Abstract
Participants in the coastal socio-economy of the Mediterranean Sea, such as industries, aquaculture, urban populations, conglomerates, and tourists, create intense anthropogenic pressures on marine ecosystems (such as the release of trace metals). This raises concerns about their impact on the surrounding environment and on marine organisms, including those collected for human consumption. This study introduces the possibility of using Patella caerulea (Linnaeus 1758), indigenous to the Mediterranean Sea, as a biosentinel of marine pollution. This study proposes coupling environmental (bioaccumulation) and toxicological (redox homeostasis) measures of bioavailability with genetic variability (COI mtDNA) assessments. Concentrations of six trace metals (cadmium, copper, iron, lead, nickel, and zinc) were measured in surface seawater and in P. caerulea individuals collected from four coastal stations on the Tunisian coast where different levels of metal contamination have occurred. The quantified biomarkers involved the determination of antioxidant defense enzymes, catalase (CAT), glutathione peroxidase (GPX), superoxide dismutase (SOD), and the measurement of lipid peroxidation indicated by malondialdehyde (MDA) levels. Our study identified critical levels of metal contamination among locations in the Gulf of Gabes. Concomitantly, the induction of antioxidant biomarkers (especially SOD and GPX) was observed, highlighting the potential of P. caerulea to acclimate to stressful pollution conditions. Molecular analysis of COI (mtDNA) revealed low discrimination between the four P. caerulea populations, highlighting the role of marine currents in the Mediterranean Sea in the dispersal and passive transportation of limpet larvae, allowing an exchange of individuals among physically separated, P. caerulea populations.
Similar content being viewed by others
Introduction
Most coastal developments in the Mediterranean do not address their long-term impact (WWF 2020). More than 50% of the human population of Mediterranean countries is concentrated on the coast. The region attracts more than 30% of world tourism and the trend is increasing (WWF 2020). Moreover, the Mediterranean Sea has (MS) a semienclosed configuration, inducing circular marine currents (counterclockwise from the Strait of Gibraltar) (El-Geziry and Bryden 2010). Therefore, the MS is considered one of the first seas suffering harm from anthropogenic activities, such as high metal emissions (Milano et al. 2012; Guittonny-Philippe et al. 2014). In Tunisia, coastal areas host 64% of the total human population and concentrate most of the country’s economic activities (WWF 2020). Several towns (Sousse, Monastir, Sfax, Gabes) are located along the coast, inducing intense anthropogenic pollution (urbanization, industrialization, and agricultural activities), often including metals (Ladhar-Chaabouni et al. 2012; Ghannem et al. 2014; Harrabi et al. 2018).
During recent decades, anthropogenic activities (such as industry, landfills, wastewater, and mineral extraction) have increased environmental metal emissions (Nriagu 1996), promoting their accumulation within the MS hydrological system (Debnath et al. 2021). Many studies have reported high metal contamination levels in rivers, oceans, and seas (Wang et al. 2013; Kaste et al. 2018; La Colla et al. 2021).
Metal pollution in marine ecosystems is currently among the chief environmental concerns. Metals affect the biotic community either through genetic alteration of the population (including genetic bottlenecks and species extinctions) or via physiological disruptions (e.g., growth and reproduction) (Belfiore and Anderson 2001; Ross et al. 2002; Kim et al. 2003; Matson et al. 2006; Ensibi et al. 2015; Rumisha et al. 2017). This is particularly true for species having a benthic period in their life cycle, including fish and many invertebrates that are in contact with bioavailable metals in sediments and surface waters (Matson et al. 2006). High Cd, Cu, Fe, Pb, and Zn concentrations are commonly reported in sediment, water, and biotic ecosystem components (Ansari et al. 2004; Mezghani-Chaari et al. 2015; Ayari et al. 2016). However, metals, such as Cd, Cr, Hg, Ni, and Pb, can be toxic to marine organisms even at very low concentrations (Pavalaki et al. 2016). Conversely, other metals, such as Fe, Cu, Mn, Se, and Zn, not only are essential for hormesis but also can be ecotoxic at excessive bioavailable concentrations (Calabrese and Baldwin 2003).
Trace elements persisting in ecological niches (e.g., sediments, water, or marshes) accumulate in organisms (bioaccumulation) and are passed upward through trophic networks to top predators (biomagnification) (Readman et al. 1993; Ettajani et al. 2001). Hence, these environmental contaminants are considered a threat not only to marine biodiversity but also to humans through the ingestion of contaminated marine food products (Amiard et al. 2008; Rabaoui et al. 2013; Bashir et al. 2020).
In addition to chemical measurements, biological approaches using recognized biosentinels can assist in assessing environmental risks to various species caused by pollution (Reguera et al. 2018; Louzon et al. 2020, 2021).
The most widely used species for assessing marine pollution are bivalves, such as mussels and oysters (Mytillus sp. and Crassostrea sp.), because they are easy-to-obtain filter feeders that bioaccumulate pollutants and are abundant on most coasts worldwide (Beliaeff et al. 1998; Cantillo 1998; Solé et al. 2000; Oros et al. 2005; Viñas et al. 2018). However, bivalves can be rare in some areas, underscoring the need for alternative biosentinel species especially those with different sensitivities to pollutants, belonging to different trophic levels, and exhibiting different paths of exposure (Reguera et al. 2018). In recent years, studies on marine gastropods have contributed to a better knowledge on metal accumulation in these species and simultaneously have allowed the evaluation of human health risks resulting from their consumption (Ahn et al. 2002; Bergasa et al. 2007; Yüzereroğlu et al. 2010; Shefer et al. 2015; Conti et al. 2017; Barchiesi et al. 2020).
Among intertidal grazers inhabiting rocky shores, limpets (Patella sp.) present increased opportunities for assessing environmental and toxicological bioavailability and impact (De Pirro et al. 2001; Reguera et al. 2018). Interest in the use of these organisms in biomonitoring programs is based on their wide distribution in the Northeast Atlantic and the Mediterranean (Poppe and Goto 1991; Jenkins et al. 2001), well-documented biology (Fernández et al. 2016; Reguera et al. 2018), abundance, and sedentary lifestyle (Fretter and Graham 1972). In addition, these grazer gastropods play a key role in coastal ecosystems by regulating the degree of algal coverage and even succession processes in rocky intertidal communities (Coleman et al. 2006; Reguera et al. 2018; Palladino et al. 2021). Lozano-Bilbao et al. (2021) studied the ability of P. aspera and P. candei crenata to accumulate trace metals, and these two limpet species were found to be useful as bioindicators of marine pollution based on their propensity for bioaccumulating metals, especially cadmium (Cd) and copper (Cu). Patella caerulea is a common wild species abundant on exposed shores of all Mediterranean coasts (Della Santina and Chelazzi 1991; Palladino et al. 2021). Consequently, the study of pollutant accumulation in this species can be relevant for predicting the potential transfer of contaminants through food webs by predation on contaminated limpets (Viñas et al. 2018).
Advances in cell biology support possibilities for assessing the biological and ecological significance of contaminants based on biological responses, or biomarkers, induced by exposure to xenobiotics (Douhri and Sayah 2009; Al Kaddissi 2012). Consequently, biomarkers are early warning tools that allow earlier detection of pollutants and their biological effects before perturbation at the level of entire marine communities (Moreira and Guilhermino 2005; Douhri and Sayah 2009). Biomarkers are measured in species capable of accumulating contaminants (e.g., biosentinels) (Al Kaddissi 2012). To date, only biomarkers of exposure have been studied in P. caerulea (cytochrome P450 enzymes (Yawetz et al. 1992; Bonacci et al. 2007) and ethoxyresorufin-O-deethylase (EROD) (Bresler et al. 2003)). Studies of pollution effects at the organismic level have been rare, although interest has been shown in integrated biomarker response (IBR) approaches for various biosentinel organisms used in ecotoxicology (Beliaeff and Burgeot 2002). In addition, these IBRs present a growing potential (Mleiki et al. 2020) for improving ecotoxicological risk assessment methodologies with pertinent biosentinels, such as P. caerulea.
Bioavailable metals can induce the production of reactive oxygen species (ROS) (O2−, OH−, NO−, H2O2, HOCl, O2, ONOO−) in living cells (Vlahogianni et al. 2007). H2O2 is considered a primary cellular precursor of the hydroxyl radical (HO.) and consists of a highly reactive and toxic form of ROS. Its removal is considered an important defense strategy against oxidative stress in marine organisms (Regoli et al. 2002a; Vlahogianni et al. 2007). To counteract the negative effects of ROS (lipid peroxidation, protein and DNA damage, etc.), marine organisms exhibit multiple enzymatic antioxidant defense systems (Legeay et al. 2005). The antioxidant activity of catalase (CAT) has great significance, as it prevents the conversion of Η2Ο2 to harmful OH- (Cutler 1984). CAT breaks hydrogen peroxide down into water and oxygen. This reaction is an important strategy of marine organisms against oxidative stress (Regoli et al. 2002a, 2002b). Superoxide dismutase (SOD) breaks down hydrogen peroxide and hydroperoxide radicals to harmless molecules (H2O2/alcohol and O2) (Silvestre 2005). Glutathione peroxidase (GPx), which is a selenium-containing antioxidant enzyme, effectively reduces H2O2 and lipid peroxides to water and lipid alcohols, respectively, and oxidizes glutathione to glutathione disulfide (Touyz 2004). Oxidative stress can also result in lipid peroxidation, which can be used as a biomarker (Mylonas and Kouretas 1999). Malondialdehyde (MDA) is the main active aldehyde resulting from the peroxidation of polyunsaturated fatty acids from membranes and, therefore, is used to predict cellular damage caused by ROS (Vlahogianni et al. 2007). Although we know the mechanisms of homeostasis perturbation in marine mollusks, understanding the relationship between their induction and the environmental bioavailability of metals in P. caerulea is needed.
To provide this knowledge, the first aim of this study is to look for relationships between known environmental bioavailability of metals (i.e., measured bioaccumulation) and the induction of toxicological effects using a multi-marker approach based on measures of Redox homeostasis (CAT, SOD, GPX, and MDA). The goal was to evaluate the usefulness of this species in environmental risk assessment on Mediterranean coasts. The first step toward achieving these objectives was to determine, by investigating mitochondrial COI variability, that all limpets used in the study belong to the species P. caerulea, thereby ensuring the relevance of conclusions regarding this species, and to evaluate whether distinct genetic variability patterns may exist between limpets at the different stations according to local metal pollution.
Materials and methods
Description of the stations
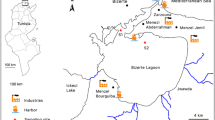
Among the 10,000 industrial companies officially registered in Tunisia that are categorized as highly polluting, 12% of these are located in Tunis, Bizerte, Sousse, Mahdia, Sfax, Gabes, and Gafsa (INS 2013) (Fig. 1). Among the polluting industries, large-scale phosphate production plants based in Gabes (and previously in Sfax), discharging approximately 12,000 tons of phosphogypsum per day, are a significant threat to the equilibrium of the marine ecosystem in Gabes city. And, beyond this, since 1970, all of the bays surrounding the Gulf of Gabes have been increasingly impacted by expanding industrialization (Bejaoui et al. 2004; Ghannem et al. 2010; Drira 2018).
