Abstract
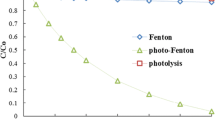
Doxorubicin (DOX) is a chemotherapeutic agent from anthracycline class, which acts unselectively on all cells; thus, it may have genotoxic and/or mutagenic effects and cause serious environmental problems. Herein, the decomposition of a diluted solution of DOX hydrochloride for injection has been investigated under photo-oxidative conditions, in ambient light and without pH modification, using hydrogen peroxide as oxidizing agent and hydrophobic siloxane-based metal-organic frameworks (MOFs) as heterogeneous catalysts. The kinetics of the photodegradation process was followed by UV-Vis spectroscopy and by ESI-MS. According to UV-Vis data, two pseudo-first-order kinetic steps describe the process, with rate constants in the order of 10−3–10−2 min−1 for the rate-determining one. ESI-MS provided more accurate information, with a rate constant of 2.6 · 10−2 min−1 calculated from the variation of DOX ion abundance. Complete decomposition of DOX was achieved after 120 min in the shade and after only 20 min by exposure to sunlight. The analysis of the residual waters by mass spectrometry and 1D and 2D NMR spectroscopy showed complete disappearance of DOX in all cases, excluded any anthracycline species, which are destroyed in the tested conditions, and proved formation of an un-harmful compound—glycerol, while no trace of metal was detected by XRF. Preliminary data also showed decomposition of oxytetracycline in similar conditions. By this study, we bring into attention a less-addressed pollution issue and we propose a mild and effective method for the removal of drug emerging pollutants.








Similar content being viewed by others
References
Abraham SA, Waterhouse DN, Mayer LD, Cullis PR, Madden TD, Bally MB (2005) The liposomal formulation of doxorubicin. Methods Enzymol 391:71–97. https://doi.org/10.1016/S0076-6879(05)91004-5
Beijnen JH, van der Houwen OAGJ, Underberg WJM (1986) Aspects of the degradation kinetics of doxorubicin in aqueous solution. Int J Pharm 32:123–131. https://doi.org/10.1016/0378-5173(86)90170-5
Bokare AD, Choi W (2014) Review of iron-free Fenton-like systems for activating H2O2 in advanced oxidation processes. J Hazard Mater 275:121–135. https://doi.org/10.1016/j.jhazmat.2014.04.054
Chartier Y, Emmanuel J, Pieper U, Pruss A, Rushbrook P, Stringer R, Townend W, Wilbum S., Zghondi R (Eds.) (2014) Safe management of wastes from health-care activities. Second ed. (World Health Organization)
Cho H, Lai TC, Tomoda K, Kwon GS (2015) Polymeric micelles for multi-drug delivery in cancer. AAPS PharmSciTech 16:10–20. https://doi.org/10.1208/s12249-014-0251-3
Chudasama V, Maruani A, Caddick S (2016) Recent advances in the construction of antibody–drug conjugates. Nat Chem 8:114–119. https://doi.org/10.1038/nchem.2415
Cvrčková O, Ciganek M (2005) Photostability of polycyclic aromatic hydrocarbons (PAHs) and nitrated polycyclic aromatic hydrocarbons (NPAHs) in dichloromethane and isooctane solutions. Polycycl Aromat Compd 25:141–156. https://doi.org/10.1080/10406630590922166
Das A, Adhikari C, Nayak D, Chakraborty A (2016) First evidence of the liposome-mediated deintercalation of anticancer drug doxorubicin from the drug−DNA complex: a spectroscopic approach. Langmuir 32:159–170. https://doi.org/10.1021/acs.langmuir.5b03702
Dhakshinamoorthy A, Li Z, Garcia H (2018) Catalysis and photocatalysis by metal organic frameworks. Chem Soc Rev 47:8134–8172. https://doi.org/10.1039/c8cs00256h
