Abstract
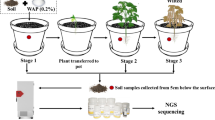
Phosphate solubilizing bacteria (PSB) can convert insoluble forms of phosphorus (P) to accessible forms. 11 strains of PSB, including five inorganic phosphate solubilizing bacteria (IPSBs) and six organic phosphate solubilizing bacteria (OPSBs), were isolated from rhizosphere soils of three plants Scirpus planiculmis, Zizania latifolia, and Phrnagmites australis in the Yeyahu Wetland of Beijing, China to investigate P-solubilizing activities. In addition, the distributions of P fractions in soil samples were also observed. All strains evaluated above 1.0 by the ratio of transparent circle diameter to colony diameter (D/d) on Ca3(PO4)2 or lecithin plates were identified by 16S rRNA sequencing. Results showed that Ca-bound P (Ca-P) was the main species of inorganic P (IP), and highly resistant organic P (HR-OP) accounted for the most part of organic P (OP). These strains were identified as bacterial species of Enterobacter asburiae, Acinetobacter sp., Bacillus cereus strain, and so on. The most efficient IPSB strain could convert over 430 mg L−1 orthophosphate, while the equivalent OPSB strain only liberated less than 4 mg L−1 in liquid culture, which indicated that IPSBs have a better P-solubilizing ability than OPSBs in rhizosphere soils of the Yeyahu Wetland and IPSBs are likely to regulate the P transformation process in this wetland.

ᅟ








Similar content being viewed by others
References
Abdol AY, Kazem K, Abdol AM, Farhad R, Habib AN (2011) Phosphate solubilizing bacteria and arbuscular mycorrhizal fungi impacts on inorganic phosphorus fractions and wheat growth. World Appl Sci J 15:1310–1318
Bai FX, Chen CL, An JY,Xiao SY,Deng XX,Pan ZY, (2014) Transcriptome responses to phosphate deficiency in Poncirus trifoliata (L.) Raf. Acta Physiologiae Plantarum 36 (12):3207-3215. https://doi.org/10.1007/s11738-014-1687-5
Berendsen RL, Pieterse CM, Bakker PA (2012) The rhizosphere microbiome and plant health. Trends Plant Sci 17:478–486. https://doi.org/10.1016/j.tplants.2012.04.001
Bolan CD, Cecco SA, Yau YY, Wesley RA, Oblitas JM, Rehak NN, Leitman SE (2003) Randomized placebo-controlled study of oral calcium carbonate supplementation in plateletpheresis: II. Metabolic effects. Transfusion 43:1414–1422. https://doi.org/10.1046/j.1537-2995.2003.00513.x
Brookes PC, Powlson DS, Jenkinson DS (1982) Measurement of microbial biomass phosphorus in soil. Soil Biol Biochem 14:319–329. https://doi.org/10.1016/0038-0717(82)90001-3
Buch A, Archana G, Naresh KG (2008) Metabolic channeling of glucose towards gluconate in phosphate-solubilizing Pseudomonas aeruginosa P4 under phosphorus deficiency. Res Microbiol 159:635–642. https://doi.org/10.1016/j.resmic.2008.09.012
Chen YP, Rekha PD, Arun AB, Shen FT, Lai WA, Yong CC (2006) Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. 34, 33-41. https://doi.org/10.1016/j.apsoil.2005.12002;
Collavino MM, Sansberro PA, Mroginski LA, Aguilar OM (2010) Comparison of in vitro solubilization activity of diverse phosphate-solubilizing bacteria native to acid soil and their ability to promote Phaseolus vulgaris growth. Biol Fertil Soils 46:727–738. https://doi.org/10.1007/s00374-010-0480-x
da Costa EM, de Lima W, Oliveira-Longatti SM, de Souza FM (2015) Phosphate-solubilising bacteria enhance Oryza sativa growth and nutrient accumulation in an oxisol fertilized with rock phosphate. Ecol Eng 83:380–385. https://doi.org/10.1016/j.ecoleng.2015.06.045
Dharni S, Srivastava AK, Samad A, Patra DD (2014) Impact of plant growth promoting Pseudomonas monteilii PsF84 and Pseudomonas plecoglossicida PsF610 on metal uptake and production of secondary metabolite (monoterpenes) by rose-scented geranium (Pelargonium graveolens cv. bourbon) grown on tannery sludge amended soil. Chemosphere 117:433–439. https://doi.org/10.1016/j.chemosphere.2014.08.001
Dinh MV, Schramm T, Spohn M, Matzner E (2016) Drying-rewetting cycles release phosphorus from forest soils. J Plant Nutr Soil Sci 179:670–678. https://doi.org/10.1002/jpln.201500577
Elaine Martins da Costa, Wellington de Lima, Silvia M. Oliveira-Longatti, Fatima M. de Souza, (2015) Phosphate-solubilising bacteria enhance Oryza sativa growth and nutrient accumulation in an oxisol fertilized with rock phosphate. Ecological Engineering 83:380-385http://dx.doi.org/10.1016/j.ecoleng.2015.06.045.