Sampling sites localization (QGIS 3.0.2). The most urbanized areas in Tunisia are mentioned in bold (GPS coordinates for sampling stations are summarized in Table S1)
Stations for this study were selected after ensuring that the selected species was present and sufficiently abundant to sample (Palladino et al. 2021). In addition, the sampling locations represent sites of ecological and economic importance with varying degrees of exposure to contaminants from discharges from neighboring urban, industrial, and agricultural regions (Amari 1984; Ben charrada 1997; INS 2013). Hence, the strategy in choosing the sampling sites assumed a gradient of pollution proceeding from north to south (Amari 1984). Four stations were selected along the Tunisian coast to include different levels of water contamination (Table 1). Station locations, from north to south, included Cap Bon (CB), belonging to the gulf of Tunis, and three distant stations belonging to the gulf of Gabes: Chebba (C), Kerkennah Islands (K), and Gabes (G) (Fig. 1). The GPS coordinates of the sampling stations are provided in the supplementary information (Table S1).
The physico-chemical properties of the water at each station were measured using a CTD multiparameter probe, YSI Pro30 CTD handmeter, as suggested by the UNESCO protocols (UNESCO 1994; UNEP/MAP/MED 2020) and also as described in ISO 10523 (2008) (Table S2). Triplicate sampling of surface seawater was performed for each station in March 2018 for the analysis of trace metals. Samples were filtered using a thin membrane (0.45 μm), acidified and stored at 4 °C for metal analyses using absorption flame emission spectroscopy (AAS) (Kremling 1976). The limits of detection (LODs) and limits of quantification (LOQs) for each metal are mentioned (Table 1). The levels of metals detected at each station (Table 1) were indicative of anthropogenic activities. Moreover, the precision and accuracy of the analytical method were validated by including certified standard reference material TORT-1 Lobster Hepatopancreas (National Research Council of Canada) (Table S3).
Sample handling
Marine gastropods accumulate pollutants, including heavy metals, from the water column and via the ingestion of contaminants accumulated in phytoplankton or adsorbed on detritus and on sediment particles. Because they occupy a benthic niche, they present a relevant model to reflect local pollution to which other taxa may be exposed including, for example, crustaceans, arthropods, corals, and free-swimming finfish (Wang et al. 1996; Griscom et al. 2000).
At each sampling station, individuals of the marine gastropod P. caerulea were collected by hand at low tide and transported in cool boxes (4 °C) to the laboratory. At least 30 individuals were sampled in each station.
Surface seawater was sampled likewise at the four stations. Seawater samples were collected in triplicate in sampling bottles at each station at a 1-m depth according to HELCOM (2019) guidelines for sampling requirements. Once in the laboratory, limpet samples were rinsed in previously filtered seawater to remove the residues of sediment and particles present in the mantle and on the visceral masses. Samples were stored at − 20 °C until measurements for bioaccumulation (“Trace metals assessment” section) and enzymatic activity (“Enzymatic biomarkers” section) were conducted in the laboratory of Biodiversity and Aquatic Ecosystems (BEA) in the Faculty of Science at Sfax University in Tunisia. Individuals used for molecular analysis (“Molecular identification of Patella caerulea” section) were stored in 70% ethanol (Pereira et al. 2011) until being transported to France for further analysis in Mer Molecules Santé laboratory (MMS) at Le Mans University in France.
Laboratory analyses
Once the samples were defrosted, they were dissected on ice plates (4 °C). Calcareous shells were gently removed using a grooved probe. Before being homogenized, samples of soft tissues were weighed (Average wet weight = 2.3 ± 0.3 g). Six samples were divided into two equal aliquots of 1.0 g per sample, one for the determination of trace metals and the other for further oxidative stress biomarker analysis. As the visceral masses were too small to allow measurements in different parts of the body, all the soft parts of P. caerulea were considered when analyzing trace metals and enzymatic biomarkers (Cravo et al. 2004; Rabaoui et al. 2013).
Molecular identification of Patella caerulea
Molecular identification of samples was conducted in the MMS laboratory at Le Mans University in France. Total genomic DNA of ten selected individuals per station was extracted from the soft bodies of samples preserved and transported to France in 70% ethanol. For DNA analyses on the individual specimens, the shell was gently removed with a sterile grooved probe, and individuals were dissected using a sterile scalpel blade. DNA extraction was performed according to the hexadecyltrimethylammonium bromide (CTAB) method adapted from Doyle and Doyle (1987). The DNA was then visualized in a 1% agarose gel, and the DNA concentration was evaluated using a NanoDrop spectrophotometer, Thermo scientific (2000). DNA identification was conducted on each individual specimen used in this study based on the mitochondrial COI sequence. The COI amplifications (481 bp) respected the following conditions: 150 ng DNA added to GoTaq®DNA polymerase kit (M3001, Promega), for PCR conducted in 25 µL total volume and including 35 cycles (1 min, 94 °C; 1 min, 49.5 °C; 1 min, 72 °C) and final elongation (10 min, 72 °C). The primers used, COIpatellaF (5′-GAATATGAGCAGGTTTAG-3′, Tm: 49.1 °C, 38.9% GC) and COIpatellaR (5′-CRGCTAAAACAGGTAAWG-3′, Tm: 50.3 °C, 41.7% GC), were designed from a multiple alignment of COI sequences of several Patella species (P. aspera: KU566778, P. caerulea: EU073896, P. depressa: KU566787, P. ferruginea: GQ469876, P. lugubris: EU073889, P. pellucida: KU566788, P. rustica: GQ469882, P. ulyssiponensis: GQ469889; P. vulgata: KU566795). The sequences were aligned using Clustal Omega (https://www.ebi.ac.uk/Tools/msa/clustalo/). The phylogenetic tree was built using MEGA X.10.1.8 software (Kumar et al. 2018). Genetic variability was estimated by the nucleotide diversity (π) and the number of variable sites (S) using DNASP 5.0. (http://www.ub.edu/dnasp/). Tajima’s D test was also performed to evaluate the hypothesis that all mutations were selectively neutral.
Trace metals assessment
Measurements of trace metals were conducted in the laboratory of Chemistry in the Faculty of Science at Sfax University in Tunisia. Six samples per location were analyzed. For each limpet sample, an amount of 1.0 g was dissected using a grooved probe and a scalpel blade, and the samples were oven dried overnight at 60 °C in porcelain crucibles to remove moisture and thereafter were weighed (average weight 0.23 ± 0.01 g) (Cravo and Bebianno 2005). The percentage moisture was calculated as the loss of weight after drying. The remaining 1.0 g portions were labeled and stored at − 20 °C until enzymatic analyses were conducted. The dried samples were placed in a muffle furnace at 500 °C for 12 h to induce calcination (Fonollosa et al. 2017; Primost et al. 2017). The ashes obtained were filtered using a solution of nitric acid HNO3 (1%) adjusted to 30 mL with deionized water. Metal concentrations of these samples were determined using atomic absorption flame emission spectroscopy (AAS) Perkin Elmer, AAnalyst 200 (SAAF) using the (UNEP/IAE/FAO 1984) method. The targeted trace metals are cadmium (Cd), copper (Cu), iron (Fe), lead (Pb), nickel (Ni), and zinc (Zn). The accuracy and precision of the analytical methods were assessed by the analysis of one aliquot of reference material, as well as one laboratory reagent blank. All samples were analyzed in triplicate, and the average metal concentration was calculated. The blank did not contain any detectable concentration of the studied metals. Recovery rates for metals were determined with certified reference materials (TORT-1, lobster hepatopancreas) between 90 and 100%, indicating the absence of analytical bias (Table S3).
Enzymatic biomarkers
Measurements of enzymatic biomarkers were conducted in the laboratory of Biodiversity and Aquatic Systems in the Faculty of Science at Sfax University in Tunisia. The labeled 1.0 g portions left from each sample used for metal assessment (six analyzed samples per station) were thawed on ice and then ground (special electric homogenizer ULTRA TURRAX) in 2 ml of tris-buffered saline (TBS) (pH = 7.4). The homogenate was centrifuged twice at 10,000 × g at 4 °C for 20 min, and the collected supernatants were kept at − 20 °C for further short-term measurements of enzymatic activities. Proteins were quantified as mg/ml according to the Lowry method (Lowry et al. 1951) using a bovine serum albumin (BSA) calibration curve (R2 = 1). Absorbance was measured at 490 nm. SOD activity was measured indirectly by the colorimetric method developed by Beauchamp and Fridovich (1971) using the riboflavin/methionine complex producing superoxide anions and nitro blue tetrazolium (NBT). NBT oxidation by the superoxide O2− anion was measured at 560 nm, and the results are expressed as units (U) of SOD activity.mg−1 protein. CAT activity was measured following the decrease in absorbance at 240 nm due to H2O2 consumption (Aebi 1984). The reaction took place in phosphate buffer (100 mM) at pH 7.0 at 25 °C with H2O2 (500 mM) and total protein (1–1.5 mg protein/ml). The CAT activity is the difference in the absorbance at that wavelength per unit of time (µmoles/min/mg protein) according to the protocol proposed by Aebi (1984). Total GPx activity was measured at 412 nm according to the protocol described by Flohé and Günzler (1984). The supernatant (v) was mixed with 1 mM GSH (2v). H2O2 (1.3 mM) was added to initiate the reaction. The reaction was stopped after 10 min by the addition of 1% trichloroacetic acid (TCA). After centrifugation, Na2HPO4 (320 mM) and Ellman’s reagent (5,5′-dithiobis-(2-nitrobenzoic acid)) (DNTB) (1 mM) were added to the supernatant. GPx activity was expressed in μmoles of oxidized GSH/min/mg protein.
Lipid peroxidation
The malondialdehyde (MDA) level in limpets was measured using the protocol defined by Niehaus and Samuelsson (1968). Briefly, 0.5 mL of tissue supernatant from a previous experiment (“Enzymatic biomarkers” section) was mixed with TBS and TCA (20%)-BHT (0.01%) and incubated at 100 °C for 30 min. Samples were then vortexed, cooled, and finally centrifuged at 10,000 × g for 10 min. Then, 1 mL of thiobarbituric acid (TBA) solution (0.67%), 40 µl of HCl (0.6 M) and 0.5 mL of supernatant were incubated for 15 min at 90 °C. Finally, the absorbance of the TBA–MDA complex was measured at 532 nm. The MDA concentration was calculated using its molar extinction coefficient (ε = 155 mM/cm). MDA was expressed in nmoles/mg protein.
