Abstract
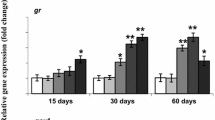
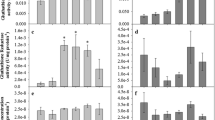
Metallic contamination is a widespread phenomena, particularly in areas impacted by human activities, and has become a relevant environmental concern. However, the toxicity of metals on fish requires full characterization in terms of short- and long-term effects. Thus, the purpose of this study was to determine the acute and chronic oxidative stress response in liver and gills of Gambusia holbrooki exposed to copper and cadmium. To assess the effects of these two metals, we adopted a strategy of analyzing the pollution effects caused by salts of the two metallic elements, and we quantified the oxidative stress biomarkers catalase, glutathione reductase, glutathione-S-transferases, and lipid peroxidation after exposure (4 and 28 days) to ecologically relevant concentrations, thus simulating actual conditions of exposure in the wild. Our results showed that copper elicited strong effects in all tested biomarkers for both acute and chronic challenges. Cadmium caused a similar response and was shown to cause significant changes particularly in catalase and glutathione-S-transferases activities. These findings evidence that ecologically relevant concentrations of common anthropogenic contaminants are causative agents of serious imbalances (namely oxidative stress) that are likely to trigger life-threatening events.


Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 6:105–121
Ahmad I, Oliveira M, Pacheco M, Santos MA (2005) Anguilla anguilla L. oxidative stress biomarkers responses to copper exposure with or without b-naphthoflavone pre-exposure. Chemosphere 61:267–275
Almeida JA, Diniz YS, Marques SFG, Faine LA, Ribas BO, Burneiko RC, Novelli ELB (2002) The use of the oxidative stress responses as biomarkers in Nile tilapia (Oreochromis niloticus) exposed to in vivo cadmium contamination. Environ Int 27:673–679
Banni M, Chouchene L, Said K, Kerkeni A, Messaoudi I (2011) Mechanisms underlying the protective effect of zinc and selenium against cadmium-induced oxidative stress in zebrafish Danio rerio. Biometals 24(6):981–992
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–310
Carlberg I, Mannervik B (1985) Glutathione reductase. Methods Enzymol 113:484–490
Espinoza HM, Williams CR, Gallagher EP (2012) Effect of cadmium on glutathione S-transferase and metallothionein gene expression in coho salmon liver, gill and olfactory tissues. Aquat Toxicol 110–111:37–44
Eyckmans M, Celis N, Horemans N, Blust R, De Boeck G (2011) Exposure to waterborne copper reveals differences in oxidative stress response in three freshwater fish species. Aquat Toxicol 103:112–120
Fernandes C, Fontaínhas-Fernandes A, Cabral D, Salgado MA (2008). Heavy metals in water, sediment and tissues of Liza saliens from Esmoriz–Paramos lagoon, Portugal. Environ Monit Assess 136:267–275
Ferreira, J.G., Simas, T., Nobre, A., Silva, M.C., Shifferegger, K., Lencart-Silva, J. (2003). Identification of Sensitive Areas and Vulnerable Zones in Transitional and Coastal Portuguese Systems – Application of the United States National Estuarine Eutrophication Assessment to the Minho, Lima, Douro, Ria de Aveiro, Mondego, Tagus, Sado, Mira, Ria Formosa and Guadiana Systems. Edited by Instituto da Água and Institute of Marine Research, Portugal
Firat O, Kargin F (2010) Effects of zinc and cadmium on erythrocyte antioxidant systems of a freshwater fish Oreochromis niloticus. J Biochem Mol Toxicol 24(4):223–229
García-Fernández AJ, Bayoumi AE, Pérez-Pertejo Y, Motas M, Reguera RM, Ordóñez C, Balaña-Fouce R, Ordóñez D (2002) Alterations of the glutathione–redox balance induced by metals in CHO-K1 cells. Comp Biochem Physiol C Toxicol Pharmacol 132(3):365–373
Gravato C, Teles M, Oliveira M, Santos MA (2006) Oxidative stress, liver biotransformation and genotoxic effects induced by copper in Anguilla anguilla L.—the influence of pre-exposure to β-naphthoflavone. Chemosphere 65(10):1821–1830
