Abstract
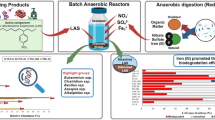
This work explores the degradation of xenobiotic compounds in aerobic and anaerobic batch reactors. Different inoculums were spiked with nine emerging contaminants at nominal concentrations ranging between 1 to 2 mg/L (ibuprofen, diclofenac, naproxen, acesulfame, sucralose, aspartame, cyclamate, linear alkylbenzene sulfonates, and secondary alkyl sulfonates). Ethanol was used as co-substrate in the anaerobic reactors. We found that the kinetic decay was faster in the aerobic reactors inoculated with a Spanish (Spn) inoculum compared to a Brazilian (Brz) inoculum, resulting in rection rates for LAS and SAS of 2.67 ± 3.6 h−1 and 5.09 ± 6 h−1 for the Brz reactors, and 1.3 ± 0.1 h−1 and 1.5 ± 0.2 h−1 for the Spn reactors, respectively. There was no evidence of LAS and SAS degradation under anaerobic conditions within 72 days; nonetheless, under aerobic conditions, these surfactants were removed by both the Brz and Spn inoculums (up to 86.2 ± 9.4% and 74.3 ± 0.7%, respectively) within 10 days. The artificial sweeteners were not removed under aerobic conditions, whereas we could observe a steady decrease in the anaerobic reactors containing the Spn inoculum. Ethanol aided in the degradation of surfactants in anaerobic environments. Proteiniphilum, Paraclostridium, Arcobacter, Proteiniclasticum, Acinetobacter, Roseomonas, Aquamicrobium, Moheibacter, Leucobacter, Synergistes, Cyanobacteria, Serratia, and Desulfobulbus were the main microorganisms identified in this study.



Similar content being viewed by others
Data availability
Molecular biology data from the next generation sequencing of this study are available on the website of the National Center for Biotechnology Information (https://www.ncbi.nlm.nih.gov/) publicly deposited under accession numbers: PRJNA595959, SRP238057, SRX7405061, SRX7405059, SRX7405058, SRX7405057, SRX7405056, SRX7405055, and SRX7405054. For other physical–chemical and chromatographic data, this question does not apply.
References
Abboud MM, Khleifat KM, Batarseh M et al (2007) Different optimization conditions required for enhancing the biodegradation of linear alkylbenzosulfonate and sodium dodecyl sulfate surfactants by novel consortium of Acinetobacter calcoaceticus and Pantoea agglomerans. Enzyme Microb Technol 41:432–439. https://doi.org/10.1016/j.enzmictec.2007.03.011
Alegbeleye OO, Opeolu BO, Jackson V (2017) Bioremediation of polycyclic aromatic hydrocarbon (PAH) compounds: (acenaphthene and fluorene) in water using indigenous bacterial species isolated from the Diep and Plankenburg rivers, Western Cape, South Africa. Brazilian J Microbiol 48:314–325. https://doi.org/10.1016/j.bjm.2016.07.027
Alygizakis NA, Urík J, Beretsou VG et al (2020) Evaluation of chemical and biological contaminants of emerging concern in treated wastewater intended for agricultural reuse. Environ Int 138:105597. https://doi.org/10.1016/j.envint.2020.105597
Alvarino T, Suárez S, Garrido M, Lema JM, Omil F (2016) A UASB reactor coupled to a hybrid aerobic MBR as innovative plant configuration to enhance the removal of organic micropollutants. Chemosphere 144:452–458. https://doi.org/10.1016/j.chemosphere.2015.09.016
Baena-Nogueras RM, González-Mazo E, Lara-Martín PA (2017) Degradation kinetics of pharmaceuticals and personal care products in surface waters: photolysis vs biodegradation. Sci Total Environ 590–591:643–654. https://doi.org/10.1016/j.scitotenv.2017.03.015
Baena-Nogueras RM, Rojas-Ojeda P, Sanz JL et al (2014) Reactivity and fate of secondary alkane sulfonates (SAS) in marine sediments. Environ Pollut 189:35–42. https://doi.org/10.1016/j.envpol.2014.02.019
Berna JL, Cassani G, Hager C-D et al (2007) Anaerobic Biodegradation of surfactants – scientific review. Tenside Surfactants Deterg 44:312–347. https://doi.org/10.3139/113.100351
Botheju D, Bakke R (2011) Oxygen effects in anaerobic digestion – A review. 1–19
