Abstract
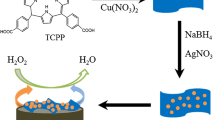
In this study, copper oxide nanoparticles (CuONPs) were prepared by a simple chemical method and then characterized by scanning electron microscope (SEM). A novel electrochemical sensor for hydrogen peroxide (H2O2) analysis was prepared by immobilizing copper oxide nanoparticles and polyalizarin yellow R (PYAR) on bare glassy carbon electrode (PAYR/CuONPs/GCE). The electrocatalytical behavior of the proposed electrochemical sensor was also studied by cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS), and differential pulse voltammetry (DPV). Based on the results, the PAYR/CuONP nanocomposite had significant electrocatalytic oxidation and reduction properties for the detection and determination of H2O2. Some parameters such as linear range, sensitivity, and detection limit for reduction peak were obtained as 0.1–140 μM, 1.4154 μA cm−2 μM−1, and 0.03 μM, respectively, by the DPV technique. Some advantages of this electrode were having widespread linear range, low detection limit, and, most importantly, ability in simultaneous oxidation and reduction of H2O2 at two applied potentials.







Similar content being viewed by others
Data Availability
The data sets used and/or analyzed during the current study are available from the corresponding author (Nader Amini) on reasonable request.
References
Abdelwahab AA, Shim YB (2014) Nonenzymatic H2O2 sensing based on silver nanoparticles capped polyterthiophene/MWCNT nanocomposite. Sensors Actuators B Chem 201:51–58
Agnieszka B, Anna B, Anna J, Mikołaj K, Jacek W, Marek L, Grzegorz DS (2021) A comparative study of electrocatalytic reduction of hydrogen peroxide at carbon rod electrodes decorated with silver particles. Mater Sci Eng B 263:114801
Chao L, Wang W, Dai M, Ma Y, Sun L, Qin X, Xie Q (2016) Step-by-step electrodeposition of a high-performance Prussian blue-gold nanocomposite for H2O2 sensing and glucose, biosensing. J Electroanal Chem 778:66–73
Chen Y, Li Q, Jiang H, Wang X (2016) Pt modified carbon fiber microelectrode for electrochemically catalytic reduction of hydrogen peroxide and its application in living cell H2O2 detection. J Electroanal Chem 781:233–237
Devasenathipathy R, Kohilarani K, Chen SM, Wang SF, Wang SC, Chen CK (2016) Electrochemical preparation of biomolecule stabilized copper nanoparticles decorated reduced graphene oxide for the sensitive and selective determination of hydrogen peroxide, Electrochim. Acta 191:55–61
Díaz Nieto CH, Granero AM, Lopez JC, Pierini GD, Levin GJ, Fernández H, Zon MA (2018) Development of a third generation biosensor to determine hydrogen peroxide based on a composite of soybean peroxidase/chemically reduced graphene oxide deposited on glassy carbon electrodes. Sensors Actuators B Chem 263:377–386
Doblhofer K (1980) Electrodes covered with thin, permeable polymer films, Electrochim. Acta 25(6):871–878
Du P, Zhou B, Cai C (2008) Development of an amperometric biosensor for glucose based on electrocatalytic reduction of hydrogen peroxide at the single-walled carbon nanotube/Nile blue A nanocomposite modified electrode. J Electroanal Chem 614(1):149–156
Ehsani M, Chaichi MJ, Hosseini SN (2017) Comparison of CuO nanoparticle and CuO/MWCNT nanocomposite for amplification of chemiluminescence immunoassay for detection of the hepatitis B surface antigen in biological samples. Sensors Actuators B Chem 247:319–328
Ensafi AA, Rezaloo F, Rezaei B (2016) Electrochemical sensor based on porous silicon/silver nanocomposite for the determination of hydrogen peroxide. Sensors Actuators B Chem 231:239–244
Guler M, Turkoglu V, Bulut A, Zahmakiran M (2018) Electrochemical sensing of hydrogen peroxide using Pd@Ag bimetallic nanoparticles decorated functionalized reduced graphene oxide, Electrochim. Acta 263:118–126
Hu M, Chen J, Li ZY, Au L, Hartland GV, Li X, Marquez M, Xia Y (2006) Gold nanostructures: engineering their plasmonic properties for biomedical applications. Chem Soc Rev 35:1084–1094