Elizabeth P, Miguel S, Ball MM, Yarza’ bal LA (2007) Isolation and characterization of mineral phosphate-solubilizing bacteria naturally colonizing a limonitic crust in the south-eastern Venezuelan region. Soil Biol Biochem 39:2905–2914. https://doi.org/10.1016/j.soilbio.2007.06.017
Glick BR (2012) Plant growth-promoting bacteria: mechanisms and applications. Scientifica (Cairo). https://doi.org/10.6064/2012/963401
Gaur R,Tiwari S, (2015) Isolation, production, purification and characterization of an organic-solvent-thermostable alkalophilic cellulase from Bacillus vallismortis RG-07. BMC Biotechnology.
Gordon H,Haygarth PM,Bardgett RD (2008) Drying and rewetting effects on soil microbial community composition and nutrient leaching.Soil Biology & Biochemistry 40:302-311. https://doi.org/10.1016/j.soilbio.2007.08.008.
Guo SL, Dang TH, Hao MD (2008) Phosphorus changes and sorption characteristics in a calcareous soil under long-term fertilization. Pedosphere 18:248–256. https://doi.org/10.1016/S1002-0160(08)60014-4
Gupta P, Kumar V (2017) Value added phytoremediation of metal stressed soils using phosphate solubilizing microbial consortium. World J Microbiol Biotechnol 33:9. https://doi.org/10.1007/s11274-016-2176-3
Gupta M, Kiran S, Gulati A, Singh B, Tewari R (2012) Isolation and identification of phosphate solubilizing bacteria able to enhance the growth and aloin-A biosynthesis of Aloe barbadensis Miller. Microbiol Res 167:358–363. https://doi.org/10.1016/j.micres.2012.02.004
Istina IN, Widiastuti H, Joy B, Antralina M (2015) Phosphate-solubilizing microbe from saprists peat soil and their potency to enhance oil palm growth and P uptake. Procedia Food Sci 3:426–435. https://doi.org/10.1016/j.profoo.2015.01.047
Ivanoff DB, Reddy KR, Robinson S (1998) Chemical fractionation of organic phosphorus in selected Histosols. Soil Sci. 163, 36–45.
Jorquera MA, Hernandez MT, Rengel Z, Marschner P, Mora MD (2008) Isolation of culturable phosphobacteria with both phytate-mineralization and phosphate-solubilization activity from the rhizosphere of plants grown in a volcanic soil. Biol Fertil Soils 44:1025–1034. https://doi.org/10.1007/s00374-008-0288-0
Karagöz K, Ateş F, Karagöz H, Kotan R, Çakmakçı R (2012) Characterization of plant growth-promoting traits of bacteria isolated from the rhizosphere of grapevine grown in alkaline and acidic soils. Eur J Soil Biol 50:144–150. https://doi.org/10.1016/j.ejsobi.2012.01.007
Li M, Zhang J, Wang GQ, Yang HJ, Whelan MJ, White SM (2013) Organic phosphorus fractionation in wetland soil profiles by chemical extraction and phosphorus-31 nuclear magnetic resonance spectroscopy. Appl Geochem 33:213–221. https://doi.org/10.1016/j.apgeochem.2013.02.014
Li YY, Yang R, Gao R, Wei HA, Chen AL, Li Y (2015) Effects of long-term phosphorus fertilization and straw incorporation on phosphorus fractions in subtropical paddy soil. J Integr Agric 14:365–373. https://doi.org/10.1016/S2095-3119(13)60684-X
Liu FP, Liu HQ, Zhou HL, Dong ZG, Bai XH, Bai P, Qiao JJ (2014) Isolation and characterization of phosphate-solubilizing bacteria from betel nut (Areca catechu) and their effects on plant growth and phosphorus mobilization in tropical soils. Biol Fertil Soils 50:927–937. https://doi.org/10.1007/s00374-014-0913-z
Liu Z, Li YC, Zhang S, Fu Y, Fan X, Patel JS, Zhang M (2015) Characterization of phosphate-solubilizing bacteria isolated from calcareous soils. Appl Soil Ecol 96:217–224. https://doi.org/10.1016/j.apsoil.2015.08.003
Mohd SA, Birtaut C, Tanweer A (2013) Bioremediation of arsenic and lead by plants and microbes from contaminated soil. Res Plant Sci 1:68–73
Othman R, Panhwar QA (2014) Phosphate-solubilizing bacteria improves nutrient uptake in aerobic rice. In: Khan MS, Zaidi A, Musarrat J (eds) Phosphate solubilizing microorganisms: principles and application of microphos technology. Springer International Publishing, Cham, pp 207–224
Panda B, Rahman H, Chatlod LR, Panda J (2013) Isolation and characterization of phosphate solubilizing bacteria from the soils of Sikkim, India. Gastroenterology 142(5):S-515
