Abstract
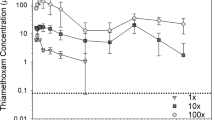
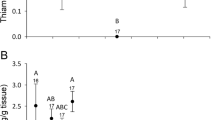
Agrochemicals including neonicotinoid insecticides and fungicides are frequently applied as seed treatments on corn, soybeans, and other common row crops. Crops grown from pesticide-treated seed are often directly planted in managed floodplain wetlands and used as a soil disturbance or food resource for wildlife. We quantified invertebrate communities within mid-latitude floodplain wetlands and assessed their response to use of pesticide-treated seeds within the floodplain. We collected and tested aqueous and sediment samples for pesticides in addition to sampling aquatic invertebrates from 22 paired wetlands. Samples were collected twice in 2016 (spring [pre-water level drawdown] and autumn [post-water level flood-up]) followed by a third sampling period (spring 2017). Meanwhile, during the summer of 2016, a portion of study wetlands were planted with either pesticide-treated or untreated corn seed. Neonicotinoid toxic equivalencies (NI-EQs) for sediment (X̅ = 0.58 μg/kg), water (X̅ = 0.02 μg/L), and sediment fungicide concentrations (X̅ = 0.10 μg/kg) were used to assess potential effects on wetland invertebrates. An overall decrease in aquatic insect richness and abundance was associated with greater NI-EQs in wetland water and sediments, as well as with sediment fungicide concentration. Post-treatment, treated wetlands displayed a decrease in insect taxa-richness and abundance before recovering by the spring of 2017. Information on timing and magnitude of aquatic insect declines will be useful when considering the use of seed treatments for wildlife management. More broadly, this study brings attention to how agriculture is used in wetland management and conservation planning.

Copyright © 2020 Esri and its licensors. All rights reserved





Similar content being viewed by others
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Agatz A, Ashauer R, Brown CD (2014) Imidacloprid perturbs feeding of Gammarus pulex at environmentally relevant concentrations. Environ Toxicol Chem 33:648–653
Alexander AC, Heard KS, Culp JM (2008) Emergent body size of mayfly survivors. Freshw Biol 53:171–180. https://doi.org/10.1111/j.1365-2427.2007.01880.x
Antweiler RC, Taylor HE, Taylor HE (2008) Evaluation of statistical treatments of left-censored environmental data using coincident uncensored data sets: II. Group Comparisons Environ Sci Technol 49:13439–13446. https://doi.org/10.1021/acs.est.5b02385
Atwood LW, Mortensen DA, Koide RT, Smith RG (2018) Evidence for multi-trophic effects of pesticide seed treatments on non-targeted soil fauna. Soil Biol Biochem 125:144–155. https://doi.org/10.1016/j.soilbio.2018.07.007
Barmentlo S, Schrama M, van Bodegom PM, de Snoo GR, Musters CJM, Vijver MG (2019) Neonicotinoids and fertilizers jointly structure naturally assembled freshwater macroinvertebrate communities. Sci Total Environ 691:1–29. https://doi.org/10.1016/j.scitotenv.2019.07.110
Bass C, Denholm I, Williamson MS, Nauen R (2015) The global status of insect resistance to neonicotinoid insecticides. Pestic Biochem Physiol 121:78–87
Beketov MA, Schäfer RB, Marwitz A, Paschke A, Liess M (2008) Long-term stream invertebrate community alterations induced by the insecticide thiacloprid: effect concentrations and recovery dynamics. Sci. Total Environ. 405:96–108. https://doi.org/10.1016/j.scitotenv.2008.07.001
