Abstract
The use of silver nanoparticles (AgNPs) in agriculture and many consumer products has led to a significant release of Ag in the environment. Although Ag toxicity in terrestrial organisms has been studied extensively, very little is known about the accumulation capacity and coping mechanisms of organisms in Ag-contaminated soil. In this context, we exposed Eisenia fetida earthworms to artificial OECD soil spiked with a range of concentrations of Ag (AgNPs or AgNO3). The main aims were to (1) identify the location and form of accumulation of Ag in the exposed earthworms and (2) better understand the physiological mechanisms involved in Ag detoxification. The results showed that similar doses of AgNPs or AgNO3 did not have the same effect on E. fetida survival. The two forms of Ag added to soil exhibited substantial differences in speciation at the end of exposure, but the Ag speciation and content of Ag in earthworms were similar, suggesting that biotransformation of Ag occurred. Finally, 3D images of intact earthworms obtained by X-ray micro-computed tomography revealed that Ag accumulated preferentially in the chloragogen tissue, coelomocytes, and nephridial epithelium. Thus, E. fetida bioaccumulates Ag, but a regulation mechanism limits its impact in a very efficient manner. The location of Ag in the organism, the competition between Ag and Cu, and the speciation of internal Ag suggest a link between Ag and the thiol-rich proteins that are widely present in these tissues, most probably metallothioneins, which are key proteins in the sequestration and detoxification of metals.





Similar content being viewed by others
References
Baccaro M, Undas AK, de Vriendt J, van den Berg JHJ, Peters RJB, van den Brink NW (2018) Ageing, dissolution and biogenic formation of nanoparticles: how do these factors affect the uptake kinetics of silver nanoparticles in earthworms? Environ Sci Nano 5:1107–1116. https://doi.org/10.1039/C7EN01212H
Bernard F, Brulle F, Douay F, Lemière S, Demuynck S, Vandenbulcke F (2010) Metallic trace element body burdens and gene expression analysis of biomarker candidates in Eisenia fetida, using an “exposure/depuration” experimental scheme with field soils. Ecotoxicol Environ Saf 73:1034–1045. https://doi.org/10.1016/j.ecoenv.2010.01.010
Bourdineaud J-P, Štambuk A, Šrut M, Radić Brkanac S, Ivanković D, Lisjak D, Sauerborn Klobučar R, Dragun Z, Bačić N, Klobučar GIV (2019) Gold and silver nanoparticles effects to the earthworm Eisenia fetida – the importance of tissue over soil concentrations. Drug Chem Toxicol:1–18. https://doi.org/10.1080/01480545.2019.1567757
Brulle F, Cocquerelle C, Wamalah AN, Morgan AJ, Kille P, Leprêtre A, Vandenbulcke F (2008) cDNA cloning and expression analysis of Eisenia fetida (Annelida: Oligochaeta) phytochelatin synthase under cadmium exposure. Ecotoxicol Environ Saf 71:47–55. https://doi.org/10.1016/j.ecoenv.2007.10.032
Carbonell G, Pro J, Gómez N, Babín MM, Fernández C, Alonso E, Tarazona JV (2009) Sewage sludge applied to agricultural soil: ecotoxicological effects on representative soil organisms. Ecotoxicol Environ Saf 72:1309–1319. https://doi.org/10.1016/j.ecoenv.2009.01.007
Chaurand P, Liu W, Borschneck D, Levard C, Auffan M, Paul E, Collin B, Kieffer I, Lanone S, Rose J, Perrin J (2018) Multi-scale X-ray computed tomography to detect and localize metal-based nanomaterials in lung tissues of in vivo exposed mice. Sci Rep 8:1–11. https://doi.org/10.1038/s41598-018-21862-4
Chhipa H (2019) Applications of nanotechnology in agriculture. Methods Microbiol:115–142
Courtois P, Rorat A, Lemiere S, Guyoneaud R, Attard E, Levard C, Vandenbulcke F (2019) Ecotoxicology of silver nanoparticles and their derivatives introduced in soil with or without sewage sludge: a review of effects on microorganisms, plants and animals. Environ Pollut 253:578–598. https://doi.org/10.1016/j.envpol.2019.07.053
Coutris C, Hertel-Aas T, Lapied E, Joner EJ, Oughton DH (2011) Bioavailability of cobalt and silver nanoparticles to the earthworm Eisenia fetida. Nanotoxicology 6:186–195. https://doi.org/10.3109/17435390.2011.569094
Curieses Silvana P, García-Velasco N, Urionabarrenetxea E, Elena SM, Bilbao E, Di Marzio Walter D, Soto M (2017) Responses to silver nanoparticles and silver nitrate in a battery of biomarkers measured in coelomocytes and in target tissues of Eisenia fetida earthworms. Ecotoxicol Environ Saf 141:57–63. https://doi.org/10.1016/j.ecoenv.2017.03.008
Davidson SK, Powell R, James S (2013) A global survey of the bacteria within earthworm nephridia. Mol Phylogenet Evol 67:188–200. https://doi.org/10.1016/j.ympev.2012.12.005
Demuynck S, Grumiaux F, Mottier V, Schikorski D, Lemière S, Leprêtre A (2006) Metallothionein response following cadmium exposure in the oligochaete Eisenia fetida. Comp Biochem Physiol Toxicol Pharmacol CBP 144:34–46. https://doi.org/10.1016/j.cbpc.2006.05.004
Diez-Ortiz M, Lahive E, George S, Ter Schure A, Van Gestel CAM, Jurkschat K, Svendsen C, Spurgeon DJ (2015a) Short-term soil bioassays may not reveal the full toxicity potential for nanomaterials; bioavailability and toxicity of silver ions (AgNO3) and silver nanoparticles to earthworm Eisenia fetida in long-term aged soils. Environ Pollut 203:191–198. https://doi.org/10.1016/j.envpol.2015.03.033
Diez-Ortiz M, Lahive E, Kille P, Powell K, Morgan AJ, Jurkschat K, Van Gestel CAM, Mosselmans JFW, Svendsen C, Spurgeon DJ (2015b) Uptake routes and toxicokinetics of silver nanoparticles and silver ions in the earthworm Lumbricus rubellus. Environ Toxicol Chem 34:2263–2270. https://doi.org/10.1002/etc.3036
Fischer E (1993) The myelo-erythroid nature of the chloragogenous-like tissues of the annelids. Comp Biochem Physiol A Physiol 106:449–453. https://doi.org/10.1016/0300-9629(93)90237-X
Garcia-Velasco N, Gandariasbeitia M, Irizar A, Soto M (2016) Uptake route and resulting toxicity of silver nanoparticles in Eisenia fetida earthworm exposed through standard OECD tests. Ecotoxicology 25:1543–1555. https://doi.org/10.1007/s10646-016-1710-2
Garcia-Velasco N, Peña-Cearra A, Bilbao E, Zaldibar B, Soto M (2017) Integrative assessment of the effects produced by Ag nanoparticles at different levels of biological complexity in Eisenia fetida maintained in two standard soils (OECD and LUFA 2.3). Chemosphere 181:747–758. https://doi.org/10.1016/j.chemosphere.2017.04.143
Gomes SIL, Hansen D, Scott-Fordsmand JJ, Amorim MJB (2015) Effects of silver nanoparticles to soil invertebrates: oxidative stress biomarkers in Eisenia fetida. Environ Pollut 199:49–55. https://doi.org/10.1016/j.envpol.2015.01.012
Hamed SS, Kauschke E, Cooper EL (2002) Cytochemical properties of earthworm coelomocytes enriched by Percoll, in: a new model for analyzing antimicrobial peptides with biomedical applications. NATO Science Series. https://doi.org/10.3923/ijzr.2005.74.83
Hayashi Y, Heckmann L-H, Simonsen V, Scott-Fordsmand JJ (2013) Time-course profiling of molecular stress responses to silver nanoparticles in the earthworm Eisenia fetida. Ecotoxicol Environ Saf 98:219–226. https://doi.org/10.1016/j.ecoenv.2013.08.017
Heckmann L-H, Hovgaard MB, Sutherland DS, Autrup H, Besenbacher F, Scott-Fordsmand JJ (2011) Limit-test toxicity screening of selected inorganic nanoparticles to the earthworm Eisenia fetida. Ecotoxicology 20:226–233. https://doi.org/10.1007/s10646-010-0574-0
Homa J, Rorat A, Kruk J, Cocquerelle C, Plytycz B, Vandenbulcke F (2015) Dermal exposure of Eisenia andrei earthworms: effects of heavy metals on metallothionein and phytochelatin synthase gene expressions in coelomocytes. Environ Toxicol Chem 34:1397–1404. https://doi.org/10.1002/etc.2944
Kaegi R, Voegelin A, Sinnet B, Zuleeg S, Hagendorfer H, Burkhardt M, Siegrist H (2011) Behavior of metallic silver nanoparticles in a pilot wastewater treatment plant. Environ Sci Technol 45:3902–3908. https://doi.org/10.1021/es1041892
Kaegi R, Voegelin A, Ort C, Sinnet B, Thalmann B, Krismer J, Hagendorfer H, Elumelu M, Mueller E (2013) Fate and transformation of silver nanoparticles in urban wastewater systems. Water Res 47:3866–3877. https://doi.org/10.1016/j.watres.2012.11.060
Khan MR, Rizvi TF (2017) Application of nanofertilizer and nanopesticides for improvements in crop production and protection. In: Ghorbanpour M, Manika K, Varma A (eds) Nanoscience and plant–soil systems. Soil Biology. Springer International Publishing, Cham, pp 405–427. https://doi.org/10.1007/978-3-319-46835-8_15
Klein CL, Stahlmecke B, Romazanov J, Kuhlbusch TAJ, Van Doren E, De Temmerman P-J, Mast J, Wick P, Krug H, Locoro G, Hund-Rinke K, Kördel W, Friedrichs S, Maier G, Werner J, Linsinger T, Gawlik BM, Comero S, Institute for Health and Consumer Protection, European Commission, Joint Research Centre, Institute for Environment and Sustainability, Institute for Reference Materials and Measurements (2011) NM-series of representative manufactured nanomaterials: NM-300 silver characterisation, stability, homogeneity. Publications Office, Luxembourg
Lapied E, Moudilou E, Exbrayat J-M, Oughton DH, Joner EJ (2010) Silver nanoparticle exposure causes apoptotic response in the earthworm Lumbricus terrestris (Oligochaeta). Nanomed. 5:975–984. https://doi.org/10.2217/nnm.10.58
Levard C, Hotze EM, Lowry GV, Brown GE (2012) Environmental transformations of silver nanoparticles: impact on stability and toxicity. Environ Sci Technol 46:6900–6914. https://doi.org/10.1021/es2037405
Ma R, Levard C, Judy JD, Unrine JM, Durenkamp M, Martin B, Jefferson B, Lowry GV (2014) Fate of zinc oxide and silver nanoparticles in a pilot wastewater treatment plant and in processed biosolids. Environ Sci Technol 48:104–112. https://doi.org/10.1021/es403646x
McGillicuddy E, Murray I, Kavanagh S, Morrison L, Fogarty A, Cormican M, Dockery P, Prendergast M, Rowan N, Morris D (2017) Silver nanoparticles in the environment: sources, detection and ecotoxicology. Sci Total Environ 575:231–246. https://doi.org/10.1016/j.scitotenv.2016.10.041
Mendes LA, Maria VL, Scott-Fordsmand JJ, Amorim MJB (2015) Ag nanoparticles (Ag NM300K) in the terrestrial environment: effects at population and cellular level in Folsomia candida (Collembola). Int J Environ Res Public Health 12:12530–12542. https://doi.org/10.3390/ijerph121012530
