Abstract
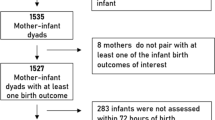
Gestational arsenic (As) exposure is associated with intrauterine growth restriction (IUGR). This study explored the association among gestational As exposure, IUGR, and reduction of folate content in maternal and umbilical plasma from 530 mother-and-singleton-offspring pairs. Birth weight (BW) was negatively correlated with As in maternal plasma (r=−0.194, P<0.001) and umbilical plasma (r=−0.235, P<0.001). By contrast, a positive correlation was found between BW and maternal folate content (r=0.198, P<0.001). The subjects were divided into As-L and As-H groups. The influence of As-H on small for gestational age (SGA) infants, a marker of IUGR, was evaluated by multivariate logistic regression that excludes interferences of gestational age, infant sex, and other confounding factors. Mothers with As-H had an elevated risk of SGA infants (adjusted OR, 2.370; P<0.05). Interestingly, maternal folate content was lower in subjects with As-H than those with As-L (22.4±10.7 vs 11.2±6.7 nmol/L, P<0.001). Linear correlation models show that As level was negatively correlated with folate content in maternal plasma (r=−0.615, P<0.001) and umbilical plasma (r=−0.209, P<0.001). Moreover, maternal folate reduction has an obvious mediating effect between increased As and decreased BW (β=−0.078, P<0.05). Our results indicate that folate reduction may be a mediator between gestational As exposure and IUGR.
Graphical abstract





Similar content being viewed by others
Data availability
Relevant data and materials are in the manuscripts and supplementary materials.
References
Abuawad A, Bozack AK, Saxena R, Gamble MV (2021) Nutrition, one-carbon metabolism and arsenic methylation. Toxicology 457:152803. https://doi.org/10.1016/j.tox.2021.152803
Ahmed OG, Shehata GA, Ali RM, Makboul R, Abd Allah ESH, Abd El-Rady NM (2021) Folic acid ameliorates neonatal isolation-induced autistic like behaviors in rats: epigenetic modifications of BDNF and GFAP promotors. Appl Physiol Nutr Metab 46(8):964–975. https://doi.org/10.1139/apnm-2020-0923
Baron RM, Kenny DA (1986) The moderator-mediator variable distinction in social psychological research: conceptual, strategic, and statistical considerations. J Pers Soc Psychol 51(6):1173–1182. https://doi.org/10.1037//0022-3514.51.6.1173
Blencowe H, Cousens S, Modell B, Lawn J (2010) Folic acid to reduce neonatal mortality from neural tube disorders. Int J Epidemiol 39:i110–i121. https://doi.org/10.1093/ije/dyq028
Bozack AK, Saxena R, Gamble MV (2018) Nutritional influences on one-carbon metabolism: effects on arsenic methylation and toxicity. Annu Rev Nutr 38:401–429. https://doi.org/10.1146/annurev-nutr-082117-051757
Bulloch RE, Wall CR, Thompson JMD, Taylor RS, Poston L, Roberts CT, Dekker GA, Kenny LC, Simpson NAB, Myers JE, McCowan LME, SCOPE Consortium (2020) Folic acid supplementation is associated with size at birth in the screening for pregnancy endpoints (SCOPE) international prospective cohort study. Early Hum Dev 147:105058. https://doi.org/10.1016/j.earlhumdev.2020.105058
Concha G, Vogler G, Lezcano D, Nermell B, Vahter M (1998) Exposure to inorganic arsenic metabolites during early human development. Toxicol Sci 44(2):185–190. https://doi.org/10.1006/toxs.1998.2486
Gu JF, Zhou H, Tang HL, Yang WT, Zeng M, Liu ZM, Peng PQ, Liao BH (2019) Cadmium and arsenic accumulation during the rice growth period under in situ remediation. Ecotoxicol Environ Saf 171:451–459. https://doi.org/10.1016/j.ecoenv.2019.01.003