Bendis RJ, Relyea RA (2016) Wetland defense: naturally occurring pesticide resistance in zooplankton populations protects the stability of aquatic communities. Oecologia. https://doi.org/10.1007/s00442-016-3574-9
Berheim EH, Jenks JA, Lundgren JG, Michel ES, Grove D, Jensen WF (2019) Effects of neonicotinoid insecticides on physiology and reproductive characteristics of captive female and fawn white-tailed deer. Sci Rep 9:1–10. https://doi.org/10.1038/s41598-019-40994-9
Bonmatin J-MM, Giorio C, Girolami V, Goulson D, Kreutzweiser DP, Krupke C, Liess M, Long E, Marzaro M, Mitchell EAD, Noome DA, Simon-Delso N, Tapparo A (2015) Environmental fate and exposure; neonicotinoids and fipronil. Environ Sci Pollut Res 22:35–67. https://doi.org/10.1007/s11356-014-3332-7
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Mächler M, Bolker BM (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://doi.org/10.3929/ETHZ-B-000240890
Cavallaro MC, Liber K, Headley JV, Peru KM, Morrissey CA (2017a) Community-level and phenological responses of emerging aquatic insects exposed to three neonicotinoid insecticides: an in situ wetland limnocorral approach. Environ Toxicol Chem. https://doi.org/10.1002/ieam.2007
Cavallaro MC, Main AR, Liber K, Phillips ID, Headley JV, Peru KM, Morrissey CA (2019) Neonicotinoids and other agricultural stressors collectively modify aquatic insect communities. Chemosphere 226:945–955. https://doi.org/10.1016/j.chemosphere.2019.03.176
Cavallaro MC, Morrissey CA, Headley JV, Peru KM, Liber K (2017b) Comparative chronic toxicity of imidacloprid, clothianidin, and thiamethoxam to Chironomus dilutus and estimation of toxic equivalency factors. Environ Toxicol Chem 36:372–382. https://doi.org/10.1002/etc.3536
Chagnon M, Kreutzweiser D, Mitchell E, a D., Morrissey, C. a., Noome, D. a., Van der Sluijs, J.P., (2014) Risks of large-scale use of systemic insecticides to ecosystem functioning and services. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-014-3277-x
Collier KJ, Probert PK, Jeffries M (2016) Conservation of aquatic invertebrates: concerns, challenges and conundrums. Aquat Conserv Mar Freshw Ecosyst 26:817–837. https://doi.org/10.1002/aqc.2710
Covich AP, Palmer MA, Crowl TA (1999) The role of benthic invertebrate species in freshwater ecosystems: zoobenthic species influence energy flows and nutrient cycling. Bioscience 49:119–127. https://doi.org/10.1525/bisi.1999.49.2.119
Davis CA, Smith LM (1998) Ecology and management of migrant shorebirds in the Playa Lakes Region of Texas. Wildlife Monogr. 140:3–45
Dawoud M, Bundschuh M, Goedkoop W, McKie BG (2017) Interactive effects of an insecticide and a fungicide on different organism groups and ecosystem functioning in a stream detrital food web. Aquat Toxicol 186:215–221. https://doi.org/10.1016/j.aquatox.2017.03.008
Douglas MR, Tooker JF (2015) Large-scale deployment of seed treatments has driven rapid increase in use of neonicotinoid insecticides and preemptive pest management in U.S. field crops. Environ Sci Technol 49:5088–5097. https://doi.org/10.1021/es506141g
Birch ANE, Begg GS, Squire GR (2011) How agro-ecological research helps to address food security issues under new IPM and pesticide reduction policies for global crop production systems. J. Exp. Bot. 62:3251–3261. https://doi.org/10.1093/jxb/err064
Englert D, Bundschuh M, Schulz R (2012) Thiacloprid affects trophic interaction between gammarids and mayflies. Environ Pollut 167:41–46. https://doi.org/10.1016/j.envpol.2012.03.024
EPA (1997) Azoxystrobin Pesticide Fact Sheet.
Epanchin PN, Knapp RA, Lawler SP, Ecology S, August N, Knapp A, Lawler P (2010) Ecological Society of America Nonnative trout impact an alpine-nesting bird by altering aquatic-insect subsidies. Ecology 91:2406–2415
Felsot AS, Ruppert JR (2002) Imidacloprid residues in Willapa Bay (Washington State) water and sediment following application for control of burrowing shrimp. J Agric Food Chem 50:4417–4423. https://doi.org/10.1021/jf011358x
Fowler DN, Webb EB, Vrtiska MP (2020) Condition bias of decoy-harvested light geese during the conservation order. J Wildl Manage 84:33–44. https://doi.org/10.1002/jwmg.21770
Fredrickson LH, Taylor TS (1982) Management of seasonally flooded impoundments for wildlife. Washington, D.C
Gleason R, Laubhan M, Tangen B, Kermes K (2008) Ecosystem services derived from wetland conservation practices in the United States Prairie Pothole Region with an emphasis on the U.S. Department of Agriculture Conservation Reserve and Wetlands Reserve Programs
Goulson D (2013) An overview of the environmental risks posed by neonicotinoid insecticides. J Appl Ecol 50:977–987. https://doi.org/10.1111/1365-2664.12111
Gray MJ, Hagy HM, Nyman JA, Stafford JD (2013) Chapter 4: management of wetlands for wildlife, wetland techniques: volume 3: applications and management. https://doi.org/10.1007/978-94-007-6907-6
Hallmann CA, Foppen RPB, Van Turnhout CAM, Kroon H. De, Jongejans E (2014) Declines in insectivorous birds are associated with high neonicotinoid concentrations. Nature 511:341–343. https://doi.org/10.1038/nature13531
Hayasaka D, Korenaga T, Suzuki K, Saito F, Sánchez-Bayo F, Goka K (2012) Cumulative ecological impacts of two successive annual treatments of imidacloprid and fipronil on aquatic communities of paddy mesocosms. Ecotoxicol Environ Saf 80:355–362. https://doi.org/10.1016/j.ecoenv.2012.04.004
Helsel D (2010) Much ado about next to nothing: incorporating nondetects in science. Ann Occup Hyg 54:257–262. https://doi.org/10.1093/annhyg/mep092
Hitaj C, Smith DJ, Code A, Wechsler S, Esker PD, Douglas MR (2020) Sowing uncertainty: what we do and don’t know about the planting of pesticide-treated seed. Bioscience 1–14. https://doi.org/10.1093/biosci/biaa019
Hladik ML, Corsi SR, Kolpin DW, Baldwin AK, Blackwell BR, Cavallin JE (2018) Year-round presence of neonicotinoid insecticides in tributaries to the Great Lakes, USA. Environ Pollut. https://doi.org/10.1016/j.envpol.2018.01.013
Hladik ML, Kolpin DW (2015) First national-scale occurrence of neonicotinoid insecticides in streams across the U.S.A. Environ Chem 13:12–20. https://doi.org/10.1071/EN15061
Hladik ML, Kolpin DW, Kuivila KM (2014) Widespread occurrence of neonicotinoid insecticides in streams in a high corn and soybean producing region. USA Environ Pollut 193:189–196. https://doi.org/10.1016/j.envpol.2014.06.033
Hobbs NT, Andrén H, Persson J, Aronsson M, Chapron G (2012) Native predators reduce harvest of reindeer by Sámi pastoralists. Ecol Appl 22:1640–1654. https://doi.org/10.1890/11-1309.1
Jeschke P, Nauen R (2008) Neonicotinoids-from zero to hero in insecticide chemistry. Pest Manag Sci 64:1084–1098. https://doi.org/10.1002/ps.1631
Jeschke P, Nauen R, Schindler M, Elbert A (2011) Overview of the status and global strategy for neonicotinoids. J Agric Food Chem 59:2897–2908. https://doi.org/10.1021/jf101303g
Kogan M (1998) Integrated pest management: historical perspectives and contemporary developments. Annu Rev Entomol 43:243–270https://doi.org/10.1146/annurev.ento.43.1.243
Kreutzweiser DP, Thompson DG, Scarr TA (2009) Imidacloprid in leaves from systemically treated trees inhibit litter breakdown by non-target invertebrates. Ecotoxicol Environ Saf 72:1053–1057
Kuechle KJ, Webb EB, Mengel D, Main AR (2019) Factors influencing neonicotinoid insecticide concentrations in floodplain wetland sediments across Missouri. Environ Sci Technol 53. https://doi.org/10.1021/acs.est.9b01799
Kunz JL, Ingersoll CG, Smalling KL, Elskus AA, Kuivila KM (2017) Chronic toxicity of azoxystrobin to freshwater amphipods, midges, cladocerans, and mussels in water-only exposures. Environ Toxicol Chem 36:2308–2315. https://doi.org/10.1002/etc.3764