Morgan JE, Morgan AJ (1993) Seasonal changes in the tissue-metal (Cd, Zn and Pb) concentrations in two ecophysiologically dissimilar earthworm species: pollution-monitoring implications. Environ Pollut 82:1–7. https://doi.org/10.1016/0269-7491(93)90155-H
Morgan AJ, Stürzenbaum SR, Winters C, Grime GW, Aziz NAA, Kille P (2004) Differential metallothionein expression in earthworm (Lumbricus rubellus) tissues. Ecotoxicol Environ Saf 57:11–19. https://doi.org/10.1016/j.ecoenv.2003.08.022
Nordberg GF (1989) Modulation of metal toxicity by metallothionein. Biol Trace Elem Res 21:131–135. https://doi.org/10.1007/BF02917245
Novo M, Lahive E, Díez-Ortiz M, Matzke M, Morgan AJ, Spurgeon DJ, Svendsen C, Kille P (2015) Different routes, same pathways: molecular mechanisms under silver ion and nanoparticle exposures in the soil sentinel Eisenia fetida. Environ Pollut 205:385–393. https://doi.org/10.1016/j.envpol.2015.07.010
OCDE (1984) Ver de terre, essai de toxicité aigüe, Ligne directrice N°207. Ligne directrice de l’OCDE pour les essais de produits chimiques, OCDE, Paris
Pradas del Real AE, Vidal V, Carrière M, Castillo-Michel H, Levard C, Chaurand P, Sarret G (2017) Silver nanoparticles and wheat roots: a complex interplay. Environ Sci Technol 51:5774–5782. https://doi.org/10.1021/acs.est.7b00422
Proux O, Biquard X, Lahera E, Menthonnex J-J, Prat A, Ulrich O, Soldo Y, Trévisson P, Kapoujyan G, Perroux G, Taunier P, Grand D, Jeantet P, Deléglise M, Roux J-P, Hazemann J-L (2005) FAME : a new beamline for X-ray absorption investigations of very-diluted systems of environmental, material and biological interests. Phys Scr 115:970–973. https://doi.org/10.1238/Physica.Topical.115a00970
R Core Team (2008) R: a language and environment for statistical computing. In: R Foundation for statistical computing. Austria. URL, Vienna https://www.R-project.org/
Ravel B, Newville M (2005) Athena, Artemis, Hephaestus: data analysis for X-ray absorption spectroscopy using IFEFFIT. J Synchrotron Radiat 12:537–541. https://doi.org/10.1107/S0909049505012719
Roubalová R, Płytycz B, Procházková P, Navarro Pacheco NI, Bilej M (2018) Annelida: environmental interactions and ecotoxicity in relation to the earthworm immune system. In: Cooper EL (ed) Advances in comparative immunology. Springer International Publishing, Cham, pp 933–951. https://doi.org/10.1007/978-3-319-76768-0_27
Shoults-Wilson WA, Reinsch BC, Tsyusko OV, Bertsch PM, Lowry GV, Unrine JM (2010) Effect of silver nanoparticle surface coating on bioaccumulation and reproductive toxicity in earthworms ( Eisenia fetida ). Nanotoxicology 5:432–444. https://doi.org/10.3109/17435390.2010.537382
Sugawara N, Sugawara C (1984) Comparative study of effect of acute administration of cadmium and silver on ceruloplasmin and metallothionein: involvement of disposition of copper, iron, and zinc. Environ Res 35:507–515. https://doi.org/10.1016/0013-9351(84)90157-9
Unrine, J., Bertsch, P., Hunyadi, S., 2008. Bioavailability, trophic transfer, and toxicity of manufactured metal and metal oxide nanoparticles in terrestrial environments, in: Nanoscience and nanotechnology. John Wiley & Sons, Ltd, pp. 345–366. https://doi.org/10.1002/9780470396612.ch14
Usman K, Khan S, Ghulam S, Khan MU, Khan N, Khan MA, Khalil SK (2012) Sewage sludge: an important biological resource for sustainable agriculture and its environmental implications. Am J Plant Sci 03:1708–1721. https://doi.org/10.4236/ajps.2012.312209
