Abstract
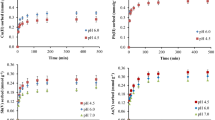
Composite mineral-biochars of a homogeneous biomass (cellulose) and heterogeneous biomass (oak leaves) were fabricated with either 5 wt% or 10 wt% minerals (montmorillonite (MMT), kaolinite, and sand) and then pyrolyzed at 600 °C for 60 min. Characterizations including proximate analysis, ultimate analysis, surface area and porosity, morphology, and surface chemistry confirmed that minerals were present on the surface of biochar, and MMT/kaolinite-biochar composites showed a strengthening in the chars’ aromatic structures, as well as increases in oxygen-containing surface functional groups. Methylene blue adsorption isotherms indicated that the MMT/kaolinite-biochars had higher adsorption capacities than pure biomass or biomass-sand biochars (110 mgMB/gchar and 24 mgMB/gchar for MMT-cellulose char and cellulose char, respectively). A multilinear model relating adsorption capacity and adsorbent properties was developed to measure the relative contribution of biochar properties to adsorption behavior. The model indicates that pore volume and hydrogen bonding were the dominant properties in controlling the adsorption of methylene blue onto the biochars. Findings from this work indicate that composite biochars prepared from biomass and inexpensive clay minerals are a promising adsorbent for remediating organic contaminants from water.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Abou-El-Sherbini KS, Hassanien MM (2010) Study of organically-modified montmorillonite clay for the removal of copper(II). J Hazard Mater 184:654–661. https://doi.org/10.1016/j.jhazmat.2010.08.088
Ahmad M, Lee SS, Dou X, Mohan D, Sung JK, Yang JE, Ok YS (2012) Effects of pyrolysis temperature on soybean stover- and peanut shell-derived biochar properties and TCE adsorption in water. Bioresour Technol 118:536–544. https://doi.org/10.1016/j.biortech.2012.05.042
Alqadami AA, Naushad M, Alothman ZA, Ahamad T (2018) Adsorptive performance of MOF nanocomposite for methylene blue and malachite green dyes: kinetics, isotherm and mechanism. J Environ Manag 223:29–36. https://doi.org/10.1016/j.jenvman.2018.05.090
Basak BB, Saha A, Sarkar B, Kumar BP, Gajbhiye NA, Banerjee A (2020) Repurposing distillation waste biomass and low-value mineral resources through biochar-mineral-complex for sustainable production of high-value medicinal plants and soil quality improvement. Sci Total Environ 1:1–10
Basta AH, Fierro V, El-Saied H, Celzard A (2009) 2-steps KOH activation of rice straw: an efficient method for preparing high-performance activated carbons. Bioresour Technol 100:3941–3947. https://doi.org/10.1016/j.biortech.2009.02.028
Basta AH, Fierro V, Saied H, Celzard A (2011) Effect of deashing rice straws on their derived activated carbons produced by phosphoric acid activation. Biomass Bioenergy 35:1954–1959. https://doi.org/10.1016/j.biombioe.2011.01.043
Brindley GW, Thompson TD (1970) Methylene blue absorption by montmorillonites. Determinations of Surface areas and exchange capacities with different initial cation saturations (clay-organic studies XIX). Isr J Chem 8:409–415. https://doi.org/10.1002/ijch.197000047
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319 . citeulike-article-id:4074706\rdoi: https://doi.org/10.1021/ja01269a023
Carrier M, Hardie AG, Uras Ü, Görgens J, Knoetze J (2012) Production of char from vacuum pyrolysis of South-African sugar cane bagasse and its characterization as activated carbon and biochar. J Anal Appl Pyrolysis 96:24–32. https://doi.org/10.1016/j.jaap.2012.02.016
