Abstract
Purpose
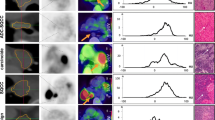
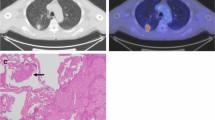
Tumor delineation within an atelectasis in lung cancer patients is not always accurate. When T staging is done by integrated 2-deoxy-2-[18F]fluoro-d-glucose ([18F]FDG)-positron emission tomography (PET)/X-ray computer tomography (CT), tumors of neuroendocrine differentiation and slowly growing tumors can present with reduced FDG uptake, thus aggravating an exact T staging. In order to further exhaust information derived from [18F]FDG-PET/CT, we evaluated the impact of CT density and maximum standardized uptake value (SUVmax) for the classification of different tumor subtypes within a surrounding atelectasis, as well as possible cutoff values for the differentiation between the primary tumor and atelectatic lung tissue.
Procedures
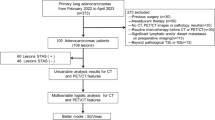
Seventy-two patients with histologically proven lung cancer and adjacent atelectasis were investigated. Non-contrast-enhanced [18F]FDG-PET/CT was performed within 2 weeks before surgery/biopsy. Boundaries of the primary within the atelectasis were determined visually on the basis of [18F]FDG uptake; CT density was quantified manually within each primary and each atelectasis.
Results
CT density of the primary (36.4 Hounsfield units (HU) ± 6.2) was significantly higher compared to that of atelectatic lung (24.3 HU ± 8.3; p < 0.01), irrespective of the histological subtype. The discrimination between different malignant tumors using density analysis failed. SUVmax was increased in squamous cell carcinomas compared to adenocarcinomas. Irrespective of the malignant subtype, a possible cutoff value of 24 HU may help to exclude the presence of a primary in lesions below 24 HU, whereas a density above a threshold of 40 HU can help to exclude atelectatic lung.
Conclusion
Density measurements in patients with lung cancer and surrounding atelectasis may help to delineate the primary tumor, irrespective of the specific lung cancer subtype. This could improve T staging and radiation treatment planning (RTP) without additional application of a contrast agent in CT, or an additional magnetic resonance imaging (MRI), even in cases of lung tumors of neuroendocrine differentiation or in slowly growing tumors with less avidity to [18F]FDG.






Similar content being viewed by others
References
Kratochwil C, Haberkorn U, Giesel FL (2010) PET/CT for diagnostics and therapy stratification of lung cancer. Radiologe 50:684–691
Abramyuk A, Appold S, Zöphel K, Hietschold V, Baumann M, Abolmaali N (2012) Quantitative modifications of TNM staging, clinical staging and therapeutic intent by FDG-PET/CT in patients with non small cell lung cancer scheduled for radiotherapy—a retrospective study. Lung Cancer 78:148–152
Flechsig P, Mehndiratta A, Haberkorn U, Kratochwil C, Giesel FL (2015) PET/MRI and PET/CT in lung lesions and thoracic malignancies. Semin Nucl Med 45:268–281
Manenti G, Raguso M, D'Onofrio S et al (2013) Pancoast tumor: the role of magnetic resonance imaging. Case Rep Radiol 2013:479120
MacManus M, Everitt S, Schimek-Jasch T, Li XA, Nestle U, Kong FMS (2017) Anatomic, functional and molecular imaging in lung cancer precision radiation therapy: treatment response assessment and radiation therapy personalization. Transl Lung Cancer Res 6:670–688
Kwee TC, Torigian DA, Alavi A (2013) Oncological applications of positron emission tomography for evaluation of the thorax. J Thorac Imaging 28:11–24
Gondi V, Bradley K, Mehta M, Howard A, Khuntia D, Ritter M, Tomé W (2007) Impact of hybrid fluorodeoxyglucose positron-emission tomography/computed tomography on radiotherapy planning in esophageal and non-small-cell lung cancer. Int J Radiat Oncol Biol Phys 67:187–195
