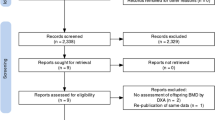
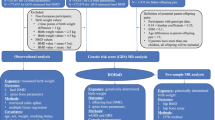
Abstract
Maternal smoking during pregnancy has detrimental effects on fetal development. The current review examined the differences in offspring’s bone mineral density (BMD) between mothers smoked during pregnancy and those who did not. A systematic review and meta-analysis on the studies investigating the influence of maternal smoking during pregnancy on children or neonates’ bone measures published up to October 30, 2018, was performed. BMD results measured at different body sites were pooled and then fixed or random effect models were used based on the presence of heterogeneity. The desired pooled effect size was the offspring’s BMD mean difference with 95% confidence interval between smoker and non-smoker mothers. Sensitivity analysis was performed for birth weight and current weight, two important mediator/confounders causing heterogeneity. Overall, eight studies consisting of 17,931 participants aged from infancy to 18 years were included. According to the fixed effect model, the mean of BMD in offspring whose mothers smoked during pregnancy was 0.01 g/cm2 lower than those with non-smoker mothers (95% CI = − 0.02 to − 0.002). However, subgroup meta-analysis adjusted for birth weight and current weight demonstrated no significant mean difference between BMD of children with smoker and non-smoker mothers (d = 0.06, 95% CI = −0.04 to 0.16, p value = 0.25 and d = − 0.005, 95% CI = − 0.01 to 0.004, p value = 0.28, respectively). According to available studies, it is suggested that maternal smoking during pregnancy does not have direct effect on the offspring’s BMD. Instead, this association might be confounded by other factors such as placental weight, birth weight, and current body size of children.



Similar content being viewed by others
References
Ackerman A, Thornton JC, Wang J, Pierson RN Jr, Horlick M (2006) Sex difference in the effect of puberty on the relationship between fat mass and bone mass in 926 healthy subjects, 6 to 18 years old. Obesity 14:819–825
Baird J, Kurshid MA, Kim M, Harvey N, Dennison E, Cooper C (2011) Does birthweight predict bone mass in adulthood? A systematic review and meta-analysis. Osteoporos Int 22:1323–1334
Brand JS, Hiyoshi A, Cao Y, Lawlor DA, Cnattingius S, Montgomery S (2020) Maternal smoking during pregnancy and fractures in offspring: national register based sibling comparison study. Bmj 368:l7057. https://doi.org/10.1136/bmj.l7057
Clark E, Ness A, Tobias J, Parents ALSo, team CS (2006a) Adipose tissue stimulates bone growth in prepubertal children. J Clin Endocrinol Metab 91:2534–2541
Clark EM, Ness AR, Bishop NJ, Tobias JH (2006b) Association between bone mass and fractures in children: a prospective cohort study. J Bone Miner Res 21:1489–1495
Clark EM, Ness AR, Tobias JH (2008) Bone fragility contributes to the risk of fracture in children, even after moderate and severe trauma. J Bone Miner Res 23:173–179
Cooper C, Cawley M, Bhalla A, Egger P, Ring F, Morton L, Barker D (1995) Childhood growth, physical activity, and peak bone mass in women. J Bone Miner Res 10:940–947
Cooper C, Fall C, Egger P, Hobbs R, Eastell R, Barker D (1997) Growth in infancy and bone mass in later life. Ann Rheum Dis 56:17–21
Cooper C, Westlake S, Harvey N, Javaid K, Dennison E, Hanson M (2006) Developmental origins of osteoporotic fracture. Osteoporos Int 17:337–347
Ducher G, Bass SL, Naughton GA, Eser P, Telford RD, Daly RM (2009) Overweight children have a greater proportion of fat mass relative to muscle mass in the upper limbs than in the lower limbs: implications for bone strength at the distal forearm. Am J Clin Nutr 90:1104–1111
Dürmuş B et al (2011) Parental smoking during pregnancy, early growth, and risk of obesity in preschool children: the Generation R Study. Am J Clin Nutr 94:164–171
Fenercioglu AK, Tamer I, Karatekin G, Nuhoglu A (2009) Impaired postnatal growth of infants prenatally exposed to cigarette smoking. Tohoku J Exp Med 218:221–228
Garnero P, Delmas P (2004) Contribution of bone mineral density and bone turnover markers to the estimation of risk of osteoporotic fracture in postmenopausal women. J Musculoskelet Nueronal Interact 4:50
Ghadimi R, Hosseini SR, Asefi S, Bijani A, Heidari B, Babaei M (2018) Influence of smoking on bone mineral density in elderly men. Int J Prev Med 9:111. https://doi.org/10.4103/ijpvm.IJPVM_234_16