Greco L, Serrano R, Blanes MA, Serrano E, Capri E (2010) Bioaccumulation markers and biochemical responses in European sea bass (Dicentrarchus labrax) raised under different environmental conditions. Ecotoxicol Environ Saf 73:38–45
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7137
Jia X, Zhang H, Liu X (2011) Low levels of cadmium exposure induce DNA damage and oxidative stress in the liver of Oujiang colored common carp Cyprinus carpio var. color. Fish Physiol Biochem 37(1):97–103
Jiang W-D, Wua P, Kuang S-Y, Liu Y, Jiang J, Hu K, Li S-H, Tang L, Feng L, Zhou X-Q (2011) Myo-inositol prevents copper-induced oxidative damage and changes in antioxidant capacity in various organs and the enterocytes of juvenile Jian carp (Cyprinus carpio var. Jian). Aquat Toxicol 105:543–551
Jin Y, Zhang X, Shu L, Chen L, Sun L, Qian H, Liu W, Fu Z (2010) Oxidative stress response and gene expression with atrazine exposure in adult female zebrafish (Danio rerio). Chemosphere 78:846–852
Johnston BD, Scown TM, Moger J, Cumberland SA, Baalousha M, Linge K, van Aerle R, Jarvis K, Lead JR, Tyler CR (2010) Bioavailability of nanoscale metal oxides TiO2, CeO2, and ZnO to fish. Environ Sci Technol 44:1144–1151
Jomova K, Valko M (2011) Advances in metal-induced oxidative stress and human disease. Toxicology 283:65–87
Lapointe D, Pierron F, Couture P (2011) Individual and combined effects of heat stress and aqueous or dietary copper exposure in fathead minnows (Pimephales promelas). Aquat Toxicol 104:80–85
Liu X-D, Thiele DJ (1997) Yeast metallothionein gene expression in response to metals and oxidative stress. Methods 11(3):289–299
López-Galindo C, Vargas-Chacoff L, Nebot E, Casanueva JF, Rubio D, Solé M, Mancera JM (2010) Biomarker responses in Solea senegalensis exposed to sodium hypochlorite used as antifouling. Chemosphere 78:885–893
Lushchak VI (2011a) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101:13–30
Lushchak VI (2011b) Adaptive response to oxidative stress: bacteria, fungi, plants and animals. Comp Biochem Physiol C 153:175–190
Modesto KA, Martinez CBR (2010) Roundup (R) causes oxidative stress in liver and inhibits acetylcholinesterase in muscle and brain of the fish Prochilodus lineatus. Chemosphere 78:294–299
Mucha AP, Vasconcelos MT, Bordalo AA (2003) Macrobenthic community in the Douro estuary: relations with trace metals and natural sediment characteristics. Environ Pollut 121(2):169–180
Ninh Pham A, Xing G, Miller CJ, Waite TD (2013) Fenton-like copper redox chemistry revisited: hydrogen peroxide and superoxide mediation of copper-catalyzed oxidant production. J Catal 301:54–64
Nunes B, Carvalho F, Guilhermino L (2004) Acute and chronic effects of clofibrate and clofibric acid on the enzymes acetylcholinesterase, lactate dehydrogenase and catalase of the mosquitofish, Gambusia holbrooki. Chemosphere 57:1581–1589
Nunes B, Gaio R, Carvalho F, Guilhermino L (2008) Behaviour and biomarkers of oxidative stress in Gambusia holbrooki after acute exposure to widely used pharmaceuticals and a detergent. Ecotoxicol Environ Saf 71:341–354
OECD (1992) Test no. 203: fish, acute toxicity test. OECD Guidel Test Chem 1(2):1–9
OECD (2000) Test no. 215: fish, juvenile growth test. OECD Guidel Test Chem 1(2):1–16
OECD (2002) Guidance document on aquatic ecotoxicology in the context of the Directive 91/414/EEC. European Commission Health & Consumer Protection Directorate-General, Directorate E-Food Safety: plant health, animal health and welfare, international questions, E1-Plant health: 1–61
Ognjanovic BI, Dorordevic NZ, Perendndija BR, Despotootovic SG, Žikic RV, Štajn AS, Saicic ZS (2008) Concentration of antioxidant compounds and lipid peroxidation in the liver and white muscle of hake (Merluccius merluccius L.) in the Adriatic Sea. Arch Biol Sci 60:601–607
Olivari FA, Hernández PP, Allende ML (2008) Acute copper exposure induces oxidative stress and cell death in lateral line hair cells of zebrafish larvae. Brain Res 1244:1–12