Kruser TJ, Bradley KA, Bentzen SM, Anderson BM, Gondi V, Khuntia D, Perlman SB, Tome WA, Chappell RJ, Walker WL, Mehta MP (2009) The impact of hybrid PET-CT scan on overall oncologic management, with a focus on radiotherapy planning: a prospective, blinded study. Technol Cancer Res Treat 8:149–158
Schaarschmidt BM, Grueneisen J, Metzenmacher M, Gomez B, Gauler T, Roesel C, Heusch P, Ruhlmann V, Umutlu L, Antoch G, Buchbender C (2017) Thoracic staging with (18)F-FDG PET/MR in non-small cell lung cancer—does it change therapeutic decisions in comparison to 18F-FDG PET/CT? Eur Radiol 27:681–688
Lee SM, Goo JM, Park GM et al (2016) Preoperative staging of non-small cell lung cancer: prospective comparison of PET/MR and PET/CT. Eur Radiol 26:3850–3857
Heusch P, Buchbender C, Köhler J et al (2014) Thoracic staging in lung cancer: prospective comparison of 18F-FDG PET/MR imaging and 18F-FDG PET/CT. J Nucl Med 55:373–378
Huellner MW, de Galiza BF, Husmann L et al (2016) TNM staging of non-small cell lung cancer: comparison of PET/MR and PET/CT. J Nucl Med 57:21–26
Lambin P, Leijenaar RTH, Deist TM, Peerlings J, de Jong EEC, van Timmeren J, Sanduleanu S, Larue RTHM, Even AJG, Jochems A, van Wijk Y, Woodruff H, van Soest J, Lustberg T, Roelofs E, van Elmpt W, Dekker A, Mottaghy FM, Wildberger JE, Walsh S (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762
Chen B, Zhang R, Gan Y, Yang L, Li W (2017) Development and clinical application of radiomics in lung cancer. Radiat Oncol 12:154
Bashir U, Siddique MM, Mclean E, Goh V, Cook GJ (2016) Imaging heterogeneity in lung cancer: techniques, applications, and challenges. AJR Am J Roentgenol 207:534–543
Lambin P, Rios-Velazquez E, Leijenaar R, Carvalho S, van Stiphout RGPM, Granton P, Zegers CML, Gillies R, Boellard R, Dekker A, Aerts HJWL (2012) Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer 48:441–446
Avanzo M, Stancanello J, El Naqa I (2017) Beyond imaging: the promise of radiomics. Phys Med 38:122–139
Kumar V, Gu Y, Basu S, Berglund A, Eschrich SA, Schabath MB, Forster K, Aerts HJWL, Dekker A, Fenstermacher D, Goldgof DB, Hall LO, Lambin P, Balagurunathan Y, Gatenby RA, Gillies RJ (2012) Radiomics: the process and the challenges. Magn Reson Imaging 30:1234–1248
Wilson R, Devaraj A (2017) Radiomics of pulmonary nodules and lung cancer. Transl Lung Cancer Res 6:86–91
Coroller TP, Agrawal V, Huynh E, Narayan V, Lee SW, Mak RH, Aerts HJWL (2017) Radiomic-based pathological response prediction from primary tumors and lymph nodes in NSCLC. J Thorac Oncol 12:467–476
Jiao Y, Ren Y, Zheng X (2017) Quantitative imaging assessment of tumor response to chemoradiation in lung cancer. Zhongguo Fei Ai Za Zhi 20:407–414
Huang Y, Liu Z, He L, Chen X, Pan D, Ma Z, Liang C, Tian J, Liang C (2016) Radiomics signature: a potential biomarker for the prediction of disease-free survival in early-stage (I or II) non-small cell lung cancer. Radiology 281:947–957
Jin JY, Kong FM (2016) Personalized radiation therapy (PRT) for lung cancer. Adv Exp Med Biol 890:175–202
Thawani R, McLane M, Beig N, Ghose S, Prasanna P, Velcheti V, Madabhushi A (2018) Radiomics and radiogenomics in lung cancer: a review for the clinician. Lung Cancer 115:34–41
Constanzo J, Wei L, Tseng HH, El Naqa I (2017) Radiomics in precision medicine for lung cancer. Transl Lung Cancer Res 6:635–647
Shao T, Yu L, Li Y, Chen M (2015) Density and SUV ratios from PET/CT in the detection of mediastinal lymph node metastasis in non-small cell lung cancer. Zhongguo Fei Ai Za Zhi 18:155–160
Flechsig P, Kratochwil C, Schwartz LH, Rath D, Moltz J, Antoch G, Heussel CP, Rieser M, Warth A, Zabeck H, Kauczor HU, Haberkorn U, Giesel FL (2014) Quantitative volumetric CT-histogram analysis in N-staging of 18F-FDG-equivocal patients with lung cancer. J Nucl Med 55:559–564