Data analysis
Stations are presented in maps drawn with QGIS software version 3.0.2. Data analysis was performed with R studio (version 4.0.3). Data are presented as the mean ± standard deviation. Normality and homogeneity of the variance were checked with the Shapiro–Wilk test and Bartlett test, respectively. Comparison of contamination degrees of the studied stations and metal concentrations was conducted using one-way ANOVA followed by a post hoc test (Tukey HSD). The differences were considered significant when the p value was less than 0.05.
Results and discussion
Molecular identification and genetic diversity of Patella caerulea
The evolutionary history is inferred by using the maximum likelihood method and the Hasegawa-Kishino-Yano model (Hasegawa et al. 1985). The tree with the highest log likelihood (− 1709.28) is presented in Fig. 2. The percentage of trees in which the associated taxa clustered together is shown next to their respective branches. Initial tree(s) for the heuristic search were obtained automatically by applying neighbor-joining and BioNJ algorithms to a matrix of pairwise distances estimated using the maximum composite likelihood (MCL) approach and then selecting the topology with superior log likelihood value. A discrete gamma distribution was used to model evolutionary rate differences among sites (5 categories (+ G, parameter = 1.5614)). The rate variation model allowed some sites to be invariable evolutionarily ([+ I], 54.13% sites). Codon positions included were 1st + 2nd + 3rd + Noncoding. There was a total of 353 positions in the final dataset. The phylogenetic tree (Fig. 2) showed that all individuals used in this study were included in the P. caerulea group (robust monophyletic group), confirming the species identity across all sample stations. The nucleotide diversity (π: 0.00381), the number of polymorphic sites (S: 19 on 481 sites) and Tajima ‘D statistic (D: − 1.73907) indicated a low intraspecific polymorphism. The haplotype network of COI (481 bp) sequences of P. caerulea inhabiting the Mediterranean Sea (Fig. 3) highlights the presence of shared haplotypes (haplogroups) between individuals from at least four different countries. Tunisian individuals are found in shared haplotypes (for example, with individuals from Morocco, Greece, and Spain) but also in separate haplotypes. This seems to show a great mixing of populations within the semienclosed environment of the Mediterranean. This low P. caerulea variability could be explained by marine currents in the Mediterranean Sea, which rotate counterclockwise and allow larval limpet dispersion along the Tunisian coasts (El-Geziry and Bryden 2010), transporting them far from their place of origin and thus creating genetic mixing between Mediterranean P. caerulea populations. This result of shared haplotypes between individuals from different Mediterranean locations is in agreement with the findings of Wesselmann et al. (2018) on Pinna nobilis, who highlighted the importance of ocean currents and pelagic larval transport time in shaping the population connectivity of the species in the Mediterranean basin. This genetic connectivity has also been described for several Mediterranean fish species (Galarza et al. 2009).
Evolutionary relationships (maximum likelihood method, HKY + G + I model, MEGA X, 100 bootstrap) of Patella sp. from the Mediterranean Sea based on COI information (543 bp). The codes of Tunisian P. caerulea sequences are B (Bizerte), C (Chebba), G (Gabes) and K (Kerkennah). Littorina littorea (marine gastropod, Littorinidae) was used as the outgroup
Haplotype network of COI sequences of Patella caerulea inhabiting the Mediterranean Sea. Each circle represents a haplotype, and the size of circles corresponds to the number of individuals within the haplotype and the number of haplotypes within one haplogroup. Connection lines between circles represent mutations. Colors indicate separate locations (COI = 481 pb). Map generated with Networks
Metal concentrations in limpets
The average concentrations and the bioaccumulation factors (BAFs) (calculated as the ratio between concentrations in limpets and concentrations in water samples for the same metal) of each metal assessed in the soft tissues of limpet P. caerulea from different stations are summarized in Table 2.
Concerning the variation between stations, most of the highest concentrations were found in limpets from Gabes city, for Cu (0.06 ± 0.07 µg/g DW), Cd (0.23 ± 0.21 µg/g DW), Ni (0.60 ± 0.25 µg/g DW), Pb (1.29 ± 0.36 µg/g DW), and Zn (8.21 ± 7.48 µg/g DW) (Table 2), but not for iron, which was slightly higher in limpets belonging to Cap Bon station (9.10 ± 4.72 µg/g DW) than in those coming from Gabes (8.03 ± 5.71 µg/g DW), although there was no significant difference (p value = 0.2 > 0.05). This is because Gabes city is under strong anthropogenic aggression from the phosphoric acid industry (Bejaoui et al. 2004; Ghannem et al. 2010; Drira 2018).
On the other hand, the lowest mean values of the studied metals were scattered among the different stations. Fe, Pb, and Zn (0.73 ± 0.31, 0.21 ± 0.11, and 1.54 ± 1.24 µg/g DW, respectively) were found at Chebba station, and Cu and Ni (0.014 ± 0.01 µg/g DW for Cu and under the limit of detection (0.07) for Ni, respectively) were found at Cap Bon station (Table 2).
Cd, which is considered one of the most toxic heavy metals (Burger 2008; Rabaoui et al. 2013), was detected only in limpets from Gabes station (0.23 ± 0.21 µg/g DW) with a BAF = 1.
The highest concentrations of iron (9.10 ± 4.72 µg/g DW) and second highest concentration of lead (1.08 ± 0.37 µg/g DW) were recorded in limpets collected at Cap Bon station but this station was presumed to be the control site (Belkhodja et al. 2012).
Significant interstation differences were noted for each trace metal assessed in P. caerulea, except for copper, which did not present any difference between stations and which was represented by the lowest concentrations found in limpet tissues from each location, with 0.014 ± 0.01 µg/g DW at Cap Bon station, 0.03 ± 0.04 µg/g DW at Chebba station, 0.04 ± 0.03 µg/g DW at Kerkennah Islands station, and 0.06 ± 0.07 µg/g DW at Gabes station. Considering the values of BAFs, copper also presented the lowest BAFs at all the studied stations (0.004 at both the Chebba and Cap Bon stations, 0.023 at the Gabes station, and 0.007 at the Kerkennah station). However, Zn and Fe appear to have the highest BAFs among all the studied metals per studied station except for Gabes station, where the highest BAFs were represented by Zn and Cd.
Comparing our results (Table 3) with levels reported for other geographical areas for P. caerulea and other Patella species (Table 3), cadmium concentrations recorded within the present study are the lowest compared to the results of previous studies in the Gulf of Gabes (0.605 ± 0.0252–1.065 ± 0.0858 µg/g DW) by Rabaoui et al. (2013), in the north coast of Tunisia (0.78 ± 0.24–1.63 ± 0.27 µg/g DW) by Belkhodja and Romdhane (2013), in Iskenderun Gulf in Turkey (0.24 ± 0.03–0.68 ± 0.03 µg/g DW) by Yüzereroğlu et al. (2010), and in Italian islands (3.30 ± 0.37–6.30 ± 2.08, 1.7–11.8 and 0.104 ± 0.03–2.318 ± 2.99 µg/g DW) by Campanella et al. (2001), Cubadda et al. (2001) and Conti et al. (2017), respectively. However, our Cd levels were higher than those determined in the Ionian Sea in Italy (0.15 ± 0.06–0.16 ± 0.05 µg/g DW) by Storelli and Marcotrigiano (2005). Studies on other limpet species showed lower Cd concentrations for Patella piperata in the Canary Islands (0.039 ± 0.011 µg/g DW) by Bergasa et al. (2007) and for Patella candei and Patella aspera (0.041 ± 0.013–0.102 ± 0.011 µg/g DW, respectively) by Lozano-Bilbao et al. (2021). Based on the above-mentioned results, Cubadda et al. (2001) and Yüzereroğlu et al. (2010) suggested that limpets of the genus Patella may be suitable species for biomonitoring Cd availability. However, the data from the study of Espinosa and Rivera-Ingraham (2016) on Patella ferruginea in Ceuta, Spain, do not fit with previous reports for Cd, suggesting that P. ferruginea was an exception since the Cd concentrations detected in soft tissues were independent of the existing pollution gradient.
Belkhodja et al. (2012) considered Cap Bon station as a control station in their study on limpets in 2012. The present study was conducted 6 years later and showed that waters around the Cap station are becoming more polluted with metals. This could be explained by the fact that this station receives domestic wastewater and effluent discharges from the three treatment plants in the surrounding areas through Wadi Soltane, Wadi Meliane, and Wadi el Bey (Ennouri et al. 2010; Belkhodja et al. 2012). For example, Wadi el Bey drains 60% of the pollutants discharged from several urban centers in northeastern Tunisia. The main sources of pollution in Wadi El Bey are tannery, stationery, brewery, tomato processing, and slaughterhouse discharges (COMETE engineering-Bceom-IHE 2008). Pollution impacts linked to increasing urbanization and tourism are significant along this part of the coast (WWF 2020).
Our range levels for lead (0.21 ± 0.11–1.29 ± 0.36 µg/g DW) were clearly lower than those reported earlier on the north coast of Tunisia (3.51 ± 0.67–3.61 ± 0.8 µg/g DW) (Belkhodja and Romdhane 2013) and on Pontine Islands in Italy (3.58 ± 0.94–0.479 ± 0.14 µg/g DW) (Conti et al. 2017), although our results for Pb were comparable with Pb ranges reported for Patella specimens in other studies (Campanella et al. 2001; Cubadda et al. 2001; Storelli and Marcotrigiano 2005; Bergasa et al. 2007; Yüzereroğlu et al. 2010; Rabaoui et al. 2013; Lozano-Bilbao et al. 2021) (Table 3).
Our range levels (Table 2) for Ni (0.11 ± 0.04–0.60 ± 0.25 µg/g DW) were clearly lower than those reported earlier in P. caerulea in contaminated areas from the north coast of Tunisia (Belkhodja et al. 2012) and on Pontine Islands in Italy (Conti et al. 2017). However, our range levels for Ni were generally comparable to those reported for P. candei and P. aspera in the Canary Islands (Lozano-Bilbao et al. 2021) and for P. caerulea in the Iskenderum Gulf in Turkey (Yüzereroğlu et al. 2010) (Table 3).
Our range levels for Cu are clearly lower than those reported in Patella specimens from other geographic areas (i.e., those previously reported in P. caerulea in the Italian Islands by Campanella et al. (2001) and Cubadda et al. (2001) and in P. candei and P. aspera in the Canary Islands by Lozano-Bilbao et al. (2021)) (Table 3). The lower-than-expected differences between our copper levels assessed in tissues from areas potentially expected to have varying degrees of pollution can be explained, as suggested by Cubadda et al. 2001, by the strong capacity of the species P. caerulea to regulate their body levels of copper. This regulatory mechanism in marine invertebrates might involve enhanced metal elimination, resistance to metal uptake, or some combination of the two, or storing excess metals in a nontoxic form until further use (Depledge and Rainbow 1990). However, laboratory experiments of copper exposure of P. aspera, P. caerulea, and P. rustica did not mention any copper regulation ability in the tested individuals, even though they showed interspecific differences in short-term copper intake and excretion dynamics (De Pirro et al. 2001; Reguera et al. 2018). This may also be linked to the stressful effect generated by cadmium (BAF = 1) as a replacement for copper in various proteins, such as ferritin, by promoting its release from biological membranes, thus contributing to an increase in its intracellular concentrations (Casalino et al. 1997; Dorta et al. 2003; Watjen and Beyersmann 2004; Al Kaddissi 2012).