Park JH, Bolan N (2013) Lead immobilization and bioavailability in microbial and root interface. J Hazard Mater 261(13):777–783. https://doi.org/10.1016/j.jhazmat.2013.02.010
Park JH, Bolan N, Megharaj M, Naidu R (2011) Isolation of phosphate solubilizing bacteria and their potential for lead immobilization in soil. J Hazard Mater 186:829–836. https://doi.org/10.1016/j.jhazmat.2010.09.095
Paul D, Sinha NS (2013) Bacteria showing phosphate solubilizing efficiency in river sediment. Electronic J Biosci 1:1–5
Pereira SIA, Castro PML (2014) Phosphate-solubilizing rhizobacteria enhance Zea mays growth in agricultural P-deficient soils. Ecol Eng 73:526–535. https://doi.org/10.1016/j.ecoleng.2014.09.060
Porder S, Vitousek PM, Chadwick OA, Chamberlain CP, Hilley GE (2007) Uplift, erosion, and phosphorus limitation in terrestrial ecosystems. Ecosystems 10(1):158–170. https://doi.org/10.1007/s10021-006-9011-x
Qian Y, Shi J, Chen Y, Lou L, Cui X, Cao R, Li P, Tang J (2010) Characterization of phosphate solubilizing bacteria in sediments from a shallow eutrophic lake and a wetland: isolation, molecular identification and phosphorus release ability determination. Molecules 15:8518–8533. https://doi.org/10.3390/molecules15118518
Raliya R, Tarafdar JC, Biswas P (2016) Enhancing the mobilization of native phosphorus in the mung bean rhizosphere using ZnO nanoparticles synthesized by soil fungi. J Agric Food Chem 64:3111–3118. https://doi.org/10.1021/acs.jafc.5b05224
Ribeiro DC, Martins G, Nogueira R, Cruz JV, Brito AG (2008) Phosphorus fractionation in volcanic lake sediments (Azores - Portugal). Chemosphere 70(7):1256–1263. https://doi.org/10.1016/j.chemosphere.2007.07.064
Rolewicz M, Rusek P, Mikos-Szymańska M, Cichy B, Dawidowicz M (2016) Obtaining of suspension fertilizers from incinerated sewage sludge ashes (ISSA) by a method of solubilization of phosphorus compounds by Bacillus megaterium bacteria. Waste Biomass Valoriz 7:871–877. https://doi.org/10.1007/s12649-016-9618-x
Ruban V, Lopez-Sanchez JF, Pardo P, Rauret G, Muntau H, Quevauviller P (2001) Development of a harmonised phosphorus extraction procedure and certification of a sediment reference material. J Environ Monit 3:121–125. https://doi.org/10.1039/b005672n
Sanchez-Roman M, Rivadeneyra MA, Vasconcelos C, McKenzie JA (2007) Biomineralization of carbonate and phosphate by moderately halophilic bacteria. FEMS Microbiol Ecol 61:273–284. https://doi.org/10.1111/j.1574-6941.2007.00336.x
Selvaraj P, Madhaiyan M, Sa T (2008) Isolation and identification of phosphate solubilizing bacteria from Chinese cabbage and their effect on growth and phosphorus utilization of plants. J Microbiol Biotechnol 18(4):773–777
Shaheen SM, Tsadilas CD, Starnatiadis S (2007) Inorganic phosphorus forms in some entisols and aridisols of Egypt. Geoderma 142:217–225. https://doi.org/10.1016/j.geoderma.2007.08.013
Sharma SB, Sayyed RZ, Trivedi MH, Gobi TA (2013) Phosphate solubilizing microbes: sustainable approach for managing phosphorus deficiency in agricultural soils. Springerplus 2:587. https://doi.org/10.1186/2193-1801-2-587
Son HJ, Park GT, Cha MS, Heo MS (2006) Solubilization of insoluble inorganic phosphates by a novel salt- and pH-tolerant Pantoea agglomerans R-42 isolated from soybean rhizosphere. Bioresour Technol 97:204–210. https://doi.org/10.1016/j.biortech.2005.02.021
Swati RT, Preeti T (2015) Phosphate solubilizing rhizospheric bacterial communities of different crops of Korea District of Chhattisgarh, India. Afr J Microbiol Res 9:1629–1636. https://doi.org/10.5897/AJMR2015.7522
Teng ZD, Zhu YY, Li M, Whelan MJ (2018) Microbial community composition and activity controls phosphorus transformation in rhizosphere soils of the Yeyahu Wetland in Beijing, China. Sci Total Environ 628-629:1266–1277. https://doi.org/10.1016/j.scitotenv.2018.02.115