Godfrey K, Walker-Bone K, Robinson S, Taylor P, Shore S, Wheeler T, Cooper C (2001) Neonatal bone mass: influence of parental birthweight, maternal smoking, body composition, and activity during pregnancy. J Bone Miner Res 16:1694–1703
Hackshaw A, Rodeck C, Boniface S (2011) Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls. Hum Reprod Update 17:589–604
Harvey N et al (2010) Maternal predictors of neonatal bone size and geometry: the Southampton Women’s Survey. J Dev Orig Health Dis 1:35–41
Heppe D, Medina-Gomez C, Hofman A, Rivadeneira F, Jaddoe V (2015) Does fetal smoke exposure affect childhood bone mass? The Generation R Study. Osteoporos Int 26:1319–1329
Herrmann M, King K, Weitzman M (2008) Prenatal tobacco smoke and postnatal secondhand smoke exposure and child neurodevelopment. Curr Opin Pediatr 20:184–190
Högler W, Schmid A, Raber G, Sölder E, Eibl G, Heinz-Erian P, Kapelari K (2003) Perinatal bone turnover in term human neonates and the influence of maternal smoking. Pediatr Res 53:817–822
Howe LD, Matijasevich A, Tilling K, Brion M-J, Leary SD, Smith GD, Lawlor DA (2012) Maternal smoking during pregnancy and offspring trajectories of height and adiposity: comparing maternal and paternal associations. Int J Epidemiol 41:722–732
Jaddoe VW, Verburg BO, de Ridder MA, Hofman A, Mackenbach JP, Moll HA, Steegers EA, Witteman JC (2007) Maternal smoking and fetal growth characteristics in different periods of pregnancy: the generation R study. Am J Epidemiol 165:1207–1215
Jauniaux E, Burton GJ (2007) Morphological and biological effects of maternal exposure to tobacco smoke on the feto-placental unit. Early Hum Dev 83:699–706
Javaid MK, Cooper C (2002) Prenatal and childhood influences on osteoporosis. Best Pract Res Clin Endocrinol Metab 16:349–367
Jones G, Riley M, Dwyer T (1999) Maternal smoking during pregnancy, growth, and bone mass in prepubertal children. J Bone Miner Res 14:146–151
Jones G, Hynes K, Dwyer T (2013) The association between breastfeeding, maternal smoking in utero, and birth weight with bone mass and fractures in adolescents: a 16-year longitudinal study. Osteoporos Int 24:1605–1611
Juonala M et al (2019) Childhood exposure to passive smoking and bone health in adulthood. The Cardiovascular Risk in Young Finns Study. J Clin Endocrinol Metab. https://doi.org/10.1210/jc.2018-02501
Leonard MB, Shults J, Wilson BA, Tershakovec AM, Zemel BS (2004) Obesity during childhood and adolescence augments bone mass and bone dimensions. Am J Clin Nutr 80:514–523
Lieberman E, Gremy I, Lang JM, Cohen AP (1994) Low birthweight at term and the timing of fetal exposure to maternal smoking. Am J Public Health 84:1127–1131
Macdonald-Wallis C, Tobias JH, Smith GD, Lawlor DA (2011) Parental smoking during pregnancy and offspring bone mass at age 10 years: findings from a prospective birth cohort. Osteoporos Int 22:1809–1819
Martínez-Mesa J, Menezes AM, González DA, Horta BL, Matijasevich A, Gigante DP, Hallal PC (2012) Life course association of maternal smoking during pregnancy and offspring's height: data from the 1993 Pelotas (Brazil) birth cohort. J Adolesc Health 51:S53–S57
Martinez-Mesa J, Restrepo-Méndez M, González D, Wehrmeister F, Horta B, Domingues M, Menezes A (2013) Life-course evidence of birth weight effects on bone mass: systematic review and meta-analysis. Osteoporos Int 24:7–18
Martínez-Mesa J, Menezes AM, Howe LD, Wehrmeister FC, Muniz LC, González-Chica DA, Assunção MC, Gonçalves H, Barros FC (2014) Lifecourse relationship between maternal smoking during pregnancy, birth weight, contemporaneous anthropometric measurements and bone mass at 18 years old. The 1993 Pelotas Birth Cohort. Early Hum Dev 90:901–906
Meredith HV (1975) Relation between tobacco smoking of pregnant women and body size of their progeny: a compilation and synthesis of published studies. Hum Biol 47:451–472
Parviainen R, Auvinen J, Pokka T, Serlo W, Sinikumpu J-J (2017) Maternal smoking during pregnancy is associated with childhood bone fractures in offspring–a birth-cohort study of 6718 children. Bone 101:202–205
Rubin C et al (2009) Timing of maturation and predictors of menarche in girls enrolled in a contemporary British cohort. Paediatr Perinat Epidemiol 23:492–504. https://doi.org/10.1111/j.1365-3016.2009.01055.x
Sayers A, Tobias JH (2010) Fat mass exerts a greater effect on cortical bone mass in girls than boys. J Clin Endocrinol Metab 95:699–706