Our results for Zn and Fe concentration ranges were generally comparable to those reported for Patella specimens in other localities (Table 3), while for Fe, concentration ranges were lower than those recently reported in P. candei and P. aspera on Canary Islands (Lozano-Bilbao et al. 2021), and for Zn, concentration ranges were lower than those reported in P. caerulea in Pontine Islands, Italy, (Conti et al. 2017) and along the Iranian coasts (Bordbar et al., 2015). High bioaccumulation levels of Zn (Table 2), especially at the Gabes station (BAF = 1.546), can be explained by the presence of large quantities of Zn in the Mediterranean environment and in sediments (Drira 2018). However, Devez (2004) showed that zinc accumulation is not necessarily correlated with environmental contamination. Another possibility is that this metal is a biologically essential element in sea organisms (Trevizani et al. 2016).
Studies on limpets indicate that, regardless of environmental quality, these animals tend to preferentially accumulate Fe and Zn (Cravo and Bebianno 2005; Belkhodja et al. 2012; Espinosa and Rivera-Ingraham 2016), and, as seen in the present study, P. caerulea is not an exception (highest concentrations in limpets and highest BAFs are recorded with these two metals (Table 2)). Apparently, the high concentration of Fe in marine invertebrate soft tissues is not surprising (Espinosa and Rivera-Ingraham 2016), and for the specific case of limpets, their teeth (radula) are 1.46% formed by an iron-based mineral material called goethite (Davies and Cliffe 2000). Zn, on the other hand, easily accumulates in soft tissues through passive uptake and is a constituent of metabolically significant enzymes and pigments, along with Fe (Rivera Ingraham et al. 2013).
Limpets represent the most important herbivorous grazers of temperate rocky shores (Palladino et al. 2021). They accumulate pollutants, including heavy metals, from the water column and via the ingestion of contaminants accumulated in phytoplankton or adsorbed on detritus and on sediment particles (Wang et al. 1996; Griscom et al. 2000). Therefore, it would be important to consider food composition and contaminant bioavailability in resources when studying bioavailability for limpet species, as recommended by Espinosa and Rivera-Ingraham (2016). Ozaki (2019) demonstrated that diet richness and/or composition can be affected by pollution-induced changes in trophic resources and that the type of food items consumed by mice affected their trophic exposure to trace metals. In the same context of factors determining the environmental bioavailability of contaminants, Espinosa and Rivera-Ingraham (2016) showed that females of P. ferruginea in Spain presented the highest values for trace metals, and this result was due only to sex and not to the size differences between males and females. Lozano-Bilbao et al. (2021) showed that environmental bioavailability can also be species-dependent when they compared the ability of accumulating metals between two limpet species and revealed that P. aspera showed a higher concentration of elements than P. candei crenata. From this perspective, in future studies considering the influence of various ecophysiological factors on metal bioaccumulation, it would be relevant to normalize the sampling protocol and suggest threshold guide values (TGVs) in assessing what may be considered abnormal internal concentrations in Patella, as well as the environmental bioavailability of metals, such as those existing for Gastropoda subadults in terrestrial ecosystems (ISO 24032:2021; Louzon et al. 2021).
Measurement of antioxidant responses and lipid peroxidation levels in P. caerulea
Protein quantification
The statistical analysis of total levels of protein in P. caerulea did not reveal any significant difference between stations, except for Gabes station (p > 0.05), where the highest protein concentration (8.33 ± 4.48 mg/ml) was reported, which seems similar to the cadmium contamination level at the same station. This agrees with the findings of Khebbeb et al. (2010), who noted a positive correlation between total levels of protein and cadmium concentration in the clam Ruditapes decussatus contaminated for a period of 21 days. However, Douhri and Sayah (2009) observed a protein concentration decrease in Patella vulgata collected from impacted sites in the Bay of Tangier in Morocco, resulting from their degradation or a reduction in their biosynthesis by xenobiotics (Douhri and Sayah 2009). Several studies related the measurement of the total level of protein not as a biomarker, but for the expression of enzymatic activities such as CAT, SOD, and GPx (de Almeida et al. 2004; Attig et al. 2010; Buffet et al. 2011; Soltani et al. 2012). For our study, the concentrations of proteins SOD, CAT, GPx, and MDA are represented in Fig. 4.
Antioxidant activities (Catalase: CAT/Superoxide Dismutase: SOD/Glutathione Peroxidase: GPx) and Malondialdehyde (MDA) levels in Patella caerulea (limpets, n = 6) from distinct Tunisian stations (CB: Cap Bon/C: Chebba/G: Gabes/K: Kerkennah). Error bars indicate absolute deviation from the mean. Different letters indicate statistically significant differences (P ≤ 0.05)
SOD activity
The findings, represented in Fig. 4, showed that the SOD activities (U/mg protein) measured in P. caerulea collected at the Kerkennah and Gabes stations (6 ± 1.9 U/mg protein and 5.25 ± 0.7 U/mg protein, respectively) were significantly higher (p = 0.01) than the SOD activities in limpets from Cap Bon and Chebba stations (2.9 ± 0.4 U/mg protein and 3.9 ± 0.7 U/mg protein, respectively). Zelko et al. (2002) suggested that active sites of SOD isoenzymes require the presence of catalytic metals (Cu, Fe, Mn, Ni, Zn) for their activation. This confirms our results because the highest BAFs for Cu, Fe, Ni, and Zn were recorded in limpets from Gabes and Kerkennah stations (except for BAF (Fe), which was the highest in Gabes but not in Kerkennah station (Table 2). Our results are in agreement with those of Espinosa and Rivera-Ingraham (2016) on P. ferruginea in Spain, who found significant SOD activity in the area with high levels of trace metals. Vlahogianni et al. (2007) showed the same result of increased SOD activity in mussels (M. galloprovincialis) challenged by high levels of Cu, Ni, and Zn. Several authors have shown that the increase in SOD activity in marine organisms is linked to the presence of heavy metals (Farombi et al. 2007; Fernández et al. 2010; Lushchak 2011; Batista et al. 2014). Farombi et al. (2007) explained that the accumulation of trace metals causes oxidative stress, which leads to the production of superoxide anions that induce SOD to catalytically scavenge superoxide radicals to H2O2, providing a defense against this type of oxygen toxicity.
CAT activity
No significant difference was shown for CAT activity measurements in limpets among the sites. Even though the Gabes station presented the highest CAT activity (16.32 ± 9.95 μmol/min/mg protein), it did not present any significant difference, as seen in Fig. 4.
In the same context, it was reported previously that CAT activity was lower in individuals of P. ferruginea inhabiting polluted sites (Espinosa and Rivera-Ingraham 2016). Lionetto et al. (2003) showed a similar result of no significance in the variation of CAT activity in mussels from sites exposed to the highest anthropogenic activities on Italian coasts. Similarly, da Silva et al. (2005) reported a lack of CAT activity change in the oyster Crassostrea rhizophorae exposed to diesel fuel. In contrast, individuals of P. vulgata inhabiting marine polluted areas of Moroccan coasts showed a significant increase in CAT activity (Douhri and Sayah 2009). Vlahogianni et al. (2007) and Jebali et al. (2007) also observed an increase in CAT activity in mussels (Mytilus galloprovincialis) from polluted areas of the Saronikos Gulf of Greece and in clams (Ruditapes decussatus) from Tunisian polluted marine ecosystems. In the present study, the lack of marked differences in this enzymatic activity between limpets from the different stations seems to confirm the suggestion of previous field (Regoli 1998) and laboratory (Cajaraville et al. 1997; Lionetto et al. 2003) studies that reported the possibility of compensatory mechanisms occurring in the CAT response of mussels (M. galloprovincialis) from chronically polluted populations that may not show expected biological responses. This could be the case for the present study and could explain the lack of significant differences in CAT activities among the studied stations. Another explanation by Regoli et al. (2002b) suggested that complex interactions between prooxidants and their cellular targets may confound the assessment of oxidative stress conditions based solely on antioxidant levels. Thus, it is not uncommon to see how, in the face of a certain stressor, some antioxidants increase while others remain unchanged or even decrease (Regoli et al. 2002b). Therefore, chemically induced perturbation of the cellular redox status was suggested to demonstrate cell damage as an impact of degraded environmental quality (Espinosa and Rivera-Ingraham 2016).
Other studies found that CAT activity and Fe concentration are interdependent because iron is an essential element of the active center of some proteins (Jurczuk et al. 2004; Al Kaddissi 2012).
GPx activity
Similar to SOD, the results (Fig. 4) showed that GPx activities (nmoles GSH/min/mg of proteins) measured at Gabes (9.77 ± 1.9 nmoles GSH/min/mg of proteins) and Kerkennah (10.85 ± 2.72 nmoles GSH/min/mg of proteins) stations were significantly higher than the other stations. GPx activity recorded in limpets from Cap Bon station (2.9 ± 0.8 nmoles GSH/min/mg of proteins) was significantly lower than that of all the other stations. Thus, the pollution trend from north to south is supported by the increasing GPx activity in P. caerulea from Cap Bon toward the southern stations.
Espinosa and Rivera-Ingraham (2016) reported comparable results while assessing heavy metals in the endangered limpet species P. ferruginea from the strait of Gibraltar, where individuals sampled from the most polluted areas presented the highest levels of GPx activity. Elevated GPx activity was also reported in mussels exposed to high metal pollution on the coasts of Ria de Vigo (Vidal-Liñán et al. 2014) and the North Tyrrhenian Sea (Regoli and Principato 1995).
Lipid peroxidation
Induced MDA concentrations (nmols/mg protein) were noted in all studied Tunisian stations (Fig. 4), indicating cellular stress resulting from changes in the environment, most likely through chemical contamination (Vlahogianni et al. 2007). However, no significant differences were recorded between the different stations. Our results are in agreement with those of Viarengo et al. (1991) and Pampanin et al. (2005) in Italy and Vlahogianni et al. (2007) in the Saronikos Gulf in Athens, who studied the toxic effects of heavy metals on lipid peroxidation in Mytilus galloprovincialis. They demonstrated an increase in MDA levels after metal exposure. Moreover, the absence of a significant difference between our four sampling stations is in agreement with the explanation supplied by Viarengo et al. (1991), who measured MDA in the digestive gland of mussels from Italy and showed that MDA levels fluctuate significantly according to time of year and that no significant difference in MDA activity is observed during spring (and particularly in March). This is in agreement with our findings, especially because our sampling was conducted during March. This finding is presumably linked to the seasonal metabolic status of the animals, which depends on gametogenesis (Gabbott 1975; Lowe et al. 1982) and food availability (Hawkins and Bayne 1984).
Conclusions
The Tunisian coast has increasing levels of pollution from the north (Cap Bon) to the south (the city of Gabes), with Gabes being the only station where we detected Cd in both water and animal tissue. Moreover, levels of the most toxic metals (e.g., Cd and Pb) have reached critical limits at the four investigated stations. Our results confirmed that stations belonging to the Gulf of Gabes (Chebba, Kerkennah, and Gabes) were the most polluted (the highest BAFs and highest total metal concentrations). These results may be explained either by the specific hydrodynamic features of the Gulf of Gabes, which are responsible for transporting pollutants between the different localities of the Gulf, and/or by the specific characteristics of domestic and industrial wastes. This region is industrially exploited and is the location of the largest chemical industries in Tunisia, including the phosphoric acid industry. The area is also frequented by tourists, which means that all domestic waste of urban areas, including tourist resorts, is channeled directly into the sea. Domestic waste was reported to be a source of heavy metal pollution (Wei and Yang 2010).
The present study confirms that limpets can be used as biosentinel organisms for identifying early warning responses to long-term ecological damage. It is known that risk assessment of environmental pollution cannot be based solely on chemical analysis, which does not provide a clear indication of toxic effects of xenobiotics on aquatic biota (Livingstone 2001). The interpretation of risk linked to pollution was supported by our study of biomarkers. Based on the results obtained in this study, there is a correlation between the level of pollution in the investigated sites and the response of animal-related biochemical parameters (CAT, GPx, SOD, MDA). It would be useful to measure trace metal content in sediments to identify contaminant increases in animal tissues (e.g., gills, muscle, digestive glands), especially considering ecophysiological factors (e.g., seasons, sexual maturity), in determining whether there are species-specific mechanisms involved in the bioaccumulation of trace metals. However, in the present study, the lack of marked differences in the enzymatic activity of CAT between limpets from the different stations appears to confirm the possibility of compensatory or adaptive mechanisms operating in chronically polluted marine animal populations.
Our molecular COI investigation on intraspecific variability allowed us to suggest that P. caerulea presents a genetic continuum along the Tunisian coasts, which can be explained by the Mediterranean Sea’s counterclockwise currents, resulting in larval limpet dispersion along the Tunisian coasts (El-Geziry and Bryden 2010). This transports larvae far from their place of origin, thus creating genetic mixing between Mediterranean P. caerulea populations. The same approach is being developed on genetically close but geographically distinct species to determine whether genetic variability exists between different populations exposed to the same pollution. Nevertheless, we suggest that additional experimental research should be conducted in considering factors of sex, size, and age of the individuals tested, carrying out exposure experiments in conditions of mono- or multi-metallic contamination to estimate with more precise approaches (such as qPCR and transcriptomic pattern) for understanding the genetic expression rates of stress biomarkers (CAT, HSP70, GPx, SOD, metallothioneins, etc.), and establishing lethal dose standards for various metals and species. This research is a work in progress. Limpets, being grazer gastropods, play a key role in coastal ecosystems by regulating the degree of algal coverage. Trophic resources are also susceptible to pollution, and we suggest that trophic transfers of trace metals should be considered in further works on environmental contamination and marine animals.
Overall, in the context of developing evidence, methodologies (combining chemical and biological approaches) can assist decision-makers in better managing coastal pollution. This work demonstrates that P. caerulea can be used as a good bioindicator in Mediterranean ecosystems and offers promising perspectives for the development of integrated biomarker indices and threshold guide values (TGVs) for evaluating environmental bioavailability (Beliaeff and Burgeot 2002; Ciliberti et al.2017; Louzon et al. 2021).
Data availability
All data generated and analyzed during this study are available from the corresponding author on reasonable request.
References
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ahn I-Y, Kim K-W, Choi HJ (2002) A baseline study on metal concentrations in the Antarctic limpet Nacella concinna (Gastropoda: Patellidae) on King George Island: variations with sex and body parts. Mar Pollut Bull 44(5):424–431. https://doi.org/10.1016/S0025-326X(01)00297-1
Al Kaddissi S (2012) Comparaison de la réponse (en termes d’accumulation, d’impacts cellulaires et génétiques) de l’écrevisse Procambarus clarkii après exposition à un polluant métallique (cadmium) et un polluant radiologique (uranium 238 et 233). PhD thesis, University of Bordeaux 1.
Amari A (1984) Contribution à la connaissance hydrologique et sédimentologique de la plate-forme des îles Kerkennah. PhD thesis, University of Tunis.
Amiard JC, Amiard-Triquet C, Charbonnier L, Mesnil A, Rainbow PS, Wang WX (2008) Bioaccessibility of essential and non-essential metals in commercial shellfish from Western Europe and Asia. Food Chem Toxicol 46(6):2010–2022. https://doi.org/10.1016/j.fct.2008.01.041
Ansari TM, Marr IL, Tariq N (2004) Heavy metals in marine pollution perspective — a mini review. J Appl Sci 4:1–20. https://doi.org/10.3923/jas.2004.1.20
Attig H, Dagnino A, Negri A, Jebali J, Boussetta H, Viarengo A, Dondero F, Banni M (2010) Uptake and biochemical responses of mussels Mytilus galloprovincialis exposed to sublethal nickel concentrations. Ecotoxicol Environ Saf 73(7):1712–1719. https://doi.org/10.1016/j.ecoenv.2010.08.007
Ayari J, Agnan Y, Charef A (2016) Spatial assessment and source identification of trace metal pollution in stream sediments of Oued El Maadene basin, northern Tunisia. Environ Monit Assess 188(7):397. https://doi.org/10.1007/s10661-016-5402-4
Aydın-Önen S, Öztürk M (2017) Investigation of heavy metal pollution in eastern Aegean Sea coastal waters by using Cystoseira barbata, Patella caerulea and Liza aurata as biological indicators. Environ Sci Pollut Res 24(8):7310–7334. https://doi.org/10.1007/s11356-016-8226-4
Barchiesi F, Branciari R, Latini M, Roila R, Lediani G, Filippini G, Scortichini G, Piersanti A, Rocchegiani E, Ranucci D (2020) Heavy metals contamination in shellfish: benefit-risk evaluation in central Italy. Foods 9(11):1720. https://doi.org/10.3390/foods9111720
Bashir I, Lone FA, Bhat RA, Mir SA, Dar ZA, Dar SA (2020) Concerns and Threats of Contamination on Aquatic Ecosystems. In: Hakeem KR, Bhat RA, Qadri H (eds) Bioremediation and Biotechnology: Sustainable Approaches to Pollution Degradation. Springer International Publishing, Cham, pp 1–26
Batista MTO, Rodrigues Junior E, Feijó-Oliveira M, Ribeiro AC, Rodrigues E, Suda CNK, Vani GS (2014) Tissue levels of the antioxidant enzymes superoxide dismutase and catalase in fish Astyanax bimaculatus from the Una River Basin. Rev Ambiente Amp Água 9(4):621–631. https://doi.org/10.4136/ambi-agua.1473
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44(1):276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bejaoui B, Raïs S, Koutitonsky V (2004) Modélisation de la dispersion du phosphogypse dans le golfe de Gabès. Bull Inst Natn Scien Tech Mer De Salammbô 31:103–109
Belfiore NM, Anderson SL (2001) Effects of contaminants on genetic patterns in aquatic organisms: a review. Mutat Res 489(2–3):97–122. https://doi.org/10.1016/s1383-5742(01)00065-5
Beliaeff B, Burgeot T (2002) Integrated biomarker response: a useful tool for ecological risk assessment. Environ Toxicol Chem 21(6):1316–1322. https://doi.org/10.1002/etc.5620210629
Beliaeff B, O’Connor TP, Claisse D (1998) Comparison of chemical concentrations in mussels and oysters from the United States and France. Environ Monit Assess 49(1):87–95. https://doi.org/10.1023/A:1005766321323
Belkhodja H, Romdhane M (2013) Corallina sp. and Patella caerulea (Linnaeus, 1758) as quantitative biological indicators for trace metals in the Tunisian coastal waters. Rapp Comm Int Mer Mèdit 40:815. Rapp Comm Int Mer Medit 40:815.
Belkhodja H, Missaoui H, Salah Romdhane M (2012) Trace metals in molluscs Patella caerulea (Linnaeus, 1758) from Tunisian North Coasts, Mediterranean Sea. Cah Biol Mar 53(2):243–254.
Ben charrada R (1997) Etude hydrodynamique et écologique du complexe Petit golfe – Lac de Tunis. Contribution à la modélisation de l’écosystème pélagique des eaux côtière Petit golfe de Tunis. PhD thesis, University of Tunis.