Pretto A, Loro VL, Baldisserotto B, Pavanato MA, Moraes BS, Menezes C, Cattaneo R, Clasen B, Finamor IA, Dressler V (2011) Effects of water cadmium concentrations on bioaccumulation and various oxidative stress parameters in Rhamdia quelen. Arch Environ Contam Toxicol 60(2):309–318
Pytharopoulou S, Grintzalis K, Sazakli E, Leotsinidis M, Georgiou CD, Kalpaxis DL (2011) Translational responses and oxidative stress of mussels experimentally exposed to Hg, Cu and Cd: one pattern does not fit at all. Aquat Toxicol 105:157–165
Rensing C, Rosen BP (2009) Heavy metals cycle (arsenic, mercury, selenium, others). Encyclopedia of microbiology, 3rd edn. Academic, p 205–219
Sáez MI, Casas JJ, Guil-Gerrero JL, Gil MD, Cañero R, Suárez MD (2013a) Effects of organic matter, alkalinity and pH on acute copper toxicity to mosquitofish, Gambusia holbrooki: Implications for a multipurpose management of irrigation ponds. Int Rev Hydrobiol 98:1–9
Sáez MI, García-Mesa S, Casas JJ, Guil-Guerrero LJ, Venegas-Venegas CE, Morales AE, Suárez MD (2013b) Effect of sublethal concentrations of waterborne copper on lipid peroxidation and enzymatic antioxidant response in Gambusia holbrooki. Environ Toxicol Pharmacol 36:125–134
Saini N, Oelhafen S, Hua H, Georgiev O, Schaffner W, Büeler H (2010) Extended lifespan of Drosophila parkin mutants through sequestration of redox-active metals and enhancement of anti-oxidative pathways. Neurobiol Dis 40(1):82–92
Sanchez A, Palluel O, Meunier L, Coquery M, Porcher J-M, Aït-Aïssaa S (2005) Copper-induced oxidative stress in three-spined stickleback: relationship with hepatic metal levels. Environ Toxicol Pharmacol 19:177–183
Serafim A, Company R, Lopes B, Rosa J, Cavaco A, Castela G, Castela E, Olea N, Bebianno MJ (2012) Assessment of essential and nonessential metals and different metal exposure biomarkers in the human placenta in a population from the south of Portugal. J Toxicol Environ Health A 75(13–15):867–877
Shaikh ZA, Vu TT, Zaman K (1999) Oxidative stress as a mechanism of chronic cadmium-induced hepatotoxicity and renal toxicity and protection by antioxidants. Toxicol Appl Pharmacol 154:256–263
Soares SS, Martins H, Gutiérrez-Merino C, Aureliano M (2008) Vanadium and cadmium in vivo effects in teleost cardiac muscle: metal accumulation and oxidative stress markers. Comp Biochem Physiol C 147:168–178
Vergauwen L, Hagenaars A, Blusta R, Knapen D (2013) Temperature dependence of long-term cadmium toxicity in the zebrafish is not explained by liver oxidative stress: evidence from transcript expression to physiology. Aquat Toxicol 126:52–62
Vieira LR, Gravato C, Soares AMVM, Morgado F, Guilhermino L (2009) Acute effects of copper and mercury on the estuarine fish Pomatoschistus microps: linking biomarkers to behaviour. Chemosphere 76:1416–1427
Wang X, Fang H, Huang Z, Shang W, Hou T, Cheng A, Cheng H (2013) Imaging ROS signaling in cells and animals. J Mol Med 91(8):917–927
Won E-J, Rhee J-S, Kim R-O, Ra K, Kim K-T, Shin K-H, Lee J-S (2012) Susceptibility to oxidative stress and modulated expression of antioxidant genes in the copper-exposed polychaete Periunereis nuntia. Comp Biochem Physiol C 155:344–351
Acknowledgments
Joana L. Pereira is recipient of an individual scholarship by the Portuguese Foundation for Science and Technology (SFRH/BPD/44733/2008). This study was supported by FEDER funds through the program COMPETE and by national funds through FCT under the scope of the project BiOtoMetal (PTDC/AMB/70431/2006).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Nunes, B., Caldeira, C., Pereira, J.L. et al. Perturbations in ROS-related processes of the fish Gambusia holbrooki after acute and chronic exposures to the metals copper and cadmium. Environ Sci Pollut Res 22, 3756–3765 (2015). https://doi.org/10.1007/s11356-014-3580-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3580-6