Abstract
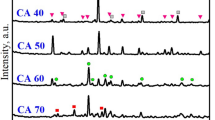
In this study, indirect carbonation was carried out by using cement kiln dust (CKD), an alkaline industrial by-product, and three chelating agents (citrate, malonate, and adipate salts) as solvents at the room temperature and atmospheric pressure. We derived the optimum conditions for eluting Ca from CKD, as well as those for storing CO2 and producing CaCO3 through carbonation. The most important factor affecting the Ca elution from CKD was the solvent concentration and that for the carbonation was the end-of-carbonation pH. Under the optimum conditions of Ca elution, the molar ratios of Ca and solvent in eluates were 1:1, 1:2, and 1:2, respectively, using citrate, malonate, and adipate solvents. Based on the results, we propose that one molecule of Ca ion and one molecule citrate that is tridentate are combined to form a complex. The bidentate malonate and adipate, on the other hand, form complexes by combining one molecule of Ca ion and two molecules of each solvent. It is essential to raise the pH while simultaneously minimizing the amount of free chelating agent in solution to produce more CaCO3 and prevent its dissolution. Besides, it is absolutely necessary to terminate the carbonation reaction at a pH of about 10.5 to improve the reuse efficiency of the chelating agent. CaCO3 produced through carbonation reaction started to dissolve at pH approximately 10.5. All of the CaCO3 produced was calcite with a purity of 98%. The efficiency of Ca elution from CKD using three solvents increased significantly with increasing stability constant of a Ca-ligand complex, but the efficiency of carbonation was the same for all solvents.






Similar content being viewed by others
References
Azdarpour A, Asadullah M, Mohammadian E, Hamidi H, Junin R, Karaei MA (2015) A review on carbon dioxide mineral carbonation through pH-swing process. Chem Eng J 279:615–630. https://doi.org/10.1016/j.cej.2015.05.064
Baldyga J, Henczka M, Sokolnicka K (2010) Utilization of carbon dioxide by chemically accelerated mineral carbonation. Mater Lett 64:702–704. https://doi.org/10.1016/j.matlet.2009.12.043
Baldyga J, Henczka M, Sokolnicka K (2011) Mineral carbonation accelerated by dicarboxylic acids as a disposal process of carbon dioxide. Chem Eng Res Des 89:1841–1854. https://doi.org/10.1016/j.cherd.2011.02.034
Eloneva S, Said A, Fogelholm CJ, Zevenhoven R (2012) Preliminary assessment of a method utilizing carbon dioxide and steelmaking slags to produce precipitated calcium carbonate. Appl Energy 90(1):329–334. https://doi.org/10.1016/j.apenergy.2011.05.045
Furia TE (1972) Stability constants (log K1) of various metal chelates in CRC handbook of food additives, 2nd ed.
Ghoorah M, Dlugogorski BZ, Balucan RD, Kennedy EM (2014) Selection of acid for weak acid processing of wollastonite for mineralization of CO2. Fuel 122:277–286. https://doi.org/10.1016/j.fuel.2014.01.015
Kim D, Kim MJ (2018a) Calcium extraction from paper sludge ash using various solvents to store carbon dioxide. KSCE J Civ Eng 22(1):4799–4805. https://doi.org/10.1007/s12205-017-0819-z
Kim MJ, Kim D (2018b) Maximization of CO2 storage for various solvent types in indirect carbonation using paper sludge ash. Environ Sci Pollut Res Int 25:30101–30109. https://doi.org/10.1007/s11356-018-2970-6
Kim MJ, Pak SY, Kim D, Jung S (2017) Optimum conditions for extracting Ca from CKD to store CO2 through indirect mineral carbonation. KSCE J Civ Eng 21(3):629–635. https://doi.org/10.1007/s12205-016-0913-7
Krevor SC, Lackner KS (2011) Enhancing serpentine dissolution kinetics for mineral carbon dioxide sequestration. Int J Greenh Gas Con 5:1073–1080. https://doi.org/10.1016/j.ijggc.2011.01.006
Li Y, Ma X, Wang W, Chi C, Shi J, Duan L (2017) Enhanced CO2 capture capacity of limestone by discontinuous addition of hydrogen chloride in carbonation at calcium looping conditions. Chem Eng J 316:438–448. https://doi.org/10.1016/j.cej.2017.01.127
Morel MM, Hering JG (1993) Principles and applications of aquatic chemistry. Wiley, Hoboken
Mun M, Cho H, Kwon J, Kim K, Kim R (2017) Investigation of the CO2 sequestration by indirect aqueous carbonation of waste cement. Am J Clim Chang 6(1):132–150. https://doi.org/10.4236/ajcc.2017.61008
Rahmani O (2018) CO2 sequestration by indirect mineral carbonation of industrial waste red gypsum. J CO2 Util 27:374–380. https://doi.org/10.1016/j.jcou.2018.08.017
Sanna A, Uibu M, Caramanna G, Kuusik R, Maroto-Valer MM (2014) A review of mineral carbonation technologies to sequester CO2. Chem Soc Rev 43:8049–8080. https://doi.org/10.1039/c4cs00035h
Santos RM, Chiang YW, Elsen J, Gerven TV (2014) Distinguishing between carbonate and non-carbonate precipitates from the carbonation of calcium-containing organic acid leachates. Hydrometallurgy 147-148:90–94. https://doi.org/10.1016/j.hydromet.2014.05.001
Song K, Kim W, Park S, Bang JH, Jeon CW, Ahn JW (2016) Effect of polyacrylic acid on direct aqueous mineral carbonation of flue gas desulfurization gypsum. Chem Eng J 301:51–57. https://doi.org/10.1016/j.cej.2016.04.142
Sun Y, Parikh V, Zhang L (2012) Sequestration of carbon dioxide by indirect mineralization using Victorian brown coal fly ash. J Hazard Mater 209-210:458–466. https://doi.org/10.1016/j.jhazmat.2012.01.053
Swaddle TW (1997) Inorganic chemistry: an industrial and environmental perspective. Academic Press, San Diego
Westin KJ, Rasmuson AC (2003) Precipitation of calcium carbonate in the presence of citrate and EDTA. Desalination 159:107–118. https://doi.org/10.1016/S0011-9164(03)90063-4
Zhao H, Park Y, Lee DH, Park AH (2013) Tuning the dissolution kinetics of wollastonite via chelating agents for CO2 sequestration with integrated synthesis of precipitated calcium carbonates. Phys Chem Chem Phys 15(36):15185–15192. https://doi.org/10.1039/c3cp52459k
Acknowledgments
This work was supported by a National Research Foundation (NRF) of Korea grant funded by the Korean government (NRF-2017R1A2B4008114) and by the Open Laboratory Operational Business Developing and Diffusing the Regional Specialization Technology funded by the Busan Institute of S&T Evaluation and Planning (B0060327000771). This research was also a part of the project titled “Production of highly functional calcium-magnesium composites based on CCUS using seawater and shell and raw material standardization,” funded by the Ministry of Oceans and Fisheries, Korea.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, MJ., Jung, S. Calcium elution from cement kiln dust using chelating agents, and CO2 storage and CaCO3 production through carbonation. Environ Sci Pollut Res 27, 20490–20499 (2020). https://doi.org/10.1007/s11356-020-08403-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08403-1