Bergasa O, Ramírez R, Collado C, Hernández-Brito JJ, Gelado-Caballero MD, Rodríguez-Somozas M, Haroun RJ (2007) Study of metals concentration levels in Patella piperata throughout the Canary Islands, Spain. Environ Monit Assess 127(1):127–133. https://doi.org/10.1007/s10661-006-9266-x
Bonacci S, Iacocca A, Fossi S, Lancini L, Caruso T, Corsi I, Focardi S (2007) Biomonitoring aquatic environmental quality in a marine protected area: a biomarker approach. Ambio 36(4):308–315. https://doi.org/10.1579/0044-7447(2007)36[308:baeqia]2.0.co;2
Bordbar L, Dassenakis M, Catsiki VA, Megalofonou P (2015) Influence of a ferronickel smelting plant activity on the coastal zone through investigation of metal bioaccumulation on two gastropod species (Patella caerulea and Phorcus turbinatus). J Environ Anal Toxicol s7:1–9. https://doi.org/10.4172/2161-0525.S7-004
Bresler V, Fishelson L, Feldstein T, Rosenfeld M, Mokady O (2003) Marine molluscs in environmental monitoring. Helgol Mar Res 57:157–165. https://doi.org/10.1007/s10152-003-0151-5
Buffet P-E, Tankoua OF, Pan J-F, Berhanu D, Herrenknecht C, Poirier L, Amiard-Triquet C, Amiard J-C, Bérard J-B, Risso C, Guibbolini M, Roméo M, Reip P, Valsami-Jones E, Mouneyrac C (2011) Behavioural and biochemical responses of two marine invertebrates Scrobicularia plana and Hediste diversicolor to copper oxide nanoparticles. Chemosphere 84(1):166–174. https://doi.org/10.1016/j.chemosphere.2011.02.003
Burger J (2008) Assessment and management of risk to wildlife from cadmium. Sci Total Environ 389(1):37–45. https://doi.org/10.1016/j.scitotenv.2007.08.037
Cajaraville MP, Orbea A, Marigómez I, Cancio I (1997) Peroxisome proliferation in the digestive epithelium of mussels exposed to the water accommodated fraction of three oils. Comp Biochem Physiol C Pharmacol Toxicol Endocrinol 117(3):233–242. https://doi.org/10.1016/S0742-8413(97)00057-1
Calabrese EJ, Baldwin LA (2003) Inorganics and hormesis. Crit Rev Toxicol 33(3–4):215–304. https://doi.org/10.1080/713611040
Campanella L, Conti ME, Cubadda F, Sucapane C (2001) Trace metals in seagrass, algae and molluscs from an uncontaminated area in the Mediterranean. Environ Pollut Barking Essex 111(1):117–126. https://doi.org/10.1016/s0269-7491(99)00327-9
Cantillo AY (1998) Comparison of results of mussel watch programs of the United States and France with worldwide mussel watch studies. Mar Pollut Bull 36(9):712–717. https://doi.org/10.1016/S0025-326X(98)00049-6
Casalino E, Sblano C, Landriscina C (1997) Enzyme activity alteration by cadmium administration to rats: the possibility of iron involvement in lipid peroxidation. Arch Biochem Biophys 346(2):171–179. https://doi.org/10.1006/abbi.1997.0197
Ciliberti A, Chaumot A, Recoura-Massaquant R, Chandesris A, François A, Coquery M, Ferréol M, Geffard O (2017) Caged Gammarus as biomonitors identifying thresholds of toxic metal bioavailability that affect gammarid densities at the French national scale. Wat Res 118:131–140. https://doi.org/10.1016/j.watres.2017.04.031
Coleman RA, Underwood AJ, Benedetti-Cecchi L, Aberg P, Arenas F, Arrontes J, Castro J, Hartnoll RG, Jenkins SR, Paula J, Della Santina P, Hawkins SJ (2006) A continental scale evaluation of the role of limpet grazing on rocky shores. Oecologia 147(3):556–564. https://doi.org/10.1007/s00442-005-0296-9
COMETE engineering-Bceom-IHE (2008) Caractérisation du golfe de Tunis – Synthèse Générale sur l’état du Golfe de Tunis. Ministère de l’Environnement et du Développement durable, Tunis, Tunisie
Conti ME, Mele G, Finoia MG (2017) Baseline trace metals in Patella caerulea in a central Tyrrhenian ecosystem (Pontine Islands archipelago and Lazio region coastal sites, Italy). Environ Sci Pollut Res Int 24(9):8852–8865. https://doi.org/10.1007/s11356-017-8572-x
Cravo A, Bebianno MJ (2005) Bioaccumulation of metals in the soft tissue of Patella aspera: application of metal/shell weight indices. Estuar Coast Shelf Sci 65(3):571–586. https://doi.org/10.1016/j.ecss.2005.06.026
Cravo A, Bebianno MJ, Foster P (2004) Partitioning of trace metals between soft tissues and shells of Patella aspera. Environ Int 30(1):87–98. https://doi.org/10.1016/S0160-4120(03)00154-5
Cubadda F, Conti ME, Campanella L (2001) Size-dependent concentrations of trace metals in four Mediterranean gastropods. Chemosphere 45(4):561–569. https://doi.org/10.1016/S0045-6535(01)00013-3
Cutler RG (1984) Urate and ascorbate: their possible roles as antioxidants in determining longevity of mammalian species. Arch Gerontol Geriatr 3(4):321–348. https://doi.org/10.1016/0167-4943(84)90033-5
da Silva AZ, Zanette J, Fernando Ferreira J, Guzenski J, Marques MRF, Bainy ACD (2005) Effects of salinity on biomarker responses in Crassostrea rhizophorae (Mollusca, Bivalvia) exposed to diesel oil. Ecotoxicol Environ Saf 62(3):376–382. https://doi.org/10.1016/j.ecoenv.2004.12.008
Davies MS, Cliffe EJ (2000) Adsorption of metals in seawater to limpet (Patella vulgata) pedal mucus. Bull Environ Contam Toxicol 64(2):228–234. https://doi.org/10.1007/s001289910034
de Almeida EA, Miyamoto S, Bainy ACD, de Medeiros MHG, Di Mascio P (2004) Protective effect of phospholipid hydroperoxide glutathione peroxidase (PHGPx) against lipid peroxidation in mussels Perna perna exposed to different metals. Mar Pollut Bull 49(5–6):386–392. https://doi.org/10.1016/j.marpolbul.2004.02.020
De Pirro M, Chelazzi G, Borghini F, Focardi S (2001) Variations in cardiac activity following acute exposure to copper in three co-occurring but differently zoned Mediterranean limpets. Mar Pollut Bull 42(12):1390–1396. https://doi.org/10.1016/S0025-326X(01)00169-2
Debnath A, Singh PK, Sharma YC (2021) Metallic contamination of global river sediments and latest developments for their remediation. J Environ Manag 298:113378. https://doi.org/10.1016/j.jenvman.2021.113378
Della Santina P, Chelazzi G (1991) Temporal organization of foraging in two Mediterranean limpets, Patella rustica L. and P. coerulea L. J Exp Mar Biol Ecol 153(1):75–85. https://doi.org/10.1016/S0022-0981(05)80007-3
Depledge M, Rainbow P (1990) Models of regulation and accumulation of trace metals in marine invertebrates. Comp Biochem Physiol Part C Comp Pharmacol 97. https://doi.org/10.1016/0742-8413(90)90163-4
Devez A (2004) Caractérisation des risques induits par les activités agricoles sur les écosystèmes aquatiques. PhD thesis, Ecole nationale du génie rural, des eaux et des forêts (Paris, Nancy).
Dorta DJ, Leite S, DeMarco KC, Prado IMR, Rodrigues T, Mingatto FE, Uyemura SA, Santos AC, Curti C (2003) A proposed sequence of events for cadmium-induced mitochondrial impairment. J Inorg Biochem 97(3):251–257. https://doi.org/10.1016/s0162-0134(03)00314-3
Douhri H, Sayah F (2009) The use of enzymatic biomarkers in two marine invertebrates Nereis diversicolor and Patella vulgata for the biomonitoring of Tangier’s bay (Morocco). Ecotoxicol Environ Saf 72(2):394–399. https://doi.org/10.1016/j.ecoenv.2008.07.016
Doyle J, Doyle J (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Drira Z (2018) Evaluation de la qualité des eaux de la zone côtière du golfe de Gabès à travers l’analyse physico chimique et zooplanctonique : Cas des côtes sud et nord de Sfax et de la zone de Ghannouch. Habilitation à diriger des recherches. PhD thesis, University of Sfax.
El-Geziry TM, Bryden IG (2010) The circulation pattern in the Mediterranean Sea: issues for modeller consideration. J Oper Oceanogr 3(2):39–46. https://doi.org/10.1080/1755876X.2010.11020116
Ennouri R, Chouba L, Magni P, Kraiem MM (2010) Spatial distribution of trace metals (Cd, Pb, Hg, Cu, Zn, Fe and Mn) and oligo-elements (Mg, Ca, Na and K) in surface sediments of the Gulf of Tunis (Northern Tunisia). Environ Monit Assess 163(1–4):229–239. https://doi.org/10.1007/s10661-009-0829-5
Ensibi C, Pringault O, Hannaoui W, Daly Yahya MN (2015) Effects of cadmium exposure on reproduction and survival of the planktonic copepod Centropages ponticus. 5(2):1-4. https://doi.org/10.4172/2155-9910.1000159
Espinosa F, Rivera-Ingraham GA (2016) Subcellular evidences of redox imbalance in well-established populations of an endangered limpet. Reasons for alarm? Mar Pollut Bull 109(1):72–80. https://doi.org/10.1016/j.marpolbul.2016.06.019
Ettajani H, Berthet B, Amiard JC, Chevolot (2001) Determination of cadmium partitioning in microalgae and oysters: contribution to the assessment of trophic transfer. Arch Environ Contam Toxicol 40:209-221. https://doi.org/10.1007/s002440010165
Farombi EO, Adelowo OA, Ajimoko YR (2007) Biomarkers of oxidative stress and heavy metal levels as indicators of environmental pollution in African cat fish (Clarias gariepinus) from Nigeria Ogun River. Int J Environ Res Public Health 4(2):158–165. https://doi.org/10.3390/ijerph2007040011
Fernández B, Campillo JA, Martínez-Gómez C, Benedicto J (2010) Antioxidant responses in gills of mussel (Mytilus galloprovincialis) as biomarkers of environmental stress along the Spanish Mediterranean coast. Aquat Toxicol 99(2):186–197. https://doi.org/10.1016/j.aquatox.2010.04.013
Fernández N, Alborés I, Aceña-Matarranz S (2016) Spatial variability of the reproductive cycle and physiological condition of Patella spp. (Mollusca Gastropoda) in the NW of the Iberian Peninsula: Implications for exploitation. Fish Res C(179):76–85. https://doi.org/10.1016/j.fishres.2016.02.010
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Meth Enzymol, 105:114-120. https://doi.org/10.1016/S0076-6879(84)05015-1
Fonollosa E, Peñalver A, Aguilar C, Borrull F (2017) Bioaccumulation of natural radionuclides in molluscs from the Ebro Delta area. Environ Sci Pollut Res 24(1):208–214. https://doi.org/10.1007/s11356-016-7783-x
Fretter V, Graham A (1972) British Prosobranch Molluscs. Their functional anatomy and ecology. With 317 fig., 755 pp. - London: Ray Society (Ray Society Publication No. 144, 1962). Int Rev Gesamten Hydrobiol Hydrogr 57(6):953–953. https://doi.org/10.1002/iroh.19720570626
Gabbott PA (1975) Storage cycles in marine bivalve molluscs: a hypothesis concerning the relationship between glycogen metabolism and gametogenesis. In: Barnes HB (Ed.) Ninth European Marine Biology Symposium, 191–211.
Galarza JA, Carreras-Carbonell J, Macpherson E, Pascual M, Roques S, Turner GF, Rico C (2009) The influence of oceanographic fronts and early-life-history traits on connectivity among littoral fish species. Proc Natl Acad Sci 106(5):1473–1478. https://doi.org/10.1073/pnas.0806804106
Ghannem N, Azri C, Serbaji Mohammed M, Yaich C (2010) Spatial distribution of heavy metals in the coastal zone of “Sfax-Kerkennah” plateau Tunisia. Environ Prog Sustain Energy 30(2):221–233. https://doi.org/10.1002/ep.10462
Ghannem N, Gargouri D, Sarbeji MM, Yaich C, Azri C (2014) Metal contamination of surface sediments of the Sfax-Chebba coastal line, Tunisia. Environ Earth Sci 72:3419–3427. https://doi.org/10.1007/s12665-014-3248-z
Griscom SB, Fisher NS, Luoma SN (2000) Geochemical influences on assimilation of sediment-bound metals in clams and mussels. Environ Sci Technol 34(1):91–99. https://doi.org/10.1021/es981309+
Guittonny-Philippe A, Masotti V, Höhener P, Boudenne J-L, Viglione J, Laffont-Schwob I (2014) Constructed wetlands to reduce metal pollution from industrial catchments in aquatic Mediterranean ecosystems: a review to overcome obstacles and suggest potential solutions. Environ Int 64:1–16. https://doi.org/10.1016/j.envint.2013.11.016
Harrabi M, Varela Della Giustina S, Aloulou F, Rodriguez-Mozaz S, Barceló D, Elleuch B (2018) Analysis of multiclass antibiotic residues in urban wastewater in Tunisia. Environ Nanotechnol Monit Manag 10:163–170. https://doi.org/10.1016/j.enmm.2018.05.006
Hasegawa M, Kishino H, Yano T (1985) Dating of the human-ape splitting by a molecular clock of mitochondrial DNA. J Mol Evol 22(2):160–174. https://doi.org/10.1007/BF02101694
Hawkins AJS, Bayne BL (1984) Seasonal variation in the balance between physiological mechanisms of feeding and digestion in Mytilus edulis (Bivalvia: Mollusca). Mar Biol 82(3):233–240. https://doi.org/10.1007/BF00392404
HELCOM (2019) Guidelines for sampling and determination of pH in seawater — expert interview: Jannica Haldin on biodiversity. https://helcom.fi/helcom-expert-interview-jannica-haldin-on-biodiversity/. Accessed 12 Jun 2021
INS (2013) Répertoire National des Entreprises. In: Inst. Natl. Stat. http://ins.tn/fr/methode-1. Accessed 12 May 2020
ISO 10523 (2008) Water quality — Determination of pH. International Standard Organisation, Geneva
ISO 24032 (2021) Soil quality — In situ caging of snails to assess bioaccumulation of contaminants. International Standard Organisation, Geneva.
Jebali J, Banni M, Almeida EAD, Boussetta H (2007) Oxidative DNA damage levels and catalase activity in the clam Ruditapes decussatus as pollution biomarkers of Tunisian marine environment. Environ Monit Assess 124(1):195–200. https://doi.org/10.1007/s10661-006-9217-6
Jenkins SR, Arenas F, Arrontes J, Bussell J, Castro J, Coleman RA, Hawkins SJ, Kay S, Martínez B, Oliveros J, F.Roberts M, Sousa S, Thompson RC, Hartnoll RG (2001) European-scale analysis of seasonal variability in limpet grazing activity and microalgal abundance. Mar Ecol Prog Ser 211:193–203. https://doi.org/10.3354/meps211193
Jurczuk M, Brzóska MM, Moniuszko-Jakoniuk J, Gałazyn-Sidorczuk M, Kulikowska-Karpińska E (2004) Antioxidant enzymes activity and lipid peroxidation in liver and kidney of rats exposed to cadmium and ethanol. Food Chem Toxicol Int J Publ Br Ind Biol Res Assoc 42(3):429–438. https://doi.org/10.1016/j.fct.2003.10.005
Kaste Ø, Skarbøvik E, Greipsland I, Gundersen CB, Austnes K, Skancke LB, Guerrero J-L, Sample JE (2018) The Norwegian river monitoring programme — water quality status and trends 2017. Norsk institutt for vannforskning
Khebbeb ME, Nadji S, Amrani A (2010) The effect of cadmium exposure on malonedialdehyde and reduced glutathione concentrations in several tissues of a bivalve mollusc (Ruditapes decussatus) fished from Mellah lagoon (North East of Algeria). Ann Biol Res 1(2):166–173
Kim SJ, Rodriguez-Lanetty M, Suh JH, Song JI (2003) Emergent effects of heavy metal pollution at a population level: Littorina brevicula a study case. Mar Pollut Bull 46(1):74–80. https://doi.org/10.1016/S0025-326X(02)00319-3
Kremling K (1976) Determination of trace metals. In: Methods of seawater analysis, 3rd edn. Strauss Offsetdruck, Morlenbac, pp 183–191.
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol Biol Evol 35(6):1547–1549. https://doi.org/10.1093/molbev/msy096
La Colla NS, Botté SE, Simonetti P, Negrin VL, Serra AV, Marcovecchio JE (2021) Water, sediments and fishes: first multicompartment assessment of metal pollution in a coastal environment from the SW Atlantic. Chemosphere 282:131131. https://doi.org/10.1016/j.chemosphere.2021.131131
Ladhar-Chaabouni R, Machreki-Ajmi M, Hamza-Chaffai A (2012) Use of metallothioneins as biomarkers for environmental quality assessment in the Gulf of Gabès (Tunisia). Environ Monit Assess 184(4):2177–2192. https://doi.org/10.1007/s10661-011-2108-5
Legeay A, Achard-Joris M, Baudrimont M, Massabuau J-C, Bourdineaud J-P (2005) Impact of cadmium contamination and oxygenation levels on biochemical responses in the Asiatic clam Corbicula fluminea. Aquat Toxicol 74(3):242–253. https://doi.org/10.1016/j.aquatox.2005.05.015
Lionetto MG, Caricato R, Giordano ME, Pascariello MF, Marinosci L, Schettino T (2003) Integrated use of biomarkers (acetylcholinesterase and antioxidant enzymes activities) in Mytilus galloprovincialis and Mullus barbatus in an Italian coastal marine area. Mar Pollut Bull 46(3):324–330. https://doi.org/10.1016/S0025-326X(02)00403-4
Livingstone DR (2001) Contaminant-stimulated reactive oxygen species production and oxidative damage in aquatic organisms. Mar Pollut Bull 42(8):656–666. https://doi.org/10.1016/s0025-326x(01)00060-1
Louzon M, Pauget B, Gimbert F, Morin-Crini N, de Vaufleury A (2020) Ex situ environmental risk assessment of polluted soils using threshold guide values for the land snail Cantareus aspersus. Sci Total Environ 721:137789. https://doi.org/10.1016/j.scitotenv.2020.137789
Louzon M, Pauget B, Gimbert F, Morin-Crini N, Wong JWY, Zaldibar B, Natal-da-Luz T, Neuwirthova N, Thiemann C, Sarrazin B, Irazola M, Amiot C, Rieffel D, Sousa JP, Chalot M, de Vaufleury A (2021) In situ and ex situ bioassays with Cantareus aspersus for environmental risk assessment of metal(loid) and PAH-contaminated soils. Integr Environ Assess Manag 4480. https://doi.org/10.1002/ieam.4480
Lowe DM, Moore MN, Bayne BL (1982) Aspects of gametogenesis in the marine mussel Mytilus edulis L. J Mar Biol Assoc U K 62(1):133–145. https://doi.org/10.1017/S0025315400020166
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lozano-Bilbao E, Herranz I, González-Lorenzo G, Lozano G, Hardisson A, Rubio C, González-Weller D, Paz S, Gutiérrez ÁJ (2021) Limpets as bioindicators of element pollution in the coasts of Tenerife (Canary Islands). Environ Sci Pollut Res 28(31):42999–43006. https://doi.org/10.1007/s11356-021-15212-7
Lushchak VI (2011) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101(1):13–30. https://doi.org/10.1016/j.aquatox.2010.10.006
Matson CW, Lambert LM, McDonald TJ, Autenrieth RL, Donnelly KC, Islamzadeh A, Politov DI, Bickham JW (2006) Evolutionary toxicology: population-level effects of chronic contaminant exposure on the Marsh Frogs (Rana ridibunda) of Azerbaijan. Environ Health Perspect 114(4):547–552. https://doi.org/10.1289/ehp.8404
Mezghani-Chaari S, Machreki-Ajmi M, Tremolet G, Kellner K, Geffard A, Minier C, Hamza-Chaffai A (2015) The endocrine-disrupting effect and other physiological responses of municipal effluent on the clam Ruditapes decussatus. Environ Sci Pollut Res 22(24):19716–19728. https://doi.org/10.1007/s11356-015-5199-7
Milano M, Ruelland D, Fernandez S, Dezetter A, Fabre J, Servat E (2012) Facing climatic and anthropogenic changes in the Mediterranean basin: what will be the medium-term impact on water stress? Comptes Rendus Geosci 344(9):432–440. https://doi.org/10.1016/j.crte.2012.07.006
Mleiki A, El Menif NJ, Marigomez I (2020) Integrative assessment of the biological responses elicited by metal pollution in the green garden snail, Cantareus apertus: laboratory and field studies. Ecol Indic 117:106589. https://doi.org/10.1016/j.ecolind.2020.106589
Moreira SM, Guilhermino L (2005) The use of Mytilus Galloprovincialis acetylcholinesterase and glutathione S-Transferases activities as biomarkers of environmental contamination along the northwest Portuguese Coast. Environ Monit Assess 105(1):309–325. https://doi.org/10.1007/s10661-005-3854-z
Mylonas C, Kouretas D (1999) Lipid peroxidation and tissue damage. Vivo Athens Greece 13(3):295–309
Niehaus WG, Samuelsson B (1968) Formation of malonaldehyde from phospholipid arachidonate during microsomal lipid peroxidation. Eur J Biochem 6(1):126–130. https://doi.org/10.1111/j.1432-1033.1968.tb00428.x
Nriagu JO (1996) A history of global metal pollution. Science 272(5259):223. https://doi.org/10.1126/science.272.5259.223
Oros DR, Hoover D, Rodigari F, Crane D, Sericano J (2005) Levels and distribution of polybrominated diphenyl ethers in water, surface sediments, and bivalves from the San Francisco estuary. Environ Sci Technol 39(1):33–41. https://doi.org/10.1021/es048905q
Ozaki S (2019) Biodiversity and functioning of terrestrial food webs: application to transfers of trace metals. PhD thesis, University of Burgundy Franche-Comté.
Palladino G, Rampelli S, Scicchitano D, Musella M, Quero GM, Prada F, Mancuso A, Seyfarth AM, Turroni S, Candela M, Biagi E (2021) Impact of marine aquaculture on the microbiome associated with nearby holobionts: the case of Patella caerulea living in proximity of Sea Bream aquaculture cages. Microorganisms 9(2):455. https://doi.org/10.3390/microorganisms9020455
Pampanin DM, Camus L, Gomiero A, Marangon I, Volpato E, Nasci C (2005) Susceptibility to oxidative stress of mussels (Mytilus galloprovincialis) in the Venice Lagoon (Italy). Mar Pollut Bull 50(12):1548–1557. https://doi.org/10.1016/j.marpolbul.2005.06.023
Pavalaki MD, Aurajo MJ, Cardoso DN, Silva ARR, Cruz A, Mendo S, Soares AMVM, Calado R, Loureiro S (2016) Ecotoxicity and genotoxicity of cadmium in different marine trophic levels. Environ Pollut 215:203–212. https://doi.org/10.1016/j.envpol.2016.05.010
Pereira J, Chaves R, Bastos E, Leitão A, Guedes-Pinto H (2011) An efficient method for genomic DNA extraction from different molluscs species. Int J Mol Sci 12:8086–8095. https://doi.org/10.3390/ijms12118086
Poppe GT, Goto Y (1991) European seashells, Volume I: Polyplacophora, Caudofoveata, Solenogastra, Gastropoda. Christa Hemmen, Wiesbaden.oks -. Verlag Christa Hemmen.