Hu C, Chen X, Hu S (2007a) Water-soluble single-walled carbon nanotubes films: preparation, characterization and applications as electrochemical sensing films. J Electroanal Chem 586(1):77–85
Hu C, Yang C, Hu S (2007b) Hydrophobic adsorption of surfactants on water-soluble carbon nanotubes: a simple approach to improve sensitivity and antifouling capacity of carbon nanotubes-based electrochemical sensors. Electrochem Commun 9(1):128–134
Ihara K, Hasegawa SI, Naito K (2008) The separation of aluminum(III) ions from the aqueous solution on membrane filter using Alizarin Yellow R. Talanta 75(4):944–949
Kardaş F, Beytur M, Akyıldırım O, Yüksek H, LütfiYola M, Atar N (2017) Electrochemical detection of atrazine in wastewater samples by copper oxide (CuO) nanoparticles ionic liquid modified electrode. J Mol Liq 248:360–363
Liu J, Zhou D, Liu X, Wu K, Wan C (2009) Determination of kojic acid based on the interface enhancement effects of carbon nanotube/alizarin red S modified electrode. Colloids Surf B: Biointerfaces 70(1):20–24
Liu Y, Liu X, Guo ZH, Hu ZH, Xue ZH, Lu X (2017) Horseradish peroxidase supported on porous graphene as a novel sensing platform for detection of hydrogen peroxide in living cells sensitively. Biosens Bioelectron 87:101–107
Lutfullah B, Muhammet G (2019) An ultra-sensitive non-enzymatic hydrogen peroxide sensor based on SiO2-APTES supported Au nanoparticles modified glassy carbon electrode, Progress in Natural Science. Mater Int 29:390–396
Marjan SR, Milan ZM, Dirk E, Valentin M (2019) Electrochemistry of hydrogen peroxide reduction reaction on carbon paste electrodes modified by Ag- and Pt-supported carbon microspheres. J Solid State Electrochem 23:1257–1267
Muhammet G, Vedat T, Arif K, Fatih K (2018) A novel nonenzymatic hydrogen peroxide amperometric sensor based on Pd@CeO2-NH2 nanocomposites modified glassy carbon electrode. Mater Sci Eng C 90:454–460
Pandurangan P, Babu R, Narayanan S (2014) Synergetic effect of Prussian blue film with gold nanoparticle graphite–wax composite electrode for the enzyme-free ultrasensitive hydrogen peroxide sensor. J Solid State Electrochem 18:883–891
Parthasarathy S, Nandhini V, Jeyaprakash BG (2016) Improved sensing response of photo activated ZnO thin film for hydrogen peroxide detection. J Colloid Interface Sci 482:81–88
Priyanga N, Sahaya R, Mehboobali P, Al-Sehemi AG, Siew-Moi P, Yang X, Shu-Y T, Annaraj J, Sambathkumar S, Gnanakumar G (2021) Hierarchical MnS@MoS2 architectures on tea bag filter paper for flexible, sensitive, and selective non-enzymatic hydrogen peroxide sensors. J Alloys Compd 855:157103
Sahoo S, Sahoob PK, Sudipa M, Satpati AK (2020) A novel low cost nonenzymatic hydrogen peroxide sensor based on CoFe2O4/CNTs nanocomposite modified electrode. J Electroanal Chem 876:114504
Sheng Q, Yu H, Zheng J (2007) Hydrogen peroxide determination by carbon ceramic electrodes modified with pyrocatechol violet, Electrochim. Acta 52(25):7300–7306
Shobha Jeykumari DR, Narayanan SS (2008) A novel nanobiocomposite based glucose biosensor using neutral red functionalized carbon nanotubes. Biosens Bioelectron 23(9):1404–1411
Shu Y, Li B, Xu Q, Gu P, Xiao X, Liu F, Yu L, Pang H, Hu X (2017) Cube-like CoSn(OH)6 nanostructure for sensitive electrochemical detection of H2O2 in human serum sample. Sensors Actuators B 241:528–533
Sookhakian M, Zalnezhad E, Alias Y (2017) Layer-by-layer electrodeposited nanowall-like palladium-reduced graphene oxide film as a highly-sensitive electrochemical non-enzymatic sensor. Sensors Actuators B Chem 241:1–7
Thanh TD, Balamurugan J, Lee SH, Kim NH, Lee JH (2016) Novel porous gold-palladium nanoalloy network-supported graphene as an advanced catalyst for non-enzymatic hydrogen peroxide sensing. Biosens Bioelectron 85:669–678
Vesna S, Slađana Đ, Miloš O, Jelena M, Kurt K, Dalibor MS (2020) A novel nonenzymatic hydrogen peroxide amperometric sensor based on AgNp@GNR nanocomposites modified screen-printed carbon electrode. J Electroanal Chem 876:114487
