Abstract
Introduction
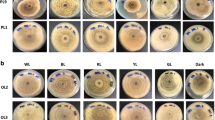
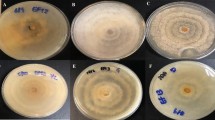
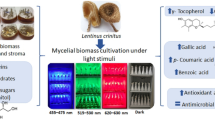
The present work identified and compared intracellular metabolites and metabolic networks in mycelial cultures of Lasiodiplodia theobromae grown under 12 natural light and 24 hours’ dark using a 1 H NMR-based metabolomics approach.
Materials and methods
Fungal cultures were grown in potato dextrose media, and metabolites were extracted by sonication with sodium phosphate-buffered saline (pH = 6.0, 10% D2O, 0.1 mM TSP) from mycelium samples collected every week over four weeks.
Results
Multivariate analyses revealed that the light exposure group showed a positive correlation within beta-hydroxybutyrate, acetoacetate, acetone, betaine, choline, glycerol, and phosphocholine. On the other hand, phenyl acetate, leucine, isoleucine, valine, and tyrosine were positively correlated with dark conditions. Light favored the oxidative degradation of valine, leucine, and isoleucine, leading to the accumulation of choline, phosphocholine, betaine, and ketone bodies (ketogenesis). Ketogenesis, gluconeogenesis, and the biosynthesis of choline, phosphocholine, and betaine, were considered discriminatory routes for light conditions. The light-sensing pathways were interlinked with fungal development, as verified by the increased production of mycelia biomass without fruiting bodies and stress signaling, as demonstrated by the increased production of pigments.




Similar content being viewed by others
References
Calvo, A. M., Wilson, R. A., Bok, J. W., & Keller, N. P. (2002). Relationship between secondary metabolism and fungal development. Microbiology and molecular biology reviews, 66(3), 447–459. https://doi.org/10.1128/MMBR.66.3.447-459.2002.
Carlini, E. A., Duarte-Almeida, J. M., Rodrigues, E., & Tabach, R. (2010). Antiulcer effect of the pepper trees Schinus terebinthifolius Raddi (aroeira-da-praia) and myracrodruon urundeuva Allemão, Anacardiaceae (aroeira-do-sertão). Revista Brasileira de Farmacognosia, 20, 140–146. https://doi.org/10.1590/S0102-695X2010000200001.
Chen, S. F., Morgan, D., Beede, R. H., & Michailides, T. J. (2013). First report of Lasiodiplodia theobromae associated with stem canker of almond in California. Plant Disease, 97(7), 994–994. https://doi.org/10.1094/PDIS-11-12-1033-PDN.
Cheong, K. K., Strub, C., Montet, D., Durand, N., Alter, P., Meile, J. C., Galindo, S. S., & Fontana, A. (2016). Effect of different light wavelengths on the growth and ochratoxin A production in aspergillus carbonarius and aspergillus westerdijkiae. Fungal Biology, 120(5), 745–751. https://doi.org/10.1016/j.funbio.2016.02.005.
Darwish, A. M. G. (2019). Fungal mycotoxins and natural antioxidants: Two sides of the same coin and significance in food safety. Microbial Biosystems, 4(1), 1–16. https://doi.org/10.21608/mb.2019.37468.
Dias, L. P., Souza, R. K., Pupin, B., & Rangel, D. E. (2021). Conidiation under illumination enhances conidial tolerance of insect-pathogenic fungi to environmental stresses. Fungal Biology, 125(11), 891–904. https://doi.org/10.1016/j.funbio.2021.06.003.
Fanelli, F., Schmidt-Heydt, M., Haidukowski, M., Susca, A., Geisen, R., Logrieco, A., & Mulè, G. (2012). Influence of light on growth, conidiation and fumonisin production by Fusarium verticillioides. Fungal biology, 116(2), 241–248. https://doi.org/10.1016/j.funbio.2011.11.007.
Góes-Neto, A., Marcelino, V. R., Verbruggen, H., da Silva, F. F., & Badotti, F. (2020). Biodiversity of endolithic fungi in coral skeletons and other reef substrates revealed with 18S rDNA metabarcoding. Coral Reefs, 39, 229–238. https://doi.org/10.1007/s00338-019-01880-y.
Gow, N. A., & Gadd, G. M. (Eds.). (2007). Growing fungus. Springer Science & Business Media.
Guedes, R. L. M., Prosdocimi, F., Fernandes, G. R., Moura, L. K., Ribeiro, H. A. L., & Ortega, J. M. (2011, December). Amino acids biosynthesis and nitrogen assimilation pathways: a great genomic deletion during eukaryotes evolution. In BMC genomics (Vol. 12, No. 4, pp. 1–13). BioMed Central. https://doi.org/10.1186/1471-2164-12-S4-S2.
Hawkins, D. M., Basak, S. C., & Mills, D. (2003). Assessing model fit by cross-validation. Journal of chemical information and computer sciences, 43(2), 579–586. https://doi.org/10.1021/ci025626i.
Hawksworth, D. L., & Lücking, R. (2017). Fungal diversity revisited: 2.2 to 3.8 million species. Microbiology spectrum, 5(4), 5–4. https://doi.org/10.1128/microbiolspec.FUNK-0052-2016.
Jami, M. S., Martín, J. F., Barreiro, C., Domínguez-Santos, R., Vasco-Cárdenas, M. F., Pascual, M., & García-Estrada, C. (2018). Catabolism of phenylacetic acid in Penicillium rubens. Proteome-wide analysis in response to the benzylpenicillin side chain precursor. Journal of proteomics, 187, 243–259. https://doi.org/10.1016/j.jprot.2018.08.006.