Liu Y, Pan X, Li J (2015) A 1961–2010 record of fertilizer use, pesticide application and cereal yields: a review. Agron Sustain Dev 35:83–93. https://doi.org/10.1007/s13593-014-0259-9
Main AR, Headley JV, Peru KM, Michel NL, Cessna AJ, Morrissey C, a., (2014) Widespread use and frequent detection of neonicotinoid insecticides in wetlands of Canada’s prairie pothole region. PLoS ONE 9:1–12. https://doi.org/10.1371/journal.pone.0092821
Main AR, Webb EB, Goyne KW, Mengel D (2018) Neonicotinoid insecticides negatively affect performance measures of non-target terrestrial arthropods: a meta-analysis. Ecol Appl 28:1232–1244. https://doi.org/10.1002/eap.1723
Miles JC, Hua J, Sepulveda MS, Krupke CH, Hoverman JT (2017) Effects of clothianidin on aquatic communities: evaluating the impacts of lethal and sublethal exposure to neonicotinoids. PLoS ONE 1–24. https://doi.org/10.4231/R7RX992T
Mohr S, Berghahn R, Schmiediche R, Hübner V, Loth S, Feibicke M, Mailahn W, Wogram J (2012) Macroinvertebrate community response to repeated short-term pulses of the insecticide imidacloprid. Aquat Toxicol 110–111:25–36. https://doi.org/10.1016/j.aquatox.2011.11.016
Morrissey CA, Mineau P, Devries JH, Sanchez-Bayo F, Liess M, Cavallaro MC, Liber K (2015) Neonicotinoid contamination of global surface waters and associated risk to aquatic invertebrates: a review. Environ Int 74:291–303. https://doi.org/10.1016/j.envint.2014.10.024
Nakanishi K, Yokomizo H, Hayashi TI (2018) Were the sharp declines of dragonfly populations in the 1990s in Japan caused by fipronil and imidacloprid? An analysis of Hill’s causality for the case of Sympetrum frequens. Environ Sci Pollut Res 1–13. https://doi.org/10.1007/s11356-018-3440-x
Nakano S, Murakami M (2001) Reciprocal subsidies: dynamic interdependence between terrestrial and aquatic food webs. Proc Natl Acad Sci 98:166–170. https://doi.org/10.1073/pnas.98.1.166
Nauen R, Denholm I (2005) Resistance of insect pests to neonicotinoid insecticides: current status and future prospects. Arch Insect Biochem Physiol 58:200–215. https://doi.org/10.1002/arch.20043
Paetzold A, Schubert CJ, Tockner K (2005) Aquatic terrestrial linkages along a braided-river: riparian arthropods feeding on aquatic insects. Ecosystems 8:748–759. https://doi.org/10.1007/s10021-005-0004-y
Pestana JLT, Alexander AC, Culp JM, Baird DJ, Cessna AJ, Soares AMVM (2009) Structural and functional responses of benthic invertebrates to imidacloprid in outdoor stream mesocosms. Environ Pollut. 157:2328–2334. https://doi.org/10.1016/j.envpol.2009.03.027
Pisa LW, Amaral-Rogers V, Belzunces LP, Bonmatin JM, Downs CA, Goulson D, Kreutzweiser DP, Krupke C, Liess M, Mcfield M, Morrissey CA, Noome DA, Settele J, Simon-Delso N, Stark JD, Van der Sluijs JP, Van Dyck H, Wiemers M (2014) Effects of neonicotinoids and fipronil on non-target invertebrates. Environ Sci Pollut Res Int 22:68–102. https://doi.org/10.1007/s11356-014-3471-x
Raby M, Nowierski M, Perlov D, Zhao X, Hao C, Poirier DG, Sibley PK (2018) Acute toxicity of six neonicotinoid insecticides to freshwater invertebrates. Environ Toxicol Chem. https://doi.org/10.1002/etc.4088
Sánchez-Bayo F, Wyckhuys KAG (2019) Worldwide decline of the entomofauna: a review of its drivers. Biol Conserv 232:8–27. https://doi.org/10.1016/j.biocon.2019.01.020
Sappington K, Ruhman M, Housenger J (2017) Imidacloprid preliminary aquatic risk assessment for imidacloprid outline US Environmental Protection Agency
Schepker TJ, Lagrange T, Webb EB (2019) Are waterfowl food resources limited during spring migration? A bioenergetic assessment of playas in Nebraska’s Rainwater Basin. Wetlands 39:173–1884. https://doi.org/10.1007/s13157-018-1047-0
Schepker TJ, Webb EB, Tillitt D, Lagrange T (2020) Neonicotinoid insecticide concentrations in agricultural wetlands and associations with aquatic invertebrate communities. Agric Ecosyst Environ 287:106678. https://doi.org/10.1016/j.agee.2019.106678
Stamm MD, Heng-Moss TM, Baxendale FP, Siegfried BD, Gaussoin RE, Snow DD, Cassada DA (2012) Effect of distribution and concentration of topically applied neonicotinoid insecticides in buffalograss, Buchloe dactyloides, leaf tissues on the differential mortality of Blissus occiduus under field conditions. Pest Manag Sci 69:285–291. https://doi.org/10.1002/ps.3387