Chcn G, Pan J, Han B, Yan H (1999) Adsorption of methylene blue on montmorillonite. J Dispers Sci Technol 20:1179–1187. https://doi.org/10.1080/01932699908943843
Chen L, Chen XL, Zhou CH, Yang HM, Ji SF, Tong DS, Zhong ZK, Yu WH, Chu MQ (2017) Environmental-friendly montmorillonite-biochar composites: facile production and tunable adsorption-release of ammonium and phosphate. J Clean Prod 156:648–659. https://doi.org/10.1016/j.jclepro.2017.04.050
Dehkhoda AM, Ellis N, Gyenge E (2016a) Effect of activated biochar porous structure on the capacitive deionization of NaCl and ZnCl2 solutions. Microporous Mesoporous Mater 224:217–228. https://doi.org/10.1016/j.micromeso.2015.11.041
Dehkhoda AM, Gyenge E, Ellis N (2016b) A novel method to tailor the porous structure of KOH-activated biochar and its application in capacitive deionization and energy storage. Biomass Bioenergy 87:107–121. https://doi.org/10.1016/j.biombioe.2016.02.023
Dou G, Goldfarb J (2017) In situ upgrading of pyrolysis biofuels by bentonite clay with simultaneous production of heterogeneous adsorbents for water treatment. Fuel 195:273–283. https://doi.org/10.1016/j.fuel.2017.01.052
Gao L, Goldfarb JL (2019) Solid waste to biofuels and heterogeneous sorbents via pyrolysis of wheat straw in the presence of fly ash as an in situ catalyst. J Anal Appl Pyrolysis 137:96–105. https://doi.org/10.1016/j.jaap.2018.11.014
Goldfarb JL, Buessing L, Gunn E, Lever M, Billias A, Casoliba EN, Schievano A, Adani F, Buessing L, Botto A, Casoliba EN, Rossoni M, Goldfarb JL (2017) Novel integrated biorefinery for olive mill waste management: utilization of secondary waste for water treatment. ACS Sustain Chem Eng 5:876–884. https://doi.org/10.1021/acssuschemeng.6b02202
Gürses A, Doǧar Ç, Yalçin M, Açikyildiz M, Bayrak R, Karaca S (2006) The adsorption kinetics of the cationic dye, methylene blue, onto clay. J Hazard Mater 131:217–228. https://doi.org/10.1016/j.jhazmat.2005.09.036
Hameed BH, Tan IAW, Ahmad AL (2008) Adsorption isotherm, kinetic modeling and mechanism of 2,4,6-trichlorophenol on coconut husk-based activated carbon. Chem Eng J 144:235–244. https://doi.org/10.1016/j.cej.2008.01.028
He K, Zeng G, Chen A, Huang Z, Peng M, Huang T, Chen G (2019) Graphene hybridized polydopamine-kaolin composite as effective adsorbent for methylene blue removal. Compos Part B Eng 161:141–149. https://doi.org/10.1016/j.compositesb.2018.10.063
Hoslett J, Ghazal H, Mohamad N, Jouhara H (2020) Removal of methylene blue from aqueous solutions by biochar prepared from the pyrolysis of mixed municipal discarded material. Sci Total Environ 714:136832. https://doi.org/10.1016/j.scitotenv.2020.136832
Istan S, Ceylan S, Topcu Y, Hintz C, Tefft J, Chellappa T, Guo J, Goldfarb JL (2016) Product quality optimization in an integrated biorefinery: conversion of pistachio nutshell biomass to biofuels and activated biochars via pyrolysis. Energy Convers Manag 127:576–588. https://doi.org/10.1016/j.enconman.2016.09.031
Jang HM, Yoo S, Choi YK, Park S, Kan E (2018) Adsorption isotherm, kinetic modeling and mechanism of tetracycline on Pinus taeda-derived activated biochar. Bioresour Technol 259:24–31. https://doi.org/10.1016/j.biortech.2018.03.013
Jiang M q, Jin X y, Lu XQ, Chen Z l (2010) Adsorption of Pb(II), Cd(II), Ni(II) and Cu(II) onto natural kaolinite clay. Desalination 252:33–39. https://doi.org/10.1016/j.desal.2009.11.005