Dong DW, Tong SW, Qi XR (2013) Comparative studies of polyethylenimine–doxorubicin conjugates with pH-sensitive and pH-insensitive linkers. J Biomed Mater Res A 101A:1336–1344. https://doi.org/10.1002/jbm.a.34450
Dumitru R, Ianculescu A, Păcurariu C, Lupa L, Pop A, Vasile B, Surdu A, Manea F (2019) BiFeO3 - synthesis, characterization and its photocatalytic activity towards doxorubicin degradation from water. Ceram Int 45:2789–2802. https://doi.org/10.1016/j.ceramint.2018.07.298
González B, Trujillano R, Vicente MA, Rives V, Korili SA, Gil A (2019) Photocatalytic degradation of trimethoprim on doped Ti-pillared montmorillonite. Appl Clay Sci 167:43–49. https://doi.org/10.1016/j.clay.2018.10.006
Hansel S, Castegnaro M, Sportouch MH, De Méo M, Milhavet JC, Laget M, Duménil G (1997) Chemical degradation of wastes of antineoplastic agents: cyclophosphamide, ifosfamide and melphalan. Int Arch Occup Environ Health 69:109–114. https://doi.org/10.1007/s004200050124
Kadu BS, Wani KD, Kaul-Ghanekar R, Chikate RC (2017) Degradation of doxorubicin to non-toxic metabolites using Fe-Ni bimetallic nanoparticles. Chem Eng J 325:715–724. https://doi.org/10.1016/j.cej.2017.05.097
Kaushik D, Bansal G (2015) Four new degradation products of doxorubicin: an application of forced degradation study and hyphenated chromatographic techniques. J Pharma Anal 5:285–295. https://doi.org/10.1016/j.jpha.2015.05.003
Khosravi A, Mokhtari J, Naimi-Jamal MR, Tahmasebi S, Panahi L (2017) Cu2(BDC)2(BPY)–MOF: an efficient and reusable heterogeneous catalyst for the aerobic Chan–Lam coupling prepared via ball-milling strategy. RSC Adv 7:46022–46027. https://doi.org/10.1039/C7RA09772G
Kosjek T, Heath E (2011) Occurrence, fate and determination of cytostatic pharmaceuticals in the environment. Trends Anal Chem 30:1065–1087. https://doi.org/10.1016/j.trac.2011.04.007
Larkin P (2011) Infrared and Raman spectroscopy. Principles and spectral interpretation. Elsevier, Amsterdam
Le Bot MA, Riche C, Guedes Y, Kernaleguen D, Simon S, Begue JM, Berthou F (1987) Study of doxorubicin photodegradation in plasma, urine and cell culture medium by HPLC. Biomed Chromatogr 2:242–244. https://doi.org/10.1002/bmc.1130020603
Le HTN, Tran TV, Phan NTS, Truong T (2015) Efficient and recyclable Cu2(BDC)2(BPY)-catalyzed oxidative amidation of terminal alkynes: role of bipyridine ligand. Catal Sci Technol 5:851–859. https://doi.org/10.1039/C4CY01074D
Lee SG, Tkaczuk M, Jankewicz G, Ambados F (2007) Surface contamination from cytotoxic chemotherapy following preparation and administration. J Pharm Pract Res 37:271–276. https://doi.org/10.1002/j.2055-2335.2007.tb00763.x
Matsumoto K, Naito T, Hori K, Suzuki N, Miyamoto Y, Takashina Y, Ohnishi K, Kawakami J (2010) Surveillance of workplace contamination and occupational exposure to antineoplastic agents in a hospital setting: establishment of a monitoring method using doxorubicin. Yakugaku Zasshi J 130:431–439
Ming Y, Purewal J, Yang J, Xu C, Soltis R, Warner J, Veenstra M, Gaab M, Müller U, Siegel DJ (2015) Kinetic Stability of MOF-5 in Humid Environments: Impact of Powder Densification, Humidity Level, andExposure Time. Langmuir 31:4988–4995. https://doi.org/10.1021/acs.langmuir.5b00833
Mordente A, Meucci E, Silvestrini A, Martorana GE, Giardina B (2009) New developments in anthracycline-induced cardiotoxicity. Curr Med Chem 16:1656–1672. https://doi.org/10.2174/092986709788186228
Nawara K, Krysinski P, Blanchard GJ (2012) Photoinduced reactivity of doxorubicin: catalysis and degradation. J Phys Chem A 116:4330–4337. https://doi.org/10.1021/jp303218r
Niu S, Zhang R, Zhou X, Zhao X, Suo H, Jiao Y, Yao H, Guo C (2018) The enhanced photocatalytic activity of Yb3+-Ho3+/Er3+ co-doped 3D BiOCl flower. Dyes and Pigments 149:462–469. https://doi.org/10.1016/j.dyepig.2017.10.026
Phan NTS, Nguyen TT, Vu PHL (2013) A copper metal–organic framework as an efficient and recyclable catalyst for the oxidative cross-dehydrogenative coupling of phenols and formamides. ChemCatChem 5:3068–3077. https://doi.org/10.1002/cctc.201300400
Pi Y, Li X, Xia Q, Wu J, Li Y, Xiao J, Li Z (2018) Adsorptive and photocatalytic removal of persistent organic pollutants (POPs) in water by metal-organic frameworks (MOFs). Chem Eng J 337:351–371. https://doi.org/10.1016/j.cej.2017.12.092
Piorecka K, Stanczyk W, Florczak M (2017) NMR analysis of antitumor drugs: doxorubicin, daunorubicin and their functionalized derivatives. Tetrahedron Lett 58:152–155. https://doi.org/10.1016/j.tetlet.2016.11.118
Racles C, Shova S, Cazacu M, Timpu D (2013) New highly ordered hydrophobic siloxane-based coordination polymers. Polymer 54:6096–6104. https://doi.org/10.1016/j.polymer.2013.09.001
Racles C, Zaltariov MF, Iacob M, Silion M, Avadanei M, Bargan A (2017) Siloxane-based metal–organic frameworks with remarkable catalytic activity in mild environmental photodegradation of azo dyes. Appl Catal B 205:78–92. https://doi.org/10.1016/j.apcatb.2016.12.034
Rafiyath SM, Rasul M, Lee B, Wei G, Lamba G, Liu D (2012) Comparison of safety and toxicity of liposomal doxorubicin vs. conventional anthracyclines: a meta-analysis. Exp Hematol Oncol 1:10 Available at http://www.ehoonline.org/content/1/1/10 [verified 27 December 2018]
Ramphal R, Bains T, Goulet G, Vaillancourt R (2015) Occupational exposure to chemotherapy of pharmacy personnel at a single centre. Can J Hosp Pharm 68:104–112
Ramu A, Mehta MM, Liu J, Turyan I, Aleksic A (2000) The riboflavin-mediated photooxidation of doxorubicin. Cancer Chemother Pharmacol 46:449–458. https://doi.org/10.1007/s002800000174
Raniolo S, Vindigni G, Ottaviani A, Unida V, Iacovelli F, Manetto A, Figini M, Stella L, Desideri A, Biocca S (2018) Selective targeting and degradation of doxorubicin-loaded folate-functionalized DNA nanocages. Nanomedicine 14:1181–1190. https://doi.org/10.1016/j.nano.2018.02.002
Revia RA, Zhang M (2016) Magnetite nanoparticles for cancer diagnosis, treatment, and treatment monitoring: recent advances. Mater Today 19:157–168. https://doi.org/10.1016/j.mattod.2015.08.022
Şentürk N, Top A (2018) PEG-peptide conjugate containing cathepsin B degradation unit as a doxorubicin carrier system. Turk J Chem 42:385–400. https://doi.org/10.3906/kim-1706-65
Somensi CA, Simionatto EL, Dalmarco JB, Gaspareto P, Radetski CM (2012) A comparison between ozonolysis and sonolysis/ozonolysis treatments for the degradation of the cytostatic drugs methotrexate and doxorubicin: kinetic and efficiency approaches. J Environ Sci Health A 47:1543–1550. https://doi.org/10.1080/10934529.2012.680414
Sun C, Li Y, Song P, Ma F (2016) An experimental and theoretical investigation of the electronic structures and photoelectrical properties of ethyl red and carminic acid for DSSC application. Materials 9(22 pp):813. https://doi.org/10.3390/ma9100813