Buerge IJ, Buser H-R, Kahle M et al (2009) Ubiquitous occurrence of the artificial sweetener acesulfame in the aquatic environment: an ideal chemical marker of domestic wastewater in groundwater. Environ Sci Technol 43:4381–4385. https://doi.org/10.1021/es900126x
Carballa M, Omil F, Ternes T, Lema JM (2007) Fate of pharmaceutical and personal care products (PPCPs) during anaerobic digestion of sewage sludge. Water Res 41:2139–2150. https://doi.org/10.1016/j.watres.2007.02.012
Carneiro RB, Gonzalez-Gil L, Londoño YA, Zaiat M, Carballa M, Lema JM (2020) Acidogenesis is a key step in the anaerobic biotransformation of organic micropollutants. J Hazard Mater 389:121888.https://doi.org/10.1016/j.jhazmat.2019.121888
Carosia MF, Okada DY, Sakamoto IK et al (2014) Microbial characterization and degradation of linear alkylbenzene sulfonate in an anaerobic reactor treating wastewater containing soap powder. Bioresour Technol 167:316–323. https://doi.org/10.1016/j.biortech.2014.06.002
Cole JR, Chai B, Farris RJ et al (2007) The ribosomal database project (RDP-II): introducing myRDP space and quality controlled public data. Nucleic Acids Res 35:D169–D172. https://doi.org/10.1093/nar/gkl889
Corada-Fernández C, González-Mazo E, Lara-Martín PA (2018) Evaluation of the anaerobic biodegradation of linear alkylbenzene sulfonates (LAS) using OECD 308 water/sediment systems. J Hazard Mater 360:24–31. https://doi.org/10.1016/j.jhazmat.2018.07.087
da Silva THG, Furtado RX de S, Zaiat M, Azevedo EB (2020) Tandem anaerobic-aerobic degradation of ranitidine, diclofenac, and simvastatin in domestic sewage. Sci Total Environ 721:137589. https://doi.org/10.1016/j.scitotenv.2020.137589
Delforno TP, Belgini DRB, Hidalgo KJ et al (2020) Anaerobic reactor applied to laundry wastewater treatment: unveiling the microbial community by gene and genome-centric approaches. Int Biodeterior Biodegradation 149:104916. https://doi.org/10.1016/j.ibiod.2020.104916
Delforno TP, Lacerda GV, Sierra-Garcia IN et al (2017) Metagenomic analysis of the microbiome in three different bioreactor configurations applied to commercial laundry wastewater treatment. Sci Total Environ 587–588:389–398. https://doi.org/10.1016/j.scitotenv.2017.02.170
Delforno TP, Macedo TZ, Midoux C et al (2019) Comparative metatranscriptomic analysis of anaerobic digesters treating anionic surfactant contaminated wastewater. Sci Total Environ 649:482–494. https://doi.org/10.1016/j.scitotenv.2018.08.328
Delforno TP, Okada DY, Polizel J et al (2012) Microbial characterization and removal of anionic surfactant in an expanded granular sludge bed reactor. Bioresour Technol 107:103–109. https://doi.org/10.1016/j.biortech.2011.12.042
Elias MT, Chandran J, Aravind UK, Aravindakumar CT (2019) Oxidative degradation of ranitidine by UV and ultrasound: identification of transformation products using LC-Q-ToF-MS. Environ Chem 16:41. https://doi.org/10.1071/EN18155
Field JA, Field TM, Poiger T et al (1995) Fate of secondary alkane sulfonate surfactants during municipal wastewater treatment. Water Res 29:1301–1307. https://doi.org/10.1016/0043-1354(94)00291-E
Floripes TC, Aquino SF, Quaresma AD, Afonso RJ, Chernicharo CA, Souza CL (2018) Ocorrência de fármacos e desreguladores endócrinos em esgoto bruto e tratado na cidade de Belo Horizonte (MG). Eng Sanit e Ambient 23:1199–1211. https://doi.org/10.1590/s1413-41522018177703
Gago-Ferrero P, Bletsou AA, Damalas DE et al (2020) Wide-scope target screening of >2000 emerging contaminants in wastewater samples with UPLC-Q-ToF-HRMS/MS and smart evaluation of its performance through the validation of 195 selected representative analytes. J Hazard Mater 387:121712. https://doi.org/10.1016/j.jhazmat.2019.121712
García-Morales JL, Nebot-Sanz E, Garcia LIR, Sales D (2001) Comparison between acidogenic and methanogenic inhibition caused by linear alkylbenzene-sulfonate (LAS). Chem Biochem Eng Q 15:13–19