Jiménez-Contreras, E., Torres-Salinas, D., Moreno, R., Baños, R., & López-Cózar, E. (2009). Response surface methodology and its application in evaluating scientific activity. Scientometrics, 79(1), 201–218. https://doi.org/10.1007/s11192-009-0413-3.
Kalra, R., Conlan, X. A., & Goel, M. (2020). Fungi as a potential source of pigments: Harnessing filamentous fungi. Frontiers in Chemistry, 8, 369. https://doi.org/10.3389/fchem.2020.00369.
Lambou, K., Pennati, A., Valsecchi, I., Tada, R., Sherman, S., Sato, H., & Latgé, J. P. (2013). Pathway of glycine betaine biosynthesis in aspergillus fumigatus. Eukaryotic cell, 12(6), 853–863. https://doi.org/10.1128/EC.00348-12.
Lange, L. (2010). The importance of fungi for a more sustainable future on our planet. Fungal Biology Reviews, 24(3–4), 90–92. https://doi.org/10.1016/j.fbr.2010.12.002.
Lin, L., & Xu, J. (2020). Fungal pigments and their roles associated with human health. Journal of Fungi, 6(4), 280. https://doi.org/10.3390/jof6040280.
Liu, R., Meng, X., Mo, C., Wei, X., & Ma, A. (2022). Melanin of fungi: From classification to application. World Journal of Microbiology and Biotechnology, 38(12), 228. https://doi.org/10.1007/s11274-022-03415-0.
Luo, F., Zhou, H., Zhou, X., Xie, X., Li, Y., Hu, F., & Huang, B. (2020). The intermediates in branched-chain amino acid biosynthesis are indispensable for conidial germination of the insect-pathogenic fungus metarhizium robertsii. Applied and environmental microbiology, 86(20), e01682–e01620. https://doi.org/10.1128/AEM.01682-20.
Maggi, O., Tosi, S., Angelova, M., Lagostina, E., Fabbri, A. A., Pecoraro, L., Altobelli, E., Pico, A. A., Savino, E., Branda, E., Turchetti, B., Zotti, M., Vizzini, A., & Buzzini, P. (2013). Adaptation of fungi, including yeasts, to cold environments. Plant Biosystems-An International Journal Dealing with all Aspects of Plant Biology, 147(1), 247–258. https://doi.org/10.1080/11263504.2012.753135.
Mann, G., Mora, S., Madu, G., & Adegoke, O. A. (2021). Branched-chain amino acids: Catabolism in skeletal muscle and implications for muscle and whole-body metabolism. Frontiers in Physiology, 12, 702826. https://doi.org/10.3389/fphys.2021.702826.
Markley, J. L., Brüschweiler, R., Edison, A. S., Eghbalnia, H. R., Powers, R., Raftery, D., & Wishart, D. S. (2017). The future of NMR-based metabolomics. Current opinion in biotechnology, 43, 34–40. https://doi.org/10.1016/j.copbio.2016.08.001.
Meruvu, H., & Dos Santos, J. C. (2021). Colors of life: A review on fungal pigments. Critical reviews in biotechnology, 41(8), 1153–1177. https://doi.org/10.1080/07388551.2021.1901647.
Middelhoven, W. J. (1993). Catabolism of benzene compounds by ascomycetous and basidiomycetous yeasts and yeastlike fungi: A literature review and an experimental approach. Antonie van Leeuwenhoek, 63, 125–144. https://doi.org/10.1007/BF00872388.
Moubasher, H. A., Balbool, B. A., Helmy, Y. A., Alsuhaibani, A. M., Atta, A. A., Sheir, D. H., & Abdel-Azeem, A. M. (2022). Insights into asparaginase from endophytic fungus Lasiodiplodia theobromae: Purification, characterization and antileukemic activity. International journal of environmental research and public health, 19(2), 680. https://doi.org/10.3390/ijerph19020680.
Murphy, D. J., & Vance, J. (1999). Mechanisms of lipid-body formation. Trends in biochemical sciences, 24(3), 109–115. https://doi.org/10.1016/S0968-0004(98)01349-8.
Nagarajan, K., Ibrahim, B., Ahmad Bawadikji, A., Lim, J. W., Tong, W. Y., Leong, C. R., Khaw, K. Y., & Tan, W. N. (2022). Recent developments in metabolomics studies of endophytic fungi. Journal of Fungi, 8(1), 28. https://doi.org/10.3390/jof8010028.
Pavan, M. E., López, N. I., & Pettinari, M. J. (2020). Melanin biosynthesis in bacteria, regulation and production perspectives. Applied Microbiology and Biotechnology, 104, 1357–1370. https://doi.org/10.1007/s00253-019-10245-y.
Pereira, A. L., Silva, G. S., & Ribeiro, V. Q. (2006). Caracterização fisiológica, cultural e patogênica de diferentes isolados de lasiodiplodia theobromae. Fitopatologia Brasileira, 31, 572–578. https://doi.org/10.1590/S0100-41582006000600006.
Pombeiro-Sponchiado, S. R., Sousa, G. S., Andrade, J. C., Lisboa, H. F., & Gonçalves, R. C. (2017). Production of melanin pigment by fungi and its biotechnological applications. Melanin, 1(4), 47–75.
Porto, V. A., da Rocha Júnior, E. R., Ursulino, J. S., Porto, