Abstract
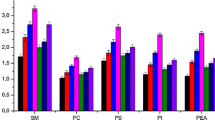
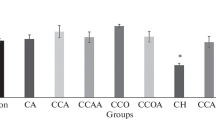
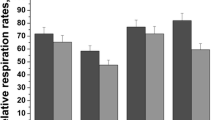
Acetaldehyde is a toxic, mutagenic and carcinogenic metabolite of alcohol which can bind to proteins, DNA and several other cellular macromolecules. Chronic alcohol consumption increases intracellular acetaldehyde levels which enhances the generation of reactive oxygen and nitrogen species (ROS and RNS). In this study, we have examined the effect of acetaldehyde on human erythrocytes under in vitro conditions. Treatment of human erythrocytes with different concentrations of acetaldehyde (0.05–2 mM) for 24 h at 37 °C increased intracellular generation of ROS and RNS. It also increased oxidation of proteins and lipids but decreased glutathione, total sulphhydryl and free amino group content. Methemoglobin level was increased accompanied by a decrease in methemoglobin reductase activity. Acetaldehyde impaired the antioxidant defence system and lowered the total antioxidant capacity of the cell. It decreased the activity of metabolic and membrane-bound enzymes and altered erythrocyte morphology. Our results show that acetaldehyde enhances the generation of ROS and RNS that results in oxidative modification of cellular components. This will lower the oxygen transporting ability of blood and shorten erythrocyte lifespan (red cell senescence).






Similar content being viewed by others
Abbreviations
- AA:
-
Acetaldehyde
- ABTS:
-
2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)
- AFR:
-
Ascorbate free radical
- ATP:
-
Adenosine 5′-triphosphate
- ATPase:
-
Adenosine triphosphatase
- CUPRAC:
-
Cupric reducing antioxidant capacity
- DCFH-DA:
-
2,7-Dichlorodihydrofluorescein diacetate
- DHE:
-
Dihydroethidium
- DPPH:
-
2,2-Diphenyl-1-picrylhydrazyl
- DTNB:
-
5,5′-Dithiobisnitrobenzoic acid
- FRAP:
-
Ferric-reducing/antioxidant power
- G6PD:
-
Glucose 6-phosphate dehydrogenase
- GSH:
-
Reduced glutathione
- GSSG:
-
Oxidized glutathione
- H2O2 :
-
Hydrogen peroxide
- K3Fe(CN)6 :
-
Potassium ferricyanide
- LDH:
-
Lactate dehydrogenase
- LPO:
-
Lipid peroxidation
- MDA:
-
Malondialdehyde
- MetHb:
-
Methemoglobin
- NAD+ and NADH:
-
Oxidized and reduced nicotinamide adenine dinucleotide
- NADP+ and NADPH:
-
Oxidized and reduced nicotinamide adenine dinucleotide phosphate
- Na,K-ATPase:
-
Sodium-potassium ATPase
- NO:
-
Nitric oxide
- PBS:
-
Phosphate-buffered saline
- PMRS:
-
Plasma membrane redox system
- ROS:
-
Reactive oxygen species
- RNS:
-
Reactive nitrogen species
- SH:
-
Sulphhydryl
- SOD:
-
Cu,Zn superoxide dismutase
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Alvarez-Llamas G, de la Cuesta F, Barderas MG, Zubiri I, Posada-Ayala M, Vivanco F (2013) Characterization of membrane and cytosolic proteins of erythrocytes. Methods Mol Biol 1000:71–80. https://doi.org/10.1007/978-1-62703-405-0_6
Ansari FA, Mahmood R (2016) Sodium nitrite enhances generation of reactive oxygen species that decrease antioxidant power and inhibit plasma membrane redox system of human erythrocytes. Cell Biol Int 40:887–894. https://doi.org/10.1002/cbin.10628
Avron M, Shavit N (1963) A sensitive and simple method for determination of ferrocyanide. Anal Biochem 6:549–554. https://doi.org/10.1016/0003-2697(63)901490
Barodka VM, Nagababu E, Mohanty JG, Nyhan D, Berkowitz DE, Rifkind JM, Strouse JJ (2014) New insights provided by a comparison of impaired deformability with erythrocyte oxidative stress for sickle cell disease. Blood Cells Mol Dis 52:230–235. https://doi.org/10.1016/j.bcmd.2013.10.004
Beckman JS, Koppenol WH (1996) Nitric oxide, superoxide, and peroxynitrite: the good, the bad and ugly. Am J Phys 271:C1427–C1435. https://doi.org/10.1152/ajpcell.1996.271.5.C1424
Benesch RE, Benesch R, Yung S (1973) Equations for the spectrophotometric analysis of hemoglobin mixtures. Anal Biochem 55:245–248. https://doi.org/10.1016/0003-2697(73)90309-6
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 239:70–76. https://doi.org/10.1006/abio.1996.0292
Bergmeyer HU, Grassl M, Walter HE (1983) In: Bergmeyer HU (ed) Methods of enzymatic analysis, volume II, 3rd edn. Verlag Chemie, Deerfield Beach (FL), pp 222–223
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–888
Boffetta P, Hashibe M (2006) Alcohol and cancer. Lancet Oncol 7:149–156. https://doi.org/10.1016/S1470-2045(06)70577-0
Bonting SL, Simon KA, Hawkins NM (1961) Studies on sodium-potassium-activated adenosine triphosphatase. I. Quantitative distribution in several tissues of the cat. Arch Biochem Biophys 95:416–423. https://doi.org/10.1016/0003-9861(61)90170-9
Bratosin D, Estaquier J, Petit F, Arnoult D, Quatannens B, Tissier JP, Slomianny C, Sartiaux C, Alonso C, Huart JJ, Montreuil J (2001) Programmed cell death in mature erythrocytes: a model for investigating death effector pathways operating in the absence of mitochondria. Cell Death Differ 8:1143–1156. https://doi.org/10.1038/sj.cdd.4400946
Braun KP, Pavlovich JG, Jones DR, Peterson CM (2006) Stable acetaldehyde adducts: structural characterization of acetaldehyde adducts of human hemoglobin N-terminal beta-globin chain peptides. Alcohol Clin Exp Res 21:40–43. https://doi.org/10.1111/j.1530-0277.1997.tb03726.x
Brovelli A, Suhail M, Pallavicini G, Sinigaglia F, Balduini C (2015) Self-digestion of human erythrocyte membranes. Role of adenosine triphosphate and glutathione. Biochem J 164:469–472. https://doi.org/10.1042/bj1640469
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310. https://doi.org/10.1016/S0076-6879(78)52032-6
Buthet LR, Maciel ME, Quintans LN, de Castro CR, Costantini MH, Fanelli SL, Castro JA, Castro GD (2013) Acetaldehyde content and oxidative stress in the deleterious effects of alcohol drinking on rat uterine horn. J Toxicol 2013:161496. https://doi.org/10.1155/2013/161496
Byrnes JR, Wolberg AS (2017) Red blood cells in thrombosis. Blood 6:72–75. https://doi.org/10.1182/blood-2017-03-745349
Carlberg I, Mannervik B (1985) Glutathione reductase. Methods Enzymol 113:484–490. https://doi.org/10.1016/S0076-6879(85)13062-4
Cazzola R, Rondanelli M, Russo-Volpe S, Ferrari E, Cestaro B (2004) Decreased membrane fluidity and altered susceptibility to peroxidation and lipid composition in overweight and obese female erythrocytes. J Lipid Res 45:1846–1851. https://doi.org/10.1194/jlr.m300509-jlr200
Çekiç SD, Kara N, Tütem E, Başkan KS, Apak R (2012) Protein-incorporated serum total antioxidant capacity measurement by a modified CUPRAC (cupric reducing antioxidant capacity). Methods Anal Lett 45:754–763. https://doi.org/10.1080/00032719.2011.653901
Comporti M, Signorini C, Leoncini S, Gardi C, Ciccoli L, Giardini A, Vecchio D, Arezzini B (2010) Ethanol-induced oxidative stress: basic knowledge. Genes Nutr 5:101–109. https://doi.org/10.1007/s12263-009-0159-9
De Grey ADNJ (2005) The plasma membrane redox system: a candidate source of aging-related oxidative stress. Age 27:129–138. https://doi.org/10.1007/s11357-005-1630-1
de Lima Santos H, Lopes ML, Maggio B, Ciancaglini P (2005) Na,K-ATPase reconstituted in liposomes: effects of lipid composition on hydrolytic activity and enzyme orientation. Colloids Surf B Biointerfaces 41:239–248. https://doi.org/10.1016/j.colsurfb.2004.12.013
Ellman GL, Courtney KD, Andres V, Feather-stone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Eriksson CJP (2001) The role of acetaldehyde in the actions of alcohol (Update 2000). Alcohol Clin Exp Res 25:15S–32S. https://doi.org/10.1111/j.1530-0277.2001.tb02369.x
Eriksson CJ, Fukunaga T (1993) Human blood acetaldehyde (update 1992). Alcohol Alcohol 2:9–25
Farag MR, Alagawany M (2018) Erythrocytes as a biological model for screening of xenobiotics toxicity. Chem Biol Interact 279:73–83. https://doi.org/10.1016/j.cbi.2017.11.007
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–120. https://doi.org/10.1016/S0076-6879(84)05015-1
Gaines KC, Salhany JM, Tuma DJ, Sorrell MF (1977) Reactions of acetaldehyde with human erythrocyte membrane proteins. FEBS Lett 75:115–119. https://doi.org/10.1016/0014-5793(77)80065-3
Gay C, Gebicki JM (2000) A critical evaluation of the effect of sorbitol on the ferric-xylenol orange hydroperoxide assay. Anal Biochem 284:217–220. https://doi.org/10.1006/abio.2000.4696
Gay C, Gebicki JM (2003) Measurement of protein and lipid hydroperoxides in biological systems by the ferric-xylenol orange method. Anal Biochem 315:29–35. https://doi.org/10.1016/S0003-2697(02)00606-1
Haorah J, Ramirez SH, Floreani N, Gorantla S, Morsey B, Persidsky Y (2008) Mechanism of alcohol-induced oxidative stress and neuronal injury. Free Radic Biol Med 45:1542–1550. https://doi.org/10.1016/j.freeradbiomed.2008.08.030
Harwood DG, Kalechstein A, Barker WW, Strauman S, George-Hyslop PS, Iglesias C, Loewenstein D, Duara R (2010) The effect of alcohol and tobacco consumption, and apolipoprotein E genotype, on the age of onset in Alzheimer’s disease. Int J Geriatr Psychiatry 25:511–518. https://doi.org/10.1002/gps.2372
Hernández-Muñoz R, Baraona E, Blacksberg I, Lieber CS (1989) Characterization of the increased binding of acetaldehyde to red blood cells in alcoholics. Alcohol Clin Exp Res 13:654–659. https://doi.org/10.1111/j.1530-0277.1989.tb00399.x
Hoffmann T, Meyer RJ, Sorrell MF, Tuma DJ (1993) Reaction of acetaldehyde with proteins: formation of stable fluorescent adducts. Alcohol Clin Exp Res 17(1):69–74
Homann N, Jousimies-Somer H, Jokelainen K, Heine R, Salaspuro M (1997) High acetaldehyde levels in saliva after ethanol consumption: methodological aspects and pathogenetic implications. Carcinogenesis 18:1739–1743
Huang JC, Li DJ, Diao JC, Hou J, Yuan JL, Zou GL (2007) A novel fluorescent method for determination of peroxynitrite using folic acid as a probe. Talanta 72:1283–1287. https://doi.org/10.1016/j.talanta.2007.01.033
Keller A, Mohamed A, Dröse S, Brandt U, Fleming I, Brandes RP (2004) Analysis of dichlorodihydrofluorescein and dihydrocalcein as probes for the detection of intracellular reactive oxygen species. Free Radic Res 38:1257–1267. https://doi.org/10.1080/10715760400022145
Kennett EC, Kuchel PW (2003) Redox reactions and electron transfer across the red cell membrane. IUBMB Life 55:375–385. https://doi.org/10.1080/15216540310001592843
Khundmiri SJ, Asghar M, Khan F, Salim S, Yusufi ANK (2004) Effect of ischemia and reperfusion on enzymes of carbohydrate metabolism in rat kidney. J Nephrol 17:377–383
Kirkman HN, Gaetani GF (2007) Mammalian catalase: a venerable enzyme with new mysteries. Trends Biochem Sci 32:44–50. https://doi.org/10.1016/j.tibs.2006.11.003