Garcia MT, Campos E, Sánchez-Leal J, Ribosa I (2006) Effect of linear alkylbenzene sulphonates (LAS) on the anaerobic digestion of sewage sludge. Water Res 40:2958–2964. https://doi.org/10.1016/j.watres.2006.05.033
Gartiser S, Urich E, Alexy R, Kümmerer K (2007) Anaerobic inhibition and biodegradation of antibiotics in ISO test schemes. Chemosphere 66:1839–1848. https://doi.org/10.1016/j.chemosphere.2006.08.040
Ghattas A-K, Fischer F, Wick A, Ternes TA (2017) Anaerobic biodegradation of (emerging) organic contaminants in the aquatic environment. Water Res 116:268–295. https://doi.org/10.1016/j.watres.2017.02.001
Gibson J, Harwood CS (2002) Metabolic diversity in aromatic compound utilization by anaerobic microbes. Annu Rev Microbiol 56:345–369. https://doi.org/10.1146/annurev.micro.56.012302.160749
Granatto CF, Grosseli GM, Sakamoto IK et al (2021) Influence of metabolic cosubstrates on methanogenic potential and degradation of triclosan and propranolol in sanitary sewage. Environ Res 199:111220. https://doi.org/10.1016/j.envres.2021.111220
Granatto CF, Grosseli GM, Sakamoto IK et al (2020) Methanogenic potential of diclofenac and ibuprofen in sanitary sewage using metabolic cosubstrates. Sci Total Environ 742:140530. https://doi.org/10.1016/j.scitotenv.2020.140530
Häggblom MM, Bossert ID (2004) Dehalogenation - microbial processes and environmental applications. Kluwer Academic Publishers, Boston
Hijosa-Valsero M, Matamoros V, Martín-Villacorta J et al (2010) Assessment of full-scale natural systems for the removal of PPCPs from wastewater in small communities. Water Res 44:1429–1439. https://doi.org/10.1016/j.watres.2009.10.032
Ilyas S, Bhatti HN (2017) Microbial diversity as a tool for wastewater treatment. Advanced materials for wastewater treatment. John Wiley & Sons Inc, Hoboken, NJ, USA, pp 171–221
Ivanković T, Hrenović J (2010) Surfactants in the environment. Arh Hig Rada Toksikol 61:95–110. https://doi.org/10.2478/10004-1254-61-2010-1943
Jyothsna TS, Tushar L, Sasikala C, Ramana CV (2016) Paraclostridium benzoelyticum gen. nov., sp. nov., isolated from marine sediment and reclassification of Clostridium bifermentans as Paraclostridium bifermentans comb. nov. Proposal of a new genus Paeniclostridium gen. nov. to accommodate Clostridium sord. Int J Syst Evol Microbiol 66:1268–1274. https://doi.org/10.1099/ijsem.0.000874
Kahl S, Kleinsteuber S, Nivala J et al (2018) Emerging biodegradation of the previously persistent artificial sweetener acesulfame in biological wastewater Treatment. Environ Sci Technol 52:2717–2725. https://doi.org/10.1021/acs.est.7b05619
Keskes S, Hmaied F, Gannoun H, Bouallagui H, Godon JJ, Hamdi M (2012) Performance of a submerged membrane bioreactor for the aerobic treatment of abattoir wastewater. Bioresour Technol 103:28–34. https://doi.org/10.1016/j.biortech.2011.09.063
Khleifat KM (2006) Biodegradation of linear alkylbenzene sulfonate by a two-member facultative anaerobic bacterial consortium. Enzyme Microb Technol 39:1030–1035. https://doi.org/10.1016/j.enzmictec.2006.02.007
Ladino-Orjuela G, Gomes E, da Silva R, et al (2016) Metabolic Pathways for degradation of aromatic hydrocarbons by bacteria. pp 105–121
Langenhoff A, Inderfurth N, Veuskens T et al (2013) Microbial removal of the pharmaceutical compounds ibuprofen and diclofenac from wastewater. Biomed Res Int 2013:1–9. https://doi.org/10.1155/2013/325806
Lara-Martín PA, Gómez-Parra A, Köchling TK et al (2007) Anaerobic degradation of linear alkylbenzene sulfonates in coastal marine sediments. Environ Sci Technol 41:3573–9
Lara-Martín PA, Gómez-Parra A, Köchling T et al (2007b) Monitoring the primary biodegradation of linear alkylbenzene sulfonates and their coproducts in anoxic sediments using liquid chromatography−mass spectrometry. Environ Sci Technol 41:3580–3586. https://doi.org/10.1021/es062373r