Kai LY, Xin JL, Hwai CO, Wei-Hsin C, Jo-Shu C, Chih-Sheng L, Pau LS, TCL (2020) Adsorptive removal of cationic methylene blue and anionic Congo red dyes using wet- torrefied microalgal biochar: equilibrium, kinetic and mechanism modeling. Environ Pollut:1–12. https://doi.org/10.1016/j.envpol.2020.115986
Khawmee K, Suddhiprakarn A, Kheoruenromne I, Singh B (2013) Surface charge properties of kaolinite from Thai soils. Geoderma 192:120–131. https://doi.org/10.1016/j.geoderma.2012.07.010
Kimetu JM, Hill JM, Husein M, Bergerson J, Layzell DB (2014) Using activated biochar for greenhouse gas mitigation and industrial water treatment. Mitig Adapt Strateg Glob Chang 21:761–777. https://doi.org/10.1007/s11027-014-9625-9
Klika Z, Pustková P, Dudová M, Čapková P, Kliková C, Grygar TM (2011) The adsorption of methylene blue on montmorillonite from acid solutions. Clay Miner 46:461–471. https://doi.org/10.1180/claymin.2011.046.3.461
Lee LY, Gan S, Yin Tan MS, Lim SS, Lee XJ, Lam YF (2016) Effective removal of Acid Blue 113 dye using overripe Cucumis sativus peel as an eco-friendly biosorbent from agricultural residue. J Clean Prod 113:194–203. https://doi.org/10.1016/j.jclepro.2015.11.016
Li K, Zhang Q (2019) Influence of micro-meso porous structure on biomass carbon’s adsorption of methylene blue. Environ Technol 37:99–104
Li Y, Wang Z, Xie X, Zhu J, Li R, Qin T (2017) Removal of Norfloxacin from aqueous solution by clay-biochar composite prepared from potato stem and natural attapulgite. Colloids Surf A Physicochem Eng Asp 514:126–136. https://doi.org/10.1016/j.colsurfa.2016.11.064
Liao P, Yuan S, Zhang W, Tong M, Wang K (2012) Mechanistic aspects of nitrogen-heterocyclic compound adsorption on bamboo charcoal. J Colloid Interface Sci 382:74–81. https://doi.org/10.1016/j.jcis.2012.05.052
Liao Y, Li L, Fan S (2019) Removal behavior and mechanism of methylene blue in aqueous solution by rice straw and rice straw-Fe3O4 composite. Acta Sci Circumst (39):359–370. https://doi.org/10.13671/j.hjkxxb.2018.0318
Liu B, Fu M, Xiang L, Feng N, Zhao H, Li Y, Cai Q, Li H, Mo C, Wong M (2020) Adsorption of microcystin contaminants by biochars derived from contrasting pyrolytic conditions: characteristics, affecting factors, and mechanisms. Sci Total Environ 1–10
Luo W, Wan J, Fan Z, Hu Q, Zhou N, Xia M, Song M, Qi Z, Zhou Z (2021) In-situ catalytic pyrolysis of waste tires over clays for high quality pyrolysis products. Int J Hydrog Energy 46:6937–6944. https://doi.org/10.1016/j.ijhydene.2020.11.170
Marrakchi F, Khanday WA, Asif M, Hameed BH (2016) Cross-linked chitosan/sepiolite composite for the adsorption of methylene blue and reactive orange 16. Int J Biol Macromol 93:1231–1239. https://doi.org/10.1016/j.ijbiomac.2016.09.069
Miao Z, Gao M, Wan K, Pei Z, He Q, Ji P, Bai L (2019) Modification of Zhaotong lignite by steam explosion treatment: pore structure and oxygen-containing functional groups. Energy Fuel 33:4033–4040
Mouni L, Belkhiri L, Bollinger JC, Bouzaza A, Assadi A, Tirri A, Dahmoune F, Madani K, Remini H (2018) Removal of Methylene Blue from aqueous solutions by adsorption on kaolin: kinetic and equilibrium studies. Appl Clay Sci 153:38–45. https://doi.org/10.1016/j.clay.2017.11.034
Murray HH (1991) Overview - clay mineral applications. Appl Clay Sci 5:379–395. https://doi.org/10.1016/0169-1317(91)90014-Z
Pan B, Xing B (2008) Adsorption mechanisms of organic chemicals on carbon nanotubes. Environ Sci Technol 42:9005–9013. https://doi.org/10.1021/es801777n