Stehle S, Schulz R (2015) Agricultural insecticides threaten surface waters at the global scale. Proc Natl Acad Sci 112:201500232. https://doi.org/10.1073/pnas.1500232112
Sur R, Stork A, Robin SUR, Stork A (2003) Uptake, translocation and metabolism of imidacloprid in plants. Bull Insectology 56:35–40
Syngenta Crop Protection, n.d. Cruiser 5FS Seed Treatment [WWW Document]. URL https://www.syngenta-us.com/current-label/cruiser_5fs
Taillebois E, Cartereau A, Jones AK, Thany SH (2018) Neonicotinoid insecticides mode of action on insect nicotinic acetylcholine receptors using binding studies. Pestic Biochem Physiol 0–1. https://doi.org/10.1016/j.pestbp.2018.04.007
Tapp JL, Webb EB (2015) Aquatic invertebrate food base for waterbirds at Wetland Reserve Program easements in the Lower Mississippi Alluvial Valley. Wetlands 35:183–192. https://doi.org/10.1007/s13157-014-0613-3
Tidwell PR, Webb EB, Vrtiska MP, Bishop AA (2013) Diets and food selection of female mallards and blue-winged teal during spring migration. J Fish Wildl Manag 4:63–74. https://doi.org/10.3996/072012-JFWM-062
Tilman D, Fargione J, Wolff B, D’Antoniio C, Dobson A, Howarth R, Schindler D, Schlesinger WH, Simberloff D, Swackhamer D (2001) Forecasting agriculturally driven global environmental change. Science (80-) 292:281–284. https://doi.org/10.1126/science.1057544
U.S. Environmental Protection Agency [WWW Document], 2019. . Aquat. Life Benchmarks Ecol. Risk Assessments Regist. Pestic.
Voshell JRJ (2002) A guide to common freshwater invertebrates of North America. The McDonald & Woodward Publishing Company, Granville
Williams N, Sweetman J (2018) Effects of neonicotinoids on the emergence and composition of chironomids in the Prairie Pothole Region. Environ. Sci. Pollut. Res. 1–12
Wolfram J, Stehle S, Bub S, Petschick LL, Schulz R (2019) Insecticide risk in United States surface waters: drivers and spatiotemporal modeling. Environ Sci Technol. https://doi.org/10.1021/acs.est.9b04285
Žabar R, Komel T, Fabjan J, Kralj MB, Trebše P (2012) Photocatalytic degradation with immobilised TiO2 of three selected neonicotinoid insecticides: imidacloprid, thiamethoxam and clothianidin. Chemosphere 89:293–301. https://doi.org/10.1016/j.chemosphere.2012.04.039
Acknowledgements
This research was funded through a cooperative agreement with the Missouri Department of Conservation (MDC) and the Missouri Cooperative Fish and Wildlife Research Unit which is jointly sponsored by MDC, the University of Missouri, the US Fish and Wildlife Service, the US Geological Survey, and the Wildlife Management Institute. We would like to thank W. Boys who was instrumental in early project development and data collection. Additionally, we thank J. Murray and the many laboratory technicians for diligently processing hundreds of invertebrate samples. We further thank D. Snow and the University of Nebraska’s Water Science Laboratory who assisted with chemical methodology. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the US Government. The authors declare no competing interests.
Funding
This research was funded through a cooperative agreement with the Missouri Department of Conservation (MDC) and the Missouri Cooperative Fish and Wildlife Research Unit which is jointly sponsored by MDC, the University of Missouri, the US Fish and Wildlife Service, the US Geological Survey, and the Wildlife Management Institute.
Author information
Authors and Affiliations
Contributions
E.B.W and D.M conceived the idea for and designed the study. K.J.K collected data, performed the data analysis, and wrote the first draft of the manuscript. E.B.W, D.M, and A.R.M provided guidance throughout data analysis and writing of earlier manuscript drafts.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Alexandros Stefanakis
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kuechle, K.J., Webb, E.B., Mengel, D. et al. Seed treatments containing neonicotinoids and fungicides reduce aquatic insect richness and abundance in midwestern USA–managed floodplain wetlands. Environ Sci Pollut Res 29, 45261–45275 (2022). https://doi.org/10.1007/s11356-022-18991-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-022-18991-9