Flechsig P, Frank P, Kratochwil C, Antoch G, Rath D, Moltz J, Rieser M, Warth A, Kauczor HU, Schwartz LH, Haberkorn U, Giesel FL (2017) Radiomic analysis using density threshold for FDG-PET/CT-based N-staging in lung cancer patients. Mol Imaging Biol 19:315–322
Giesel FL, Schneider F, Kratochwil C, Rath D, Moltz J, Holland-Letz T, Kauczor HU, Schwartz LH, Haberkorn U, Flechsig P (2017) Correlation between SUVmax and CT radiomic analysis using lymph node density in PET/CT-based lymph node staging. J Nucl Med 58:282–287
Tournoy KG, Maddens S, Gosselin R, van Maele G, van Meerbeeck JP, Kelles A (2007) Integrated FDG-PET/CT does not make invasive staging of the intrathoracic lymph nodes in non-small cell lung cancer redundant: a prospective study. Thorax 62:696–701
Cuaron J, Dunphy M, Rimner A (2012) Role of FDG-PET scans in staging, response assessment, and follow-up care for non-small cell lung cancer. Front Oncol 2:208
Brown RS, Leung JY, Kison PV, Zasadny KR, Flint A, Wahl RL (1999) Glucose transporters and FDG uptake in untreated primary human non-small cell lung cancer. J Nucl Med 40:556–565
Giesel F, Will L, Lawal I, Lengana T, Kratochwil C, Vorster M, Neels O, Reyneke F, Haberkon U, Kopka K, Sathekge M (2017) Intra-individual comparison of 18F-PSMA-1007 and 18F-DCFPyL PET/CT in the prospective evaluation of patients with newly diagnosed prostate carcinoma: a pilot study. J Nucl Med. https://doi.org/10.2967/jnumed.117.204669
Naalsund A, Maublant J (2006) The solitary pulmonary nodule--is it malignant or benign? Diagnostic performance of Tc-depreotide SPECT. Respiration 73:634–641
Halley A, Hugentobler A, Icard P, Porret E, Sobrio F, Lerochais JP, Bouvard G, Zalcman G, Agostini D (2005) Efficiency of 18F-FDG and 99mTc-depreotide SPECT in the diagnosis of malignancy of solitary pulmonary nodules. Eur J Nucl Med Mol Imaging 32:1026–1032
Menda Y, Kahn D (2002) Somatostatin receptor imaging of non-small cell lung cancer with 99mTc depreotide. Semin Nucl Med 32:92–96
Jensen TH, Bech M, Binderup T, Böttiger A, David C, Weitkamp T, Zanette I, Reznikova E, Mohr J, Rank F, Feidenhans’l R, Kjær A, Højgaard L, Pfeiffer F (2013) Imaging of metastatic lymph nodes by X-ray phase-contrast micro-tomography. PLoS One 8:e54047
Flechsig P, Choyke P, Kratochwil C, Warth A, Antoch G, Holland Letz T, Rath D, Eichwald V, Huber PE, Kauczor HU, Haberkorn U, Giesel FL (2016) Increased x-ray attenuation in malignant vs. benign mediastinal nodes in an orthotopic model of lung cancer. Diagn Interv Radiol 22:35–39
Strobel K, Thuerl CM, Hany TF (2005) How much intravenous contrast is needed in FDG-PET/CT? Nuklearmedizin 44(Suppl 1):S32–S37
Pfannenberg AC, Aschoff P, Brechtel K, Müller M, Bares R, Paulsen F, Scheiderbauer J, Friedel G, Claussen CD, Eschmann SM (2007) Low dose non-enhanced CT versus standard dose contrast-enhanced CT in combined PET/CT protocols for staging and therapy planning in non-small cell lung cancer. Eur J Nucl Med Mol Imaging 34:36–44
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the institutional review board and conducted according to the guidelines of the institutional review board and to good clinical practice according to the ethical principles that have their origin in the Declaration of Helsinki. In this retrospective analysis, the requirement of informed consent was waived by the institutional review board.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Flechsig, P., Rastgoo, R., Kratochwil, C. et al. Impact of Computer-Aided CT and PET Analysis on Non-invasive T Staging in Patients with Lung Cancer and Atelectasis. Mol Imaging Biol 20, 1044–1052 (2018). https://doi.org/10.1007/s11307-018-1196-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-018-1196-9