Patil CS, Kadam AN, Gunjal DB, Naik VM, Lee SW, Kolekar GB, Gore AH (2020) Sugarcane molasses derived carbon sheet@sea sand composite for direct removal of methylene blue from textile wastewater: industrial wastewater remediation through sustainable, greener, and scalable methodology. Sep Purif Technol 247:1–12. https://doi.org/10.1016/j.seppur.2020.116997
Qu J, Wang Y, Tian X, Jiang Z, Deng F, Tao Y, Jiang Q, Wang L, Zhang Y (2021) KOH-activation porous biochar with high specific surface area for adsorptive removal of chromium(VI) and naphathalene from water: affecting factors, mechanisms and reusability exploration. J Hazard Mater 401:1–10
Segad M, Jönsson B, Åkesson T, Cabane B (2010) Ca/Na montmorillonite: structure, forces and swelling properties. Langmuir 26:5782–5790. https://doi.org/10.1021/la9036293
Sternik D, Galaburda MV, Bogatyrov VM, Oranska OI, Charmas B, Gun’ko VM (2020) Novel porous carbon/clay nanocomposites derived from kaolinite/resorcinol-formaldehyde polymer blends: synthesis, structure and sorption properties. Appl Surf Sci 525:1–11. https://doi.org/10.1016/j.apsusc.2020.146361
Suliman W, Harsh JB, Abu-lail NI, Fortuna A, Dallmeyer I, Garcia-perez M (2016) Influence of feedstock source and pyrolysis temperature on biochar bulk and surface properties. Biomass Bioenergy 84:37–48. https://doi.org/10.1016/j.biombioe.2015.11.010
Sun P, Hui C, Khan RA, Du J, Zhang Q, Zhao YH (2015) Efficient removal of crystal violet using Fe3O4-coated biochar: the role of the Fe3O4 nanoparticles and modeling study their adsorption behavior. Sci Rep 5:1–12. https://doi.org/10.1038/srep12638
Tan X, Liu Y, Zeng G, Wang X, Hu X, Gu Y, Yang Z (2015) Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 125:70–85. https://doi.org/10.1016/j.chemosphere.2014.12.058
Tan X, Liu S, Liu Y, Gu Y, Zeng G, Hu X, Wang X, Liu S, Jiang L (2017) Biochar as potential sustainable precursors for activated carbon production: multiple applications in environmental protection and energy storage. Bioresour Technol 227:359–372. https://doi.org/10.1016/j.biortech.2016.12.083
Tang J, Zong L, Mu B, Zhu Y, Wang A (2018) Preparation and cyclic utilization assessment of palygorskite/carbon composites for sustainable efficient removal of methyl violet. Appl Clay Sci 161:317–325. https://doi.org/10.1016/j.clay.2018.04.039
Tang J, Mu B, Zong L, Wang A (2019) From waste hot-pot oil as carbon precursor to development of recyclable attapulgite/carbon composites for wastewater treatment. J Environ Sci (China) 75:346–358. https://doi.org/10.1016/j.jes.2018.05.014
Tay T, Ucar S, Karagöz S (2009) Preparation and characterization of activated carbon from waste biomass. J Hazard Mater 165:481–485. https://doi.org/10.1016/j.jhazmat.2008.10.011
Uchimiya M, Wartelle LH, Klasson KT, Fortier CA, Lima IM (2011) Influence of pyrolysis temperature on biochar property and function as a heavy metal sorbent in soil. J Agric Food Chem 59:2501–2510. https://doi.org/10.1021/jf104206c
van Zwieten L, Kimber S, Morris S, Chan KY, Downie A, Rust J, Joseph S, Cowie A (2010) Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 327:235–246. https://doi.org/10.1007/s11104-009-0050-x
Vijayalakshmi P, Bala VSS, Thiruvengadaravi KV, Panneerselvam P, Palanichamy M, Sivanesan S (2010) Removal of Acid Violet 17 from aqueous solutions by adsorption onto activated carbon prepared from pistachio nut shell. Sep Sci Technol 46:155–163. https://doi.org/10.1080/01496395.2010.484006
Visa M, Bogatu C, Duta A (2010) Simultaneous adsorption of dyes and heavy metals from multicomponent solutions using fly ash. Appl Surf Sci 256:5486–5491. https://doi.org/10.1016/j.apsusc.2009.12.145