Suter MA, Anders AM, Aagaard KM (2012) Maternal smoking as a model for environmental epigenetic changes affecting birthweight and fetal programming. Mol Hum Reprod 19:1–6
Tandon N et al (2012) Growth from birth to adulthood and peak bone mass and density data from the New Delhi Birth Cohort. Osteoporos Int 23:2447–2459
Timpson NJ, Sayers A, Davey-Smith G, Tobias JH (2009) How does body fat influence bone mass in childhood? A Mendelian randomization approach. J Bone Miner Res 24:522–533
van Leeuwen J, Koes B, Paulis W, van Middelkoop M (2017) Differences in bone mineral density between normal-weight children and children with overweight and obesity: a systematic review and meta-analysis. Obes Rev 18:526–546
Venkataraman PS, Duke JC (1991) Bone mineral content of healthy, full-term neonates: effect of race, gender, and maternal cigarette smoking. Am J Dis Child 145:1310–1312
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP (2008) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 61:344–349. https://doi.org/10.1016/j.jclinepi.2007.11.008
Von Kries R, Toschke AM, Koletzko B, Slikker W Jr (2002) Maternal smoking during pregnancy and childhood obesity. Am J Epidemiol 156:954–961
Wang X, Tager IB, Van Vunakis H, Speizer FE, Hanrahan JP (1997) Maternal smoking during pregnancy, urine cotinine concentrations, and birth outcomes. A prospective cohort study. Int J Epidemiol 26:978–988
Williams LA, Evans SF, Newnham JP (1997) Prospective cohort study of factors influencing the relative weights of the placenta and the newborn infant. Bmj 314:1864
Wong PK, Christie JJ, Wark JD (2007) The effects of smoking on bone health. Clin Sci 113:233–241
Author information
Authors and Affiliations
Corresponding authors
Additional information
Editorial Responsibility: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Search string for PubMed (www.pubmed.com)
(((((“Bone Density”[Title/Abstract] OR “Bone Densities”[Title/Abstract] OR “Density, Bone”[Title/Abstract] OR “Bone Mineral density”[Title/Abstract] OR “Bone Mineral Densities”[Title/Abstract] OR “Density, Bone Mineral”[Title/Abstract] OR “Bone Mineral Content”[Title/Abstract] OR “Bone Mineral Contents”[Title/Abstract] OR “Bone mass”[Title/Abstract] OR BMD[Title/Abstract] OR BMC[Title/Abstract])) OR “Bone Density”[Mesh])) AND ((“Maternal smoking”[Title/Abstract] OR “Maternal smoke”[Title/Abstract] OR “Maternal cigarette”[Title/Abstract] OR “ Maternal cigarette smoke”[Title/Abstract] OR “ Maternal cigarette smoking”[Title/Abstract] OR “Pregnancy smoking”[Title/Abstract] OR “Pregnancy smoke”[Title/Abstract] OR “Pregnancy cigarette”[Title/Abstract] OR “Pregnancy cigarette smoke”[Title/Abstract] OR “Pregnancy cigarette smoking”[Title/Abstract] OR “Prenatal smoking”[Title/Abstract] OR “Prenatal smoke”[Title/Abstract] OR “Prenatal cigarette”[Title/Abstract] OR “ Prenatal cigarette smoke”[Title/Abstract] OR “Prenatal cigarette smoking”[Title/Abstract])))
Search string for Web of Science
TITLE: (“Bone Density” OR “Bone Densities” OR “Density, Bone” OR “Bone Mineral density” OR “Bone Mineral Densities” OR “Density, Bone Mineral” OR “Bone Mineral Content” OR “Bone Mineral Contents” OR “Bone mass” OR BMD OR BMC) AND TITLE: (“Maternal smoking” OR “Maternal smoke” OR “Maternal cigarette” OR “ Maternal cigarette smoke” OR “ Maternal cigarette smoking” OR “Pregnancy smoking” OR “Pregnancy smoke” OR “Pregnancy cigarette” OR “Pregnancy cigarette smoke” OR “Pregnancy cigarette smoking” OR “Prenatal smoking” OR “Prenatal smoke” OR “Prenatal cigarette” OR “ Prenatal cigarette smoke” OR “Prenatal cigarette smoking”).
Timespan: All years. Indexes: SCI-EXPANDED, SSCI, CPCI-S, CPCI-SSH, ESCI.
Search string for Scopus (www.scopus.com)
(TITLE-ABS-KEY (“Bone Density” OR “Bone Densities” OR “Density, Bone” OR “Bone Mineral density” OR “Bone Mineral Densities” OR “Density, Bone Mineral” OR “Bone Mineral Content” OR “Bone Mineral Contents” OR “Bone mass” OR bmd OR bmc) AND TITLE-ABS-KEY (“Maternal smoking” OR “Pregnancy smoking” OR “Prenatal smoking”))
Rights and permissions
About this article
Cite this article
Baradaran Mahdavi, S., Daniali, S.S., Farajzadegan, Z. et al. Association between maternal smoking and child bone mineral density: a systematic review and meta-analysis. Environ Sci Pollut Res 27, 23538–23549 (2020). https://doi.org/10.1007/s11356-020-08740-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08740-1