Guo H, Mao B, Wang M, Liu Q, Yang L, Xie Y, Wang Y, He X, Cui H, Lin X, Lv L, Zhou M, Xu X, Qiu J, Zhang Y (2020) Folic acid supplementation, dietary folate intake and risk of small for gestational age in China. Public Health Nutr 23(11):1965–1973. https://doi.org/10.1017/S1368980019003331
Han Y, Liang C, Manthari RK, Yu Y, Gao Y, Liu Y, Jiang S, Tikka C, Wang J, Zhang J (2020) Arsenic influences spermatogenesis by disorganizing the elongation of spermatids in adult male mice. Chemosphere 238:124650. https://doi.org/10.1016/j.chemosphere.2019.124650
Hill DS, Wlodarczyk BJ, Finnell RH (2008) Reproductive consequences of oral arsenate exposure during pregnancy in a mouse model. Birth Defects Res B Dev Reprod Toxicol 83(1):40–47. https://doi.org/10.1002/bdrb.20142
Hill DS, Wlodarczyk BJ, Mitchell LE, Finnell RH (2009) Arsenate-induced maternal glucose intolerance and neural tube defects in a mouse model. Toxicol Appl Pharmacol 239(1):29–36. https://doi.org/10.1016/j.taap.2009.05.009
Hirano S (2020) Biotransformation of arsenic and toxicological implication of arsenic metabolites. Arch Toxicol 94(8):2587–2601. https://doi.org/10.1007/s00204-020-02772-9
Jin L, Zhang L, Li Z, Liu JM, Ye R, Ren A (2013) Placental concentrations of mercury, lead, cadmium, and arsenic and the risk of neural tube defects in a Chinese population. Reprod Toxicol 35:25–31. https://doi.org/10.1016/j.reprotox.2012.10.015
Li N, Li Z, Ye R, Liu J, Ren A (2017) Impact of periconceptional folic acid supplementation on low birth weight and small-for-gestational-age infants in china: a large prospective cohort study. J Pediatr 187:105–110. https://doi.org/10.1016/j.jpeds.2017.04.060
Li S, Liu D, Zhang R, Lei F, Liu X, Cheng Y, Li C, Xiao M, Guo L, Li M, Zhang B, Zhu Z, Shi G, Liu Y, Dang S, Yan H (2019) The association of maternal dietary folate intake and folic acid supplementation with small-for-gestational-age births: a cross-sectional study in Northwest China. Br J Nutr 122(4):459–467. https://doi.org/10.1017/S0007114519001272
Li Y, Ji L, Mi W, Xie S, Bi Y (2021) Health risks from groundwater arsenic on residents in northern China coal-rich region. Sci Total Environ 773:145003. https://doi.org/10.1016/j.scitotenv.2021.145003
Liu H, Lu S, Zhang B, Xia W, Liu W, Peng Y, Zhang H, Wu K, Xu S, Li Y (2018) Maternal arsenic exposure and birth outcomes: a birth cohort study in Wuhan, China. Environ Pollut 236:817–823. https://doi.org/10.1016/j.envpol.2018.02.012
Mullin AM, Amarasiriwardena C, Cantoral-Preciado A, Claus Henn B, Leon Hsu HH, Sanders AP, Svensson K, Tamayo-Ortiz M, Téllez-Rojo MM, Wright RO, Burris HH (2019) Maternal blood arsenic levels and associations with birth weight-for-gestational age. Environ Res 177:108603. https://doi.org/10.1016/j.envres.2019.108603
Richmond RC, Sharp GC, Herbert G, Atkinson C, Taylor C, Bhattacharya S, Campbell D, Hall M, Kazmi N, Gaunt T, McArdle W, Ring S, Davey Smith G, Ness A, Relton CL (2018) The long-term impact of folic acid in pregnancy on offspring DNA methylation: follow-up of the Aberdeen Folic Acid Supplementation Trial (AFAST). Int J Epidemiol 47(3):928–937. https://doi.org/10.1093/ije/dyy032
Roche ML, Samson KLI, Green TJ, Karakochuk CD, Martinez H (2021) Perspective: Weekly Iron and Folic Acid Supplementation (WIFAS): a critical review and rationale for inclusion in the essential medicines list to accelerate anemia and neural tube defects reduction. Adv Nutr 12(2):334–342. https://doi.org/10.1093/advances/nmaa169