Primost MA, Gil MN, Bigatti G (2017) High bioaccumulation of cadmium and other metals in Patagonian edible gastropods. Mar Biol Res 13(7):774–781. https://doi.org/10.1080/17451000.2017.1296163
Rabaoui L, Balti R, Zrelli R, Tlig-Zouari S (2013) Assessment of heavy metals pollution in the gulf of Gabes (Tunisia) using four mollusk species. Mediterr Mar Sci 15(1):45. https://doi.org/10.12681/mms.504
Readman JW, Kwong LLW, Grondin D, Bartocci J, Villeneuve JP, Mee LD (1993) Coastal water contamination from a triazine herbicide used in antifouling paints. Environ Sci Technol 27(9):1940–1942. https://doi.org/10.1021/es00046a027
Regoli F (1998) Trace metals and antioxidant enzymes in gills and digestive gland of the Mediterranean mussel Mytilus galloprovincialis. Arch Environ Contam Toxicol 34(1):48–63. https://doi.org/10.1007/s002449900285
Regoli F, Principato G (1995) Glutathione, glutathione-dependent and antioxidant enzymes in mussel, Mytilus galloprovincialis, exposed to metals under field and laboratory conditions: implications for the use of biochemical biomarkers. Aquat Toxicol 31(2):143–164. https://doi.org/10.1016/0166-445X(94)00064-W
Regoli F, Pellegrini D, Winston GW, Gorbi S, Giuliani S, Virno-Lamberti C, Bompadre S (2002a) Application of biomarkers for assessing the biological impact of dredged materials in the Mediterranean: the relationship between antioxidant responses and susceptibility to oxidative stress in the red mullet (Mullus barbatus). Mar Pollut Bull 44(9):912–922. https://doi.org/10.1016/s0025-326x(02)00120-0
Regoli F, Gorbi S, Frenzilli G, Nigro M, Corsi I, Focardi S, Winston GW (2002b) Oxidative stress in ecotoxicology: from the analysis of individual antioxidants to a more integrated approach. Mar Environ Res 54(3–5):419–423. https://doi.org/10.1016/s0141-1136(02)00146-0
Reguera P, Couceiro L, Fernández N (2018) A review of the empirical literature on the use of limpets Patella spp. (Mollusca: Gastropoda) as bioindicators of environmental quality. Ecotoxicol Environ Saf 148:593–600. https://doi.org/10.1016/j.ecoenv.2017.11.004
Rivera Ingraham GA, Malanga GF, Puntarulo SÁ, Pérez AF, Ruiz Tabares A, Maestre M, González Aranda R, Espinosa F, García Gómez JC (2013) Antioxidant defenses and trace metal bioaccumulation capacity of Cymbula nigra (Gasteropoda: Patellidae). https://doi.org/10.1007/s11270-013-1458-8
Ross K, Cooper N, Bidwell JR, Elder J (2002) Genetic diversity and metal tolerance of two marine species: a comparison between populations from contaminated and reference sites. Mar Pollut Bull 44(7):671–679. https://doi.org/10.1016/S0025-326X(01)00333-2
Rumisha C, Leermakers M, Elskens M, Mdegela RH, Gwakisa P, Kochzius M (2017) Genetic diversity of the giant tiger prawn Penaeus monodon in relation to trace metal pollution at the Tanzanian coast. Mar Pollut Bull 114(2):759–767. https://doi.org/10.1016/j.marpolbul.2016.10.057
Shefer E, Silverman J, Herut B (2015) Trace metal bioaccumulation in Israeli Mediterranean coastal marine mollusks. Quat Int 390:44–55. https://doi.org/10.1016/j.quaint.2015.10.030
Silvestre F (2005) Réponses à un stress environnemental induit par le cadmium chez un crustacé euryhalin, Eriocheir sinensis. Approche intégrative incluant une analyse du protéome. PhD Thesis, Namur University.
Solé C, Porte C, Barcelo D, Albaiges J (2000) Bivalve residue analysis for the assessment of coastal pollution in the Ebro Delta (NW Mediterranean). Marine Pollut Bull 40(9):746–753. https://doi.org/10.1016/S0025-326X(00)00011-4
Soltani N, Amira A, Sifi K, Beldi H (2012) Environmental monitoring of the Annaba Gulf (Algeria): measurements of biomarkers in Donax trunculus and metallic pollution. Bull Société Zool Fr 1–4(137):51–60
Storelli MM, Marcotrigiano GO (2005) Bioindicator organisms: heavy metal pollution evaluation in the Ionian Sea (Mediterranean Sea—Italy). Environ Monit Assess 102(1):159–166. https://doi.org/10.1007/s10661-005-6018-2
Touyz RM (1979) (2004) Reactive oxygen species, vascular oxidative stress, and redox signaling in hypertension: what is the clinical significance? Hypertens Dallas Tex 44(3):248–252. https://doi.org/10.1161/01.HYP.0000138070.47616.9d
Trevizani TH, Figueira RCL, Ribeiro AP, Theophilo CYS, Majer AP, Petti MAV, Corbisier TN, Montone RC (2016) Bioaccumulation of heavy metals in marine organisms and sediments from Admiralty Bay, King George Island. Antarctica Mar Pollut Bull 106(1):366–371. https://doi.org/10.1016/j.marpolbul.2016.02.056
UNEP/IAE/FAO (1984) Determination of total cadmium, zinc, lead and copper in selected marine organisms by flameless atomic absorption spectrophotometry. Reference methods for marine pollution studies (1):11–49.
UNEP/MAP/MED (2020) Integrated Meetings of the Ecosystem Approach Correspondence Groups on IMAP Implementation (CORMONs) - Monitoring Guidelines/Protocols for Determination of Hydrographic Physical Parameters
UNESCO (1994) Protocols for the Joint Global Ocean Flux Study (JGOFS) core measurements
Viarengo A, Canesi L, Pertica M, Livingstone DR (1991) Seasonal variations in the antioxidant defense systems and lipid peroxidation of the digestive gland of mussels. Comp Biochem Physiol Part C Comp Pharmacol 100(1):187–190. https://doi.org/10.1016/0742-8413(91)90151-I
Vidal-Liñán L, Bellas J, Etxebarria N, Nieto O, Beiras R (2014) Glutathione S-transferase, glutathione peroxidase and acetylcholinesterase activities in mussels transplanted to harbour areas. Sci Total Environ 470–471:107–116. https://doi.org/10.1016/j.scitotenv.2013.09.073
Viñas L, Pérez-Fernández B, Soriano JA, López M, Bargiela J, Alves I (2018) Limpet (Patella sp) as a biomonitor for organic pollutants. A proxy for mussel? Mar Pollut Bull 133:271–280. https://doi.org/10.1016/j.marpolbul.2018.05.046
Vlahogianni T, Dassenakis M, Scoullos MJ, Valavanidis A (2007) Integrated use of biomarkers (superoxide dismutase, catalase and lipid peroxidation) in mussels Mytilus galloprovincialis for assessing heavy metals’ pollution in coastal areas from the Saronikos Gulf of Greece. Mar Pollut Bull 54(9):1361–1371. https://doi.org/10.1016/j.marpolbul.2007.05.018
Wang WX, Fisher NS, Luoma SN (1996) Kinetic determinations of trace element bioaccumulation in the mussel Mytilus edulis. Mar Ecol Prog Ser 140:91–113. https://doi.org/10.3354/meps140091
Wang S-L, Xu X-R, Sun Y-X, Liu J-L, Li H-B (2013) Heavy metal pollution in coastal areas of South China: a review. Mar Pollut Bull 76(1–2):7–15. https://doi.org/10.1016/j.marpolbul.2013.08.025
Watjen W, Beyersmann D (2004) Cadmium-induced apoptosis in C6 glioma cells: influence of oxidative stress. Biometals Int J Role Met Ions Biol Biochem Med 17(1):65–78. https://doi.org/10.1023/a:1024405119018
Wei B, Yang L (2010) A review of heavy metal contaminations in urban soils, urban road dusts and agricultural soils from China. Microchem J 94(2):99–107. https://doi.org/10.1016/j.microc.2009.09.014
Wesselmann M, González-Wangüemert M, Serrão EA, Engelen AH, Renault L, García-March JR, Duarte CM, Hendriks IE (2018) Genetic and oceanographic tools reveal high population connectivity and diversity in the endangered pen shell Pinna nobilis. Sci Rep 8(1):4770. https://doi.org/10.1038/s41598-018-23004-2
WWF (2020) Le littoral tunisien. https://wwf.panda.org/fr/wwf_action_zones/tunisie/littoral_tunisien/. Accessed 14 Jul 2021
Yawetz A, Manelis R, Fishelson L (1992) The effects of aroclor 1254 and petrochemical pollutants on cytochrome P450 from the digestive gland microsomes of four species of Mediterranean molluscs. Comp Biochem Physiol Part C Comp Pharmacol 103(3):607–614. https://doi.org/10.1016/0742-8413(92)90188-D
Yüzereroğlu TA, Gök G, Çoğun HY, Firat Ö, Aslanyavrusu S, Maruldalı O, Kargin F (2010) Heavy metals in Patella caerulea (Mollusca, Gastropoda) in polluted and non-polluted areas from the Iskenderun Gulf (Mediterranean Turkey). Environ Monit Assess 167(1):257–264. https://doi.org/10.1007/s10661-009-1047-x
Zelko IN, Mariani TJ, Folz RJ (2002) Superoxide dismutase multigene family: a comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radic Biol Med 33(3):337–349. https://doi.org/10.1016/s0891-5849(02)00905-x
Acknowledgements
The authors are grateful to Pr. Ayadi for allowing us to do the measurements of trace metals in his laboratory at the University of Sfax in Tunisia. We kindly thank Christophe Buyse, English teacher at Le Mans University, for the time devoted to proofreading our work and Dr. Louzon for his valuable contribution in the revision of the manuscript.
Funding
Part of the PhD is financed by the Eiffel Scholarship Program of Excellence to insure MZ’s (PhD student) stay in Le Mans University, France. Otherwise, no funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
MZ: Conceptualization, Formal Analysis, Investigation, Methodology, Visualization, Validation, Writing – Original draft, Writing – Review and Editing; KA: Methodology; IM: Review; HA: Resources, Supervision; VL: Conceptualization, Methodology, Project Administration, Supervision, Writing—Original Draft, Writing – Review and Editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Invertebrate animal used in this study doesn’t require specific approval from the ethical committee. All authors consented to participate in this study.
Consent for publication
All authors consented to publish this article under its current form.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Bruno Nunes.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible Editor: Bruno Nunes
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zaidi, M., Athmouni, K., Metais, I. et al. The Mediterranean limpet Patella caerulea (Gastropoda, Mollusca) to assess marine ecotoxicological risk: a case study of Tunisian coasts contaminated by metals. Environ Sci Pollut Res 29, 28339–28358 (2022). https://doi.org/10.1007/s11356-021-18490-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-18490-3