2015) Isoelectric point and adsorption activity of porous g-C 3 N 4. Appl Surf Sci 344:188–195. https://doi.org/10.1016/j.apsusc.2015.03.086
Funding
The authors thank the Gobernación de Antioquia, SAPIENCIA, EIA, Universidad de Antioquia (acta No. 2020–38851), for their support, and I.U. COLMAYOR and the Universidad Nacional de Colombia-Sede Medellín, for their financial support (HERMES 50042). Moreover, DSM is supported by the MinCiencias and the Universidad Nacional de Colombia through a Joven Investigador scholarship HERMES 43136.
Author information
Authors and Affiliations
Contributions
Dany S. Monje: investigation, methodology, writing—original draft, writing—review and editing. D. Fabio Mercado: term, conceptualization, methodology, investigation, supervision, writing—review and editing. Gustavo A. Peñuela Mesa: methodology, supervision and editing. Gloria Cristina Valencia: methodology, investigation, supervision.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Angeles Blanco
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Monje, D.S., Mercado, D.F., Mesa, G.A.P. et al. Carbon dots decorated magnetite nanocomposite obtained using yerba mate useful for remediation of textile wastewater through a photo-Fenton treatment: Ilex paraguariensis as a platform of environmental interest—part 2. Environ Sci Pollut Res 30, 3070–3087 (2023). https://doi.org/10.1007/s11356-022-22405-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-22405-1