Samal AC, Bhattacharya P, Biswas P, Maity JP, Bundschuh J, Santra SC (2021) Variety-specific arsenic accumulation in 44 different rice cultivars and human health risks due to co-exposure of arsenic-contaminated rice and drinking water. J Hazard Mater 407:124804. https://doi.org/10.1016/j.jhazmat.2020.124804
Samson KLI, Loh SP, Lee SS, Sulistyoningrum DC, Khor GL, Shariff ZBM, Ismai IZ, Yelland LN, Leemaqz S, Makrides M, Hutcheon JA, Roche ML, Karakochuk CD, Green TJ (2020) Weekly iron-folic acid supplements containing 2.8 mg folic acid are associated with a lower risk of neural tube defects than the current practice of 0.4 mg: a randomised controlled trial in Malaysia. BMJ Glob. Health 5(12):e003897. https://doi.org/10.1136/bmjgh-2020-003897
Simanjuntak Y, Ko HY, Lee YL, Yu GY, Lin YL (2020) Preventive effects of folic acid on Zika virus-associated poor pregnancy outcomes in immunocompromised mice. PLoS Pathog 16(5):e1008521. https://doi.org/10.1371/journal.ppat.1008521
Stýblo M, Venkatratnam A, Fry RC, Thomas DJ (2021) Origins, fate, and actions of methylated trivalent metabolites of inorganic arsenic: progress and prospects. Arch Toxicol 95(5):1547–1572. https://doi.org/10.1007/s00204-021-03028-w
Timmermans S, Jaddoe VW, Hofman A, Steegers-Theunissen RP, Steegers EA (2009) Periconception folic acid supplementation, fetal growth and the risks of low birth weight and preterm birth: the Generation R Study. Br J Nutr 102(5):777–785. https://doi.org/10.1017/S0007114509288994
Van Eijsden M, Smits LJ, van der Wal MF, Bonsel GJ (2008) Association between short interpregnancy intervals and term birth weight: the role of folate depletion. Am J Clin Nutr 88(1):147–153. https://doi.org/10.1093/ajcn/88.1.147
VanderWeele TJ (2016) Mediation Analysis: A Practitioner's Guide. Annu Rev Public Health 37:17–32. https://doi.org/10.1146/annurev-publhealth-032315-021402
Wang B, Li H, Li Z, Jian L, Gao Y, Qu Y, Liu C, Xu C, Li Y, Diao Z, Lu W, Yu Y, Machaty Z, Luo H (2019) Maternal folic acid supplementation modulates the growth performance, muscle development and immunity of Hu sheep offspring of different litter size. J Nutr Biochem 70:194–201. https://doi.org/10.1016/j.jnutbio.2019.05.011
Wang H, Li J, Zhang X, Zhu P, Hao JH, Tao FB, Xu DX (2018) Maternal serum arsenic level during pregnancy is positively associated with adverse pregnant outcomes in a Chinese population. Toxicol Appl Pharmacol 356:114–119. https://doi.org/10.1016/j.taap.2018.07.030
Wang X, Zhang J, Xu W, Huang Q, Liu L, Tian M, Xia Y, Zhang W, Shen H (2016) Low-level environmental arsenic exposure correlates with unexplained male infertility risk. Sci Total Environ 571:307–313. https://doi.org/10.1016/j.scitotenv.2016.07.169
Xu W, Bao H, Liu F, Liu L, Zhu YG, She J, Dong S, Cai M, Li L, Li C, Shen H (2012) Environmental exposure to arsenic may reduce human semen quality: associations derived from a Chinese cross-sectional study. Environ Health 11:46. https://doi.org/10.1186/1476-069X-11-46
Zhang B, Shang S, Li S, Mi B, Li M, Shi G, Ma M, Wang Q, Yan H, Dang S (2020) Maternal folic acid supplementation and more prominent birth weight gain in twin birth compared with singleton birth: a cross-sectional study in northwest China. Public Health Nutr 23(16):2973–2982. https://doi.org/10.1017/S1368980019004580