Kuma F, Ishizawa S, Hirayama K, Nakajima H (1972) Studies on methemoglobin reductase. I Comparative studies of diaphorases from normal and methemoglobinemic erythrocytes. J Biol Chem 247:550–555
Leaz JKF, Adjobo-Hermans MJW, Brock R, Bosman GJCGM (2017) Acetylcholinesterase provides new insights into red blood cell ageing in vivo and in vitro. Blood Transfus 15:232–238. https://doi.org/10.2450/2017.0370-16
Lee WS, Lee JY, Sung WY, Seo SW (2014) Naproxen-induced methemoglobinemia in an alcohol-dependent patient. Am J Emerg Med 32:1493–14e9. https://doi.org/10.1016/j.ajem.2014.04.027
Levine RL, Garland D, Oliver CN, Amici A, Climent I, Lenz AG, Ahn BW, Shaltiel S, Stadtman ER (1990) Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol 186:464–478. https://doi.org/10.1016/0076-6879(90)86141-H
Linderborg K, Salaspuro M, Väkeväinen S (2011) A single sip of a strong alcoholic beverage causes exposure to carcinogenic concentrations of acetaldehyde in the oral cavity. Food Chem Toxicol 49:2103–2106
Lindros KO (1983) Human blood acetaldehyde levels: with improved methods, a clearer picture emerges. Alcohol Clin Exp Res 7:70–75
Maheshwari N, Khan FH, Mahmood R (2017) Sodium meta-arsenite induced reactive oxygen species in human red blood cells: impaired antioxidant and membrane redox systems, hemoglobin oxidation, and morphological changes. Free Radic Res 51:483–497. https://doi.org/10.1080/10715762.2017.1327714
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Maurya PK, Kumar P, Chandra P (2016) Age-dependent detection of erythrocytes glucose-6-phosphate dehydrogenase and its correlation with oxidative stress. Arch Physiol Biochem 122:61–66. https://doi.org/10.3109/13813455.2015.1136648
May JM, Qu ZC, Cobb CE (2004) Human erythrocyte recycling of ascorbic acid: relative contributions from the ascorbate free radical and dehydroascorbic acid. J Biol Chem 279:14975–14982. https://doi.org/10.1074/jbc.M312548200
Miranda KM, Espey MG, Wink DA (2001) A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide 5:62–71. https://doi.org/10.1006/niox.2000.0319
Mishra K, Ojha H, Chaudhury NK (2012) Estimation of antiradical properties of antioxidants using DPPH-assay: a critical review and results. Food Chem 247:6960–6962. https://doi.org/10.1016/j.foodchem.2011.07.127
Mohanty JG, Nagababu E, Rifkind JM (2014) Red blood cell oxidative stress impairs oxygen delivery and induces red blood cell aging. Front Physiol 5:84. https://doi.org/10.3389/fphys.2014.00084
Nagababu E, Gulyani S, Earley CJ, Cutler R, Mattson M, Rifkind J (2008) Iron-deficiency anaemia enhances red blood cell oxidative stress. Free Radic Res 42:824–829. https://doi.org/10.1080/10715760802459879
Nieminen M, Salaspuro M (2018) Local acetaldehyde-an essential role in alcohol-related upper gastrointestinal tract carcinogenesis. Cancers 10:11. https://doi.org/10.3390/cancers10010011
Novitskiy G, Traore K, Wang L, Trush MA, Mezey E (2006) Effects of ethanol and acetaldehyde on reactive oxygen species production in rat hepatic stellate cells. Alcohol Clin Exp Res 30:1429–1435. https://doi.org/10.1111/j.1530-0277.2006.00171.x
Pastorino R, Iuliano L, Vecchioni A, Arzani D, Milic M, Annunziata F, Zerbinati C, Capoluongo E, Bonassi S, McKay JD, Boccia S (2018) Effect of alcohol dehydrogenase-1B and −7 polymorphisms on blood ethanol and acetaldehyde concentrations in healthy subjects with a history of moderate alcohol consumption. Drug Test Anal 10:488–495. https://doi.org/10.1002/dta.2251
Peters AL, Van Noorden CJF (2009) Glucose-6-phosphate dehydrogenase deficiency and malaria: Cytochemical detection of heterozygous G6PD deficiency in women. J Histochem Cytochem 57:1003–1011. https://doi.org/10.1369/jhc.2009.953828