Wang I, Zhu H, Hou H, Zhang Z, Xiao X, Song Y (2011) A novel hydrogen peroxide sensor based on Ag nanoparticles electrodeposited on chitosan-graphene oxide/cysteamine-modified gold electrode. J Solid State Electrochem 16:1693–1700
Wang H, Li T, Ma J, Li K, Zuo X (2017) Silver nanoparticles selectively deposited on graphene-colloidal carbon sphere composites and their application for hydrogen peroxide sensing. Sensors Actuators B 239:1205–1212
Weina X, Jianlin L, Mingjun W, Lin C, Xue W, Chenguo H (2016) Direct growth of MnOOH nanorod arrays on a carbon cloth for high performance non-enzymatic hydrogen peroxide sensing. Anal Chim Acta 913:128–136
Williams RR, Lautenschleger M (1963) Titration of weak acids with tetramethylgnanidine as solvent. Talanta 10(7):804–808
Wu K, Hu S (2004) Deposition of a thin film of carbon nanotubes onto a glassy carbon electrode by electropolymerization. Carbon 42(15):3237–3242
Wu ZL, Li CK, Yu JG, Chen XQ (2017) MnO2/reduced graphene oxide nanoribbons: facile hydrothermal preparation and their application in amperometric detection of hydrogen peroxide. Sensors Actuators B Chem 239:544–552
Xie Y, Yu Y, Lu L, Ma X, Gong L, Huang X, Liu G, Yu Y (2018) CuO nanoparticles decorated 3D graphene nanocomposite as non-enzymatic electrochemical sensing platform for malathion detection. J Electroanal Chem 812:82–89
Yogeswaran U, Chen SM (2007) Separation and concentration effect of f-MWCNTs on electrocatalytic responses of ascorbic acid, dopamine and uric acid at f-MWCNTs incorporated with poly (neutral red) composite films. Electrochim Acta 52(19):5985–5996
Yue W, Zheng D, Hu C, Hu S (2010) Fabrication and application of poly(alizarin red S)-carbon nanotubes composite film based nitrite sensor. J Nanosci Nanotechnol 10(10):6586–6593
Yusoff N, Rameshkumar P, Mehmood MS, Pandikumar A, Lee HW, Huang NM (2017) Ternary nanohybrid of reduced graphene oxide-nafion@silver nanoparticles for boosting the sensor performance in non-enzymatic amperometric detection of hydrogen peroxide. Biosens Bioelectron 87:1020–1028
Zeng J, Wei W, Wu L, Liu X, Liu K, Li Y (2006) Fabrication of poly(toluidine blue O)/carbon nanotube composite nanowires and its stable low-potential detection of NADH. J Electroanal Chem 595(2):152–160
Zhang R, Chen W (2017) Recent advances in graphene-based nanomaterials for fabricating electrochemical hydrogen peroxide sensors. Biosens Bioelectron 89:249–268
Zhang K, Zhang Y (2010) Lable-free electrochemical DNA sensor based on gold nanoparticles/poly(neutral red) modified electrode. Electroanalysis 22(6):673–679
Zhang K, Zhang L, Xu J, Wang C, Geng T, Wang H, Zhu J (2010) A sensitive amperometric hydrogen peroxide sensor based on thionin/EDTA/carbon nanotubes—chitosan composite film modified electrode. Mikrochim Acta 171(1):139–144
Zhang K, Zhang N, Zhang L, Xu J, Wang H, Wang C, Geng T (2011) Amperometric sensing of hydrogen peroxide using a glassy cabon electode modified with silver nanoparticles on poly(alizarin yellow R). Mikrochim Acta 173:135–141
Funding
This work was partially supported by Kwangwoon University, Seoul, Korea, through Research Grant–2021.
Author information
Authors and Affiliations
Contributions
Nader Amini performed detection and determination of H2O2 using an electrochemical sensor. Bahaaldin Rashidzadeh, Nasrin Amanollahi, and Afshin Maleki evaluated the experimental data and analysis of the data set. Jae-Kyu Yang and Seung-Mok Lee provided guidance on the preparation method of the electrochemical sensor.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Responsible Editor: Angeles Blanco
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Amini, N., Rashidzadeh, B., Amanollahi, N. et al. Application of an electrochemical sensor using copper oxide nanoparticles/polyalizarin yellow R nanocomposite for hydrogen peroxide. Environ Sci Pollut Res 28, 38809–38816 (2021). https://doi.org/10.1007/s11356-021-13299-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-13299-6