Vance ME, Kuiken T, Vejerano EP, McGinnis SP, Hochella MF, Rejeski D, Hull MS (2015) Nanotechnology in the real world: redeveloping the nanomaterial consumer products inventory. Beilstein J Nanotechnol 6:1769–1780. https://doi.org/10.3762/bjnano.6.181
Vijver MG, Vink JPM, Miermans CJH, van Gestel CAM (2003) Oral sealing using glue: a new method to distinguish between intestinal and dermal uptake of metals in earthworms. Soil Biol Biochem 35:125–132. https://doi.org/10.1016/S0038-0717(02)00245-6
Vijver MG, Van Gestel CAM, Lanno RP, Van Straalen NM, Peijnenburg WJGM (2004) Internal metal sequestration and its ecotoxicological relevance: a review. Environ Sci Technol 38:4705–4712. https://doi.org/10.1021/es040354g
Yan A, Chen Z (2019) Impacts of silver nanoparticles on plants: a focus on the phytotoxicity and underlying mechanism. Int J Mol Sci 20:1003. https://doi.org/10.3390/ijms20051003
Yu S, Yin Y, Liu J (2013) Silver nanoparticles in the environment. Env Sci Process Impacts 15:78–92. https://doi.org/10.1039/C2EM30595J
Acknowledgments
The authors wish to thank Dominique Dubois, Olivier Proux, Géraldine Sarret, Ana Elena Pradas Del Real, Kerstin Hund-Rinke and Régine Leroux for their help and fruitful discussions.
Funding
This study was funded mainly by the ANSES in the ETNA2 project context, by a grant of the University of Lille and the SMRE doctoral school and by a public grant overseen by the French National Research Agency (ANR) as part of the French platform Nano-ID (EQUIPEX project ANR-10-EQPX-39-01).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Chris Lowe
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplementary 1
Scheme of the experimental design. “Control” corresponds to the microcosm without any inputs. “AgNPs” corresponds to silver nanoparticles. “Dis” corresponds to dispersant. “AgNO3” corresponds to silver nitrate. The concentrations C1, C2, C3 and C4 correspond to 33 (± 16), 71 (± 8), 117 (± 11) and 277 (± 24) mg kg-1 of Ag (dry matter) (the mean concentrations of AgNO3 and AgNPs were not significantly different). The volumes D1, D2, D3 and D4 correspond to the volumes of dispersant added to the microcosms. Dispersant was added in the same amount as in the corresponding AgNPs microcosms, that is, 1.599, 2.666, 5.331 and 10.662 mL. (PPTX 65.4 kb)
Supplementary 2
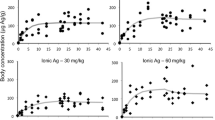
Metal contents in earthworms (mean in mg kg-1). The results were obtained by ICP analysis. “Control” corresponds to the microcosm without silver addition. “AgNPs” corresponds to silver nanoparticles. “Dis” corresponds to dispersant. “AgNO3” corresponds to silver nitrate. The concentrations C1, C2, C3 and C4 correspond to 33 (± 20), 71 (± 10), 117 (± 15) and 277 (± 45) mg kg-1 (dry matter) (the mean concentrations of AgNO3 and AgNPs were not significantly different). Stars (*) indicate significant differences between the condition with Ag and the associated control without Ag. Standard deviations are in parentheses. (DOCX 13.7 kb)
Supplementary 3
3D imaging of an earthworm by micro-CT. (top) Selection of FOV for high-resolution micro-CT scan. (bottom) 3D image analysis procedure for isolating Ag accumulation areas (normalization and thresholding step). (PPTX 692 kb)
Rights and permissions
About this article
Cite this article
Courtois, P., Rorat, A., Lemiere, S. et al. Accumulation, speciation and localization of silver nanoparticles in the earthworm Eisenia fetida. Environ Sci Pollut Res 28, 3756–3765 (2021). https://doi.org/10.1007/s11356-020-08548-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08548-z