Peters AL, Van Bruggen R, De Korte D, Van Noorden CJF, Vlaar APJ (2016) Glucose-6-phosphate dehydrogenase activity decreases during storage of leukoreduced red blood cells. Transfusion 56:427–432. https://doi.org/10.1111/trf.13378
Peterson CM, Nguyen LB (1985) Clinical implications of acetaldehyde adducts with hemoglobin. Prog Clin Biol Res 183:19–30
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269:337–341. https://doi.org/10.1006/abio.1999.4019
Qasim N, Mahmood R (2015) Diminution of oxidative damage to human erythrocytes and lymphocytes by creatine: possible role of creatine in blood. PLoS One 10(11):e0141975. https://doi.org/10.1371/journal.pone.0141975
Radi R (2013) Peroxynitrite, a stealthy biological oxidant. J Biol Chem 288:26464–26472. https://doi.org/10.1074/jbc.R113.472936
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic Biol Med 26:1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Reshamwala SMS, Patil ND (2005) Biochemical changes in erythrocyte membrane in uncontrolled type 2 diabetes mellitus. Indian J Biochem Biophys 42:250–253
Rizvi SI, Pandey KB (2010) Activation of the erythrocyte plasma membrane redox system by resveratrol: a possible mechanism for antioxidant properties. Pharmacol Reports 62:726–732. https://doi.org/10.1016/S1734-1140(10)70330-3
Rizvi SI, Kumar D, Chakravarti S, Singh P (2011) Erythrocyte plasma membrane redox system may determine maximum life span. Med Hypotheses 76:547–549. https://doi.org/10.1016/j.mehy.2010.12.014
Rocco A, Compare D, Angrisani D, Sanduzzi Zamparelli M, Nardone G (2014) Alcoholic disease: liver and beyond. World J Gastroenterol 20:14652–14659. https://doi.org/10.3748/wjg.v20.i40.14652
Schallreuter KU, Elwary SM, Gibbons NC, Rokos H, Wood JM (2004) Activation/deactivation of acetylcholinesterase by H2O2: more evidence for oxidative stress in vitiligo. Biochem Biophys Res Commun 315:502–508
Secretan B, Straif K, Baan R, Grosse Y, El Ghissassi F, Bouvard V et al (2009) A review of human carcinogens—part E: tobacco, areca nut, alcohol, coal smoke, and salted fish. Lancet Oncol 10:1033–1034
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205
Seeman JI, Dixon M, Haussmann HJ (2002) Acetaldehyde in mainstream tobacco smoke: formation and occurrence in smoke and bioavailability in the smoker. Chem Res Toxicol 11:1331–1350. https://doi.org/10.1021/tx020069f
Setshedi M, Wands JR, de la Monte SM (2010) Acetaldehyde adducts in alcoholic liver disease. Oxidative Med Cell Longev 3:177–185. https://doi.org/10.4161/oxim.3.3.12288
Sheetz MP, Singer SJ (1974) Biological membranes as bilayer couples. A molecular mechanism of drug-erythrocyte interactions. Proc Natl Acad Sci U S A 71:4457–4461. https://doi.org/10.1073/pnas.71.11.4457
Shonk CE, Boxer GE (1964) Enzyme patterns in human tissues. I. Methods for the determination of glycolytic enzymes. Cancer Res 24:709–721
Snyder SL, Sobocinski PZ (1975) An improved 2,4,6-trinitrobenzenesulfonic acid method for the determination of amines. Anal Biochem 64:284–288. https://doi.org/10.1016/0003-2697(75)90431-5
Solomon LR (1988) Effects of acetaldehyde on human red cell metabolism: evidence for the formation of enzyme inhibitors. Clin Chim Acta 175:249–265. https://doi.org/10.1016/0009-8981(88)90101-5
Tamura T, Stadtman TC (1996) A new selenoprotein from human lung adenocarcinoma cells: purification, properties, and thioredoxin reductase activity. Proc Natl Acad Sci U S A 93:1006–1011. https://doi.org/10.1073/pnas.93.3.1006