Zhang SH, Guo AJ, Wei N, Zhang R, Niu YJ (2021a) Associations of urinary dichloroacetic acid and trichloroacetic acid exposure with platelet indices: exploring the mediating role of blood pressure in the general population. J Hazard Mater 402:123452. https://doi.org/10.1016/j.jhazmat.2020.123452
Zhang GB, Wang H, Hu J, Guo MY, Wang Y, Zhou Y, Yu Z, Fu L, Chen YH, Xu DX (2016) Cadmium-induced neural tube defects and fetal growth restriction: association with disturbance of placental folate transport. Toxicol Appl Pharmacol 306:79–85. https://doi.org/10.1016/j.taap.2016.07.007
Zhang X, Liu J, Jin Y, Yang S, Song Z, Jin L, Wang L, Ren A (2019) Folate of pregnant women after a nationwide folic acid supplementation in China. Matern Child Nutr 15(4):e12828. https://doi.org/10.1111/mcn.12828
Zhang XY, Wang B, Xu S, Wang J, Gao L, Song YP, Lv JW, Xu FX, Li J, Chen J, Cui AQ, Zhang C, Wang H, Xu DX (2021b) Reactive oxygen species-evoked genotoxic stress mediates arsenic-induced suppression of male germ cell proliferation and decline in sperm quality. J Hazard Mater 406:124768. https://doi.org/10.1016/j.jhazmat.2020.124768
Zhao M, Chen YH, Dong XT, Zhou J, Chen X, Wang H, Wu SX, Xia MZ, Zhang C, Xu DX (2013) Folic acid protects against lipopolysaccharide-induced preterm delivery and intrauterine growth restriction through its anti-inflammatory effect in mice. PLoS One 8(12):e82713. https://doi.org/10.1371/journal.pone.0082713
Zheng N, Yu Y, Hou S, Xu S, Tang L, Ji X (2019) Contamination assessment and health risk of arsenic exposure to stairway dust in the zinc smelting district. Northeast China. Environ Geochem Health 41(2):729–736. https://doi.org/10.1007/s10653-018-0166-0
Acknowledgements
We are particularly thankful to all the participants in this study and to the medical staff responsible for collecting blood of recruited subjects at Wuxi Maternity and Child Health Care Hospital in China.
Funding
This work was supported by Key Project of the National Natural Science Foundation of China (81930093). The work was also supported by the major scientific research project of Wuxi Municipal Health Commission (Z201902).
Author information
Authors and Affiliations
Contributions
Fei-Xiang Xu and Xu Chen contributed equally to this work. The following are the details of the author’s work. Fei-Xiang Xu, methodology, formal analysis, and writing—original draft; Xu Chen, formal analysis; Heng Zhang, resources, supervision, and funding acquisition; Yi-Jun Fan, formal analysis; Ya-Ping Song, writing—original draft; Jin-wei Lv, methodology; Ya-Li Xie, formal analysis; Yichao Huang, methodology and formal analysis; Dao-Zhen Chen, formal analysis; Hua Wang, resources and formal analysis; De-Xiang Xu, formal analysis, conceptualization, and funding acquisition.
Corresponding author
Ethics declarations
Ethics approval
The Ethics Committee of Anhui Medical University in China approved this study (No. 20190295).
Consent to participate and consent for publication
All the participants in this study were recruited with available informed consent. And all listed authors have agreed to submit this manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 398kb)
Rights and permissions
About this article
Cite this article
Xu, FX., Chen, X., Zhang, H. et al. Association between gestational arsenic exposure and intrauterine growth restriction: the role of folate content. Environ Sci Pollut Res 29, 89652–89661 (2022). https://doi.org/10.1007/s11356-022-21961-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21961-w