Wang X, Shu L, Wang Y, Xu B, Bai Y, Tao S, Xing B (2011) Sorption of peat humic acids to multi-walled carbon nanotubes. Environ Sci Technol 45:9276–9283. https://doi.org/10.1021/es202258q
Wang X, Zhang Y, Luo W, Elzatahry AA, Cheng X, Alghamdi A, Abdullah AM, Deng Y, Zhao D (2016) Synthesis of ordered mesoporous silica with tunable morphologies and pore sizes via a nonpolar solvent-assisted Stöber method. Chem Mater 28:2356–2362. https://doi.org/10.1021/acs.chemmater.6b00499
Wu H, Lai C, Zeng G, Liang J, Chen J, Xu J, Dai J, Li X, Liu J, Chen M, Lu L, Hu L, Wan J (2017) The interactions of composting and biochar and their implications for soil amendment and pollution remediation: a review. Crit Rev Biotechnol 37:754–764. https://doi.org/10.1080/07388551.2016.1232696
Yang B, Cai Z, Zhao W (2009) Catalysis of different clay minerals to kerabitumen in hydrocarbon genesis. Ed Nat Sci 11:68–71. https://doi.org/10.19406/j.cnki.cqkjxyxbzkb.2009.01.021
Yao Y, Gao B, Fang J, Zhang M, Chen H, Zhou Y, Creamer AE, Sun Y, Yang L (2014) Characterization and environmental applications of clay-biochar composites. Chem Eng J 242:136–143. https://doi.org/10.1016/j.cej.2013.12.062
Yao S, Zhang M, Li L, Liao Y, Zhou N, Fan S, Tang J (2018) Preparation of tea waste-nano Fe3O4 composite and its removal mechanism of methylene blue from aqueous solution. Environ Chem 37:96–107
Yılmaz Ş, Zengin A, Ecer Ü, Şahan T (2019) Conversion from a natural mineral to a novel effective adsorbent: utilization of pumice grafted with polymer brush for methylene blue decolorization from aqueous environments. Colloids Surf A Physicochem Eng Asp 583:1–13. https://doi.org/10.1016/j.colsurfa.2019.123961
Zhang M, Gao B (2013) Removal of arsenic , methylene blue , and phosphate by biochar / AlOOH nanocomposite. Chem Eng J 226:286–292. https://doi.org/10.1016/j.cej.2013.04.077
Zhang M, Gao B, Varnoosfaderani S, Hebard A, Yao Y, Inyang M (2013) Preparation and characterization of a novel magnetic biochar for arsenic removal. Bioresour Technol 130:457–462. https://doi.org/10.1016/j.biortech.2012.11.132
Zhao Z, Zhou W (2019) Insight into interaction between biochar and soil minerals in changing biochar properties and adsorption capacities for sulfamethoxazole. Environ Pollut 245:208–217
Zhou L, Chen H, Jiang X, Lu F, Zhou Y, Yin W, Ji X (2009) Modification of montmorillonite surfaces using a novel class of cationic gemini surfactants. J Colloid Interface Sci 332:16.21
Zhu L, Yang H, Zhao Y, Kang K, Liu Y, He P, Wu Z, Wei Z (2019) Biochar combined with montmorillonite amendments increase bioavailable organic nitrogen and reduce nitrogen loss during composting. Bioresour Technol 294:1–9. https://doi.org/10.1016/j.biortech.2019.122224
Funding
L. Gao acknowledges the support of the Fundamental Research Funds for the Central Universities (Grant no. 2020QN37) and the support of the China Scholarship Council (CSC) under Grant CSC NO. 201606420017.
Author information
Authors and Affiliations
Contributions
LG: methodology, material preparation, origin draft preparation; JG: review and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Zhihong Xu
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 531 kb)
Rights and permissions
About this article
Cite this article
Gao, L., Goldfarb, J.L. Characterization and adsorption applications of composite biochars of clay minerals and biomass. Environ Sci Pollut Res 28, 44277–44287 (2021). https://doi.org/10.1007/s11356-021-13858-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-13858-x