Tamura M, Ito H, Matsui H, Hyodo I (2014) Acetaldehyde is an oxidative stressor for gastric epithelial cells. J Clin Biochem Nutr 55:26–31. https://doi.org/10.3164/jcbn.14-12
Taussky HH, Shorr E (1953) A microcolorimetric method for the determination of inorganic phosphorus. J Biol Chem 202:675–685
Tyulina OV, Prokopieva VD, Boldyrev AA, Johnson P (2006) Erythrocyte and plasma protein modification in alcoholism: a possible role of acetaldehyde. Biochim Biophys Acta - Mol Basis Dis 1762:558–563. https://doi.org/10.1016/j.bbadis.2006.03.003
Umbreit J (2007) Methemoglobin—it’s not just blue: a concise review. Am J Hematol 82:134–144. https://doi.org/10.1002/ajh.20738
Unt E, Kairane C, Vaher I, Zilmer M (2008) Red blood cell and whole blood glutathione redox status in endurance-trained men following a ski marathon. J Sport Sci Med 7:344–349
Vajreswari A, Rupalatha M, Rao PS (2002) Effect of altered dietary n-6-to-n-3 fatty acid ratio on erythrocyte lipid composition and membrane-bound enzymes. J Nutr Sci Vitaminol 48:365–370
Wang Y, Liu M, Li F, Peng XM, Zhang K, Peng QY, Liao ZG (2009) Scanning electron microscopic observation of erythrocytes and endothelial cells of electrified death rabbits. Leg Med Tokyo Jpn 11:S244–S247. https://doi.org/10.1016/j.legalmed.2009.01.097
Waris S, Habib S, Tantry IQ, Khan RH, Mahmood R, Ali A (2018) Acetaldehyde-induced structural and conformational alterations in human immunoglobulin G: a physicochemical and multi-spectroscopic study. Int J Biol Macromol 113:701–710. https://doi.org/10.1016/j.ijbiomac.2018.03.013
Wojtala A, Bonora M, Malinska D, Pinton P, Duszynski J, Wieckowski MR (2014) Methods to monitor ROS production by fluorescence microscopy and fluorometry. Methods Enzymol 542:243–262. https://doi.org/10.1016/B978-0-12-416618-9.00013-3
Yan T, Zhao Y (2020) Acetaldehyde induces phosphorylation of dynamin-related protein 1 and mitochondrial dysfunction via elevating intracellular ROS and Ca2+ levels. Redox Biol 28:101381. https://doi.org/10.1016/j.redox.2019.101381
Yan T, Zhao Y, Zhang X (2016) Acetaldehyde induces cytotoxicity of SH-SY5Y cells via inhibition of Akt activation and induction of oxidative stress. Oxidative Med Cell Longev 2016:4512309. https://doi.org/10.1155/2016/4512309
Yen GC, Chen HY (1995) Antioxidant activity of various tea extracts in relation to their antimutagenicity. J Agric Food Chem 43:27–32. https://doi.org/10.1021/jf00049a007
Zhang B, Zhang J, Peng S, Liu R, Li X, Hou Y, Han X, Fang J (2017) Thioredoxin reductase inhibitors: a patent review. Expert Opin Ther Pat 27:547–556. https://doi.org/10.1080/13543776.2017.1272576
Zakhari S (2013) Alcohol metabolism and epigenetic changes. Alcohol Res 35:6–16.
Acknowledgements
Infrastructural facility of the department as well as University Sophisticated Instrumentation Facility (USIF) is acknowledged. The authors are also thankful to the University Grants Commission for the award of Maulana Azad National Fellowship to SW.
Funding
The study is financially supported by the department from DST-FIST-II, UGC-SAP-DRS-III and DST-PURSE.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Mohamed Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Waris, S., Patel, A., Ali, A. et al. Acetaldehyde-induced oxidative modifications and morphological changes in isolated human erythrocytes: an in vitro study. Environ Sci Pollut Res 27, 16268–16281 (2020). https://doi.org/10.1007/s11356-020-08044-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-08044-4