Lara-Martín PA, Gómez-Parra A, Sanz JL, González-Mazo E (2010) Anaerobic degradation pathway of linear alkylbenzene sulfonates (LAS) in sulfate-reducing marine sediments. Environ Sci Technol 44:1670–1676. https://doi.org/10.1021/es9032887
Le-Minh N, Khan SJ, Drewes JE, Stuetz RM (2010) Fate of antibiotics during municipal water recycling treatment processes. Water Res 44:4295–4323. https://doi.org/10.1016/j.watres.2010.06.020
Li Z, Wang H, Tang Z et al (2008) Effects of pH value and substrate concentration on hydrogen production from the anaerobic fermentation of glucose. Int J Hydrogen Energy 33:7413–7418. https://doi.org/10.1016/j.ijhydene.2008.09.048
Macedo TZ, Delforno TP, Braga JK et al (2017) Robustness and microbial diversity of a fluidized bed reactor employed for the removal and degradation of an anionic surfactant from laundry wastewater. J Environ Eng 143:04017062. https://doi.org/10.1061/(ASCE)EE.1943-7870.0001240
Macedo TZ, Okada DY, Delforno TP et al (2015) The comparative advantages of ethanol and sucrose as co-substrates in the degradation of an anionic surfactant: microbial community selection. Bioprocess Biosyst Eng. https://doi.org/10.1007/s00449-015-1424-5
Mara D, Horan N (2003) Handbook of water and wastewater microbiology. Elsevier
Mazzoli R, Pessione E, Giuffrida MG et al (2007) Degradation of aromatic compounds by Acinetobacter radioresistens S13: growth characteristics on single substrates and mixtures. Arch Microbiol 188:55–68. https://doi.org/10.1007/s00203-007-0223-z
Mensah KA, Forster CF (2003) An examination of the effects of detergents on anaerobic digestion. Bioresour Technol 90:133–138. https://doi.org/10.1016/S0960-8524(03)00126-3
Merrettig-Bruns U, Jelen E (2009) Anaerobic biodegradation of detergent surfactants. Materials (basel) 2:181–206. https://doi.org/10.3390/ma2010181
Monteiro SC, Boxall ABA (2010) Occurrence and fate of human pharmaceuticals in the environment. pp 53–154
Motteran F, Braga JK, Silva EL, Varesche MBA (2016) Kinetics of methane production and biodegradation of linear alkylbenzene sulfonate from laundry wastewater. J Environ Sci Heal - Part A Toxic/Hazardous Subst Environ Eng 51 https://doi.org/10.1080/10934529.2016.1215197
Motteran F, Braga JK, Silva EL, Varesche MBA (2017) Influence of sucrose on the diversity of bacteria involved in nonionic surfactant degradation in fluidized bed reactor. Water Air Soil Pollut 228https://doi.org/10.1007/s11270-016-3193-4
Motteran F, Nadai BM, Braga JK et al (2018) Metabolic routes involved in the removal of linear alkylbenzene sulfonate (LAS) employing linear alcohol ethoxylated and ethanol as co-substrates in enlarged scale fluidized bed reactor. Sci Total Environ 640–641:1411–1423. https://doi.org/10.1016/j.scitotenv.2018.05.375
Nika MC, Ntaiou K, Elytis K, et al (2020) Wide-scope target analysis of emerging contaminants in landfill leachates and risk assessment using risk quotient methodology. J Hazard Mater 122493https://doi.org/10.1016/j.jhazmat.2020.122493
OECD (2006) Test No. 311: Anaerobic biodegradability of organic compounds in digested sludge: by measurement of gas production,. Paris, France
Okada DY, Delforno TP, Esteves AS et al (2012) Optimization of linear alkylbenzene sulfonate (LAS) degradation in UASB reactors by varying bioavailability of LAS, hydraulic retention time and specific organic load rate. Bioresour Technol 128C:125–133. https://doi.org/10.1016/j.biortech.2012.10.073
Okada DY, Delforno TP, Etchebehere C, Varesche MBA (2014) Evaluation of the microbial community of upflow anaerobic sludge blanket reactors used for the removal and degradation of linear alkylbenzene sulfonate by pyrosequencing. Int Biodeterior Biodegradation 96:63–70. https://doi.org/10.1016/j.ibiod.2014.09.017
Okada DY, Esteves AS, Delforno TP et al (2013) Influence of co-substrates in the anaerobic degradation of an anionic surfactant. Brazilian J Chem Eng 30:499–506. https://doi.org/10.1590/S0104-66322013000300008