Wang S, Liang X, Liu G, Li H, Liu X, Fan F, Xia W, Wang P, Ye Y, Li L, Liu Z, Zhu J (2013) Phosphorus loss potential and phosphatase activities in paddy soils. Plant Soil Environ 59:530–536. https://doi.org/10.17221/626/2013-PSE
Wang JC, Ren CQ, Chen HT, Zou YK, Bughio MA, Li QF (2017) Conversion of rainforest into agroforestry and monoculture plantation in China: consequences for soil phosphorus forms and microbial community. Sci Total Environ 595:769–778. https://doi.org/10.1016/j.scitotenv.2017.04.012
Xiang WL, Liang HZ, Liu S, Luo F, Tang J, Li MY, Che ZM (2011) Isolation and performance evaluation of halotolerant phosphate solubilizing bacteria from the rhizospheric soils of historic Dagong Brine Well in China. World J Microbiol Biotechnol 27:2629–2637
Yadav H, Gothwal RK, Nigam VK, Sinha-Roy S, Ghosh P (2013) Optimization of culture conditions for phosphate solubilization by a thermo-tolerant phosphate-solubilizing bacterium Brevibacillus sp. BISR-HY65 isolated from phosphate mines. Biocatal Agric Biotechnol 2:217–225. https://doi.org/10.1016/j.bcab.2013.04.005
Yang PX, Ma L, Chen MH, Xi JQ, He F, Duan CQ, Mo MH, Fang DH, Duan YQ, Yang FX (2012) Phosphate solubilizing ability and phylogenetic diversity of bacteria from P-rich soils around Dianchi Lake drainage area of China. Pedosphere 22:707–716. https://doi.org/10.1016/S1002-0160(12)60056-3
Yin Y, Liang CH (2013) Transformation of phosphorus fractions in paddy soil amended with pig manure. J Soil Sci Plant Nutr 13:809–818. https://doi.org/10.4067/S0718-95162013005000064
Yu X, Liu X, Zhu TH, Liu GH, Mao C (2012) Co-inoculation with phosphate-solubilzing and nitrogen-fixing bacteria on solubilization of rock phosphate and their effect on growth promotion and nutrient uptake by walnut. Eur J Soil Biol 50:112–117. https://doi.org/10.1016/j.ejsobi.2012.01.004
Yun QB, Cao XY, Song CL, Zhou YY, Li H, Sun AH, Hou J, Chen X (2014) Phosphorus fractions, sorption characteristics, and its release in sediments of a Chinese eutrophic river (Nanfei River). Fresenius Environ Bull 23:1222–1231
Zhang J, Li M, Liu S (2011) Seasonal variations and bioavailability of inorganic phosphorus in soil of Yeyahu Wetland in Beijing,China. Int J Sediment Res 26:181–192. https://doi.org/10.1016/S1001-6279(11)60085-1
Zhang Y, Huang L, Zhang Z, Wei L, Sun C, Chen D, Wu W (2016) Phosphorus fractions and phosphorus adsorption characteristics of soils from the water-level fluctuating zone of Nansi Lake, China. Pol J Environ Stud 25:865–872. https://doi.org/10.15244/pjoes/61007
Zhu J, Qu B, Li M (2017) Phosphorus mobilization in the Yeyahu Wetland: phosphatase enzyme activities and organic phosphorus fractions in the rhizosphere soils. Int Biodeterior Biodegrad 124:304–313. https://doi.org/10.1016/j.ibiod.2017.05.010
Zhu J, Li M, Whelan MJ (2018) Phosphorus activators contribute to legacy phosphorus availability in agricultural soils: a review. Sci Total Environ 612:522–537. https://doi.org/10.1016/j.scitotenv.2017.08.095
Acknowledgments
We are grateful to Jing Zhu, Yunyun Zhu, Yaoqiang Huo, Wen Shao and Kedong Gong for their assistance in laboratory work.
Funding
This study was funded by the National Natural Science Foundation of China (no. 51279004) and the University Student of Science and Technology Innovation Project of Beijing (no. S201510022107).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Diane Purchase
Highlights
• The most efficient IPSB strain could convert over 430 mg L−1 orthophosphate.
• IPSBs had a better P-solubilizing ability than OPBSs in the rhizosphere soils.
• Acidification seemed to be the main strategy for P solubilizing from IP.
• Mineralization of OP was the main strategy for P solubilizing from OP.
Rights and permissions
About this article
Cite this article
Teng, Z., Chen, Z., Zhang, Q. et al. Isolation and characterization of phosphate solubilizing bacteria from rhizosphere soils of the Yeyahu Wetland in Beijing, China. Environ Sci Pollut Res 26, 33976–33987 (2019). https://doi.org/10.1007/s11356-018-2955-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-2955-5