Sun K, Li L, Yu XL, Liu L, Meng Q, Wang F, Zhang R (2017) Functionalization of mixed ligand metal-organic frameworks as the transport vehicles for drugs. J Colloid Interface Sci 486:128–135. https://doi.org/10.1016/j.jcis.2016.09.068
Tacar O, Sriamornsak P, Dass CR (2013) Doxorubicin: an update on anticancer molecular action, toxicity and novel drug delivery systems. J Pharm Pharmacol 65:157–170. https://doi.org/10.1111/j.2042-7158.2012.01567.x
Tavoloni N, Guarino AM, Berk PD (1980) Photolytic degradation of Adriamycin. J Pharm Pharmacol 32:860–862. https://doi.org/10.1111/j.2042-7158.1980.tb13094.x
Thongrasmee W, Tohsing K, Janjai S (2018) A model for calculating daily near infrared solar radiation. J Phys Conf Ser 1144:012040. https://doi.org/10.1088/1742-6596/1144/1/012040
Vasconcelos IB, da Silva TG, Militão GCG, Soares TA, Rodrigues NM, Rodrigues MO, da Costa Jr. NB, Freiree RO, Junior SA (2012) Cytotoxicity and slow release of the anti-cancer drug doxorubicin from ZIF-8. RSC Adv 2:9437–9442. https://doi.org/10.1039/C2RA21087H
Wang CC, Li JR, Lv XL, Zhang YQ, Guo G (2014) Photocatalytic organic pollutants degradation in metal–organic frameworks. Energy Environ Sci 7:2831–2867. https://doi.org/10.1039/C4EE01299B
Wood MJ, Irwin WJ, Scott DK (1990) Photodegradation of doxorubicin, daunorubicin and epirubicin measured by high performance liquid chromatography. J Clin Pharm Ther 15:291–300. https://doi.org/10.1111/j.1365-2710.1990.tb00387.x
World Health Organization (2018) Cancer fact sheet, Available at http://www.who.int/mediacentre/factsheets/fs297/en/
Wu JCS, Chen CH (2004) A visible-light response vanadium-doped titania nanocatalyst by sol–gel method. J Photochem Photobiol A Chem 163:509–515. https://doi.org/10.1016/j.jphotochem.2004.02.007
Yang Y, Yin LC, Gong Y, Niu P, Wang JQ, Gu L, Chen X, Liu G, Wang L, Cheng HM (2018) An unusual strong visible-light absorption band in red anatase TiO2 photocatalyst induced by atomic hydrogen-occupied oxygen vacancies. Adv Mater 1704479(8pp). https://doi.org/10.1002/adma.201704479
Zhang KL, Liu CM, Huang FQ, Zheng C, Wang WD (2006) Study of the electronic structure and photocatalytic activity of the BiOCl photocatalyst. Appl Catal B-Environ 68:125–129. https://doi.org/10.1016/j.apcatb.2006.08.002
Zhang H, Jiang W, Liu R, Zhang J, Zhang D, Li Z, Luan Y (2017) Rational design of MOF nanocarrier-based co-delivery system of doxorubicin hydrochloride/verapamil hydrochloride for overcoming multidrug resistance with efficient targeted cancer therapy. ACS Appl Mater Interfaces 9:19687–19697. https://doi.org/10.1021/acsami.7b05142
Ming Y, Purewal J, Yang J, Xu C, Soltis R, Warner J, Veenstra M, Gaab M, Müller U, Siegel DJ (2015) Kinetic stability of MOF-5 in humid environments: impact of powder densification, humidity level, and exposure time. Langmuir 31 (17):4988-4995
Funding
This work was supported by a grant of Romanian Ministry of Research and Innovation, CNCS - UEFISCDI, project number PN-III-P4-ID-PCCF-2016-0050 (5D-nanoP), within PNCDI III.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible editor: Suresh Pillai
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 3143 kb)
Rights and permissions
About this article
Cite this article
Racles, C., Zaltariov, MF., Silion, M. et al. Photo-oxidative degradation of doxorubicin with siloxane MOFs by exposure to daylight. Environ Sci Pollut Res 26, 19684–19696 (2019). https://doi.org/10.1007/s11356-019-05288-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-019-05288-7