Perales JA, Manzano MA, Garrido MC et al (2006) Biodegradation kinetics of linear alkylbenzene sulphonates in sea water. Biodegradation 18:63–70. https://doi.org/10.1007/s10532-005-9036-4
Peressutti SR, Olivera NL, Babay PA et al (2008) Degradation of linear alkylbenzene sulfonate by a bacterial consortium isolated from the aquatic environment of Argentina. J Appl Microbiol 105:476–484. https://doi.org/10.1111/j.1365-2672.2008.03771.x
Petrie B, Barden R, Kasprzyk-Hordern B (2015) A review on emerging contaminants in wastewaters and the environment: Current knowledge, understudied areas and recommendations for future monitoring. Water Res 72:3–27. https://doi.org/10.1016/j.watres.2014.08.05
Rodarte-Morales AI, Feijoo G, Moreira MT, Lema JM (2012) Biotransformation of three pharmaceutical active compounds by the fungus Phanerochaete chrysosporium in a fed batch stirred reactor under air and oxygen supply. Biodegradation 23:145–156. https://doi.org/10.1007/s10532-011-9494-9
Rostamizadeh M, Jalali H, Naeimzadeh F, Gharibian S (2019) Efficient Removal of Diclofenac from Pharmaceutical Wastewater Using Impregnated Zeolite Catalyst in Heterogeneous Fenton Process. Phys Chemestry Res 7:37–52. https://doi.org/10.22036/PCR.2018.144779.1524
Salgado R, Marques R, Noronha JP et al (2012) Assessing the removal of pharmaceuticals and personal care products in a full-scale activated sludge plant. Environ Sci Pollut Res 19:1818–1827. https://doi.org/10.1007/s11356-011-0693-z
Scott MJ, Jones MN (2000) The biodegradation of surfactants in the environment. Biochim Biophys Acta - Biomembr 1508:235–251. https://doi.org/10.1016/S0304-4157(00)00013-7
Silva LG, Silva DC, Oliveira LM et al (2017) Degradação dos homólogos do alquilbenzeno linear sulfonato em lodo anaeróbio disperso. Eng Sanit e Ambient 22:391–401. https://doi.org/10.1590/s1413-41522016153982
Sun W, Krumins V, Fennell DE et al (2015) Anaerobic degradation of aromatic compounds. Manual of Environmental Microbiology. ASM Press, Washington, DC, USA, p 5.1.3-1-5.1.3-14
Talib A, Randhir TO (2017) Managing emerging contaminants in watersheds: need for comprehensive, systems-based strategies. Sustain Water Qual Ecol 9–10:1–8. https://doi.org/10.1016/j.swaqe.2016.05.002
Thomaidis NS, Asimakopoulos AG, Blettsou AA (2012) Emerging contaminants: a tutorial mini-review. Glob NEST J 14:72–79
Tijani JO, Fatoba OO, Petrik LF (2013) A Review of pharmaceuticals and endocrine-disrupting compounds: sources, effects, removal, and detections. Water, Air, Soil Pollut 224:1770. https://doi.org/10.1007/s11270-013-1770-3
Torres CI, Ramakrishna S, Chiu C-A et al (2011) Fate of sucralose during wastewater treatment. Environ Eng Sci 28:325–331. https://doi.org/10.1089/ees.2010.0227
Tran NH, Gan J, Nguyen VT, Chen H, You L, Duarah A, Zhang L, Gin KY (2015) Sorption and biodegradation of artificial sweeteners in activated sludge processes. Bioresour Technol 197:329–338. https://doi.org/10.1016/j.biortech.2015.08.083
Vásquez-Piñeros MA, Martínez-Lavanchy PM, Jehmlich N et al (2018) Delftia sp. LCW, a strain isolated from a constructed wetland shows novel properties for dimethylphenol isomers degradation. BMC Microbiol 18:108. https://doi.org/10.1186/s12866-018-1255-z
Veras TB, Ribeiro L, de Paiva A, Duarte MMMB et al (2019) Analysis of the presence of anti-inflammatories drugs in surface water: a case study in Beberibe river - PE, Brazil. Chemosphere 222:961–969. https://doi.org/10.1016/j.chemosphere.2019.01.167
Verslyppe B, De Smet W, De Baets B et al (2014) StrainInfo introduces electronic passports for microorganisms. Syst Appl Microbiol 37:42–50. https://doi.org/10.1016/j.syapm.2013.11.002
Völker J, Vogt T, Castronovo S, Wick A, Ternes TA, Joss A, Oehlmann J, Wagner M (2017) Extended anaerobic conditions in the biological wastewater treatment: Higher reduction of toxicity compared to target organic micropollutants. Water Res 116:220–230. https://doi.org/10.1016/j.watres.2017.03.030
Wang S, Wang J (2018) Degradation of emerging contaminants by acclimated activated sludge. Environ Technol 39:1985–1993. https://doi.org/10.1080/09593330.2017.1345989
Ward HF, Stroo CH (2010) In situ remediation of chlorinated solvent plumes. Springer New York, New York, NY
Zhang Q, Tang L, Zhang J, Mao Z, Jiang L (2011) Optimization of thermal-dilute sulfuric acid pretreatment for enhancement of methane production from cassava residues. Bioresour Technol 102:3958–3965. https://doi.org/10.1016/j.biortech.2010.12.031
Zhao J, Chen X, Bao L, Bao Z, He Y, Zhang Y, Li J (2016) Correlation between microbial diversity and toxicity of sludge treating synthetic wastewater containing 4-chlorophenol in sequencing batch reactors. Chemosphere 153:138–145. https://doi.org/10.1016/j.chemosphere.2016.01.086
Zumstein E, Moletta R, Godon J-J (2000) Examination of two years of community dynamics in an anaerobic bioreactor using fluorescence polymerase chain reaction (PCR) single-strand conformation polymorphism analysis. Environ Microbiol 2:69–78. https://doi.org/10.1046/j.1462-2920.2000.00072.x
Acknowledgements
This research was funded by the Research Support Foundation of the State of São Paulo (FAPESP), process number 2016/06338-1, regarding the Research Internship Scholarship Program Abroad (BEPE) for financial support. The authors also thank the Laboratory of Biological Processes at the University of São Paulo (LPB/EESC/USP) and the professors and technicians of the Department of Physical Chemistry of the Faculty of Environmental and Marine Sciences of the University of Cadiz (UCA), who provided support and helped in the execution of this study (project RTI2018-096211-B-I00 funded by Spanish Ministry of Science, Innovation and Universities). The authors declare that they have no conflict of interest.
Funding
Fabricio Motteran reports financial support was provided by State of Sao Paulo Research Foundation process number 2016/06338–1, regarding the Research Internship Scholarship Program Abroad (BEPE) and Pablo A. Lara-Martin reports equipment, drugs, or supplies was provided by Spanish Ministry of Science, Innovation and Universities (project RTI2018-096211-B-I00).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by FM and PAL-M. The first draft of the manuscript was written by FM and revised by MBAV and PAL-M. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All authors consent to participate in this manuscript.
Consent for publication
All authors are in agreement with the submission and publication of the manuscript in the journal’s rules and that all responsible authorities for both institutions involved are in agreement with what was decided by the authors. All the authors declare that the manuscript was not previously submitted to Environmental Science and Pollution Research and confirm that this manuscript has not been published elsewhere and is not under consideration by another journal. All the authors have read and have abided by the statement of ethical standards and do not have conflict of interest for the manuscript submitted to Environmental Science and Pollution Research. All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Fabrico Motteran and Pablo A. Lara-Martin. The first draft of the manuscript was written by Fabricio Motteran and revised by Maria Bernadete A. Varesche and Pablo A. Lara-Martin. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Gerald Thouand
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Motteran, F., Varesche, M.B.A. & Lara-Martin, P.A. Assessment of the aerobic and anaerobic biodegradation of contaminants of emerging concern in sludge using batch reactors. Environ Sci Pollut Res 29, 84946–84961 (2022). https://doi.org/10.1007/s11356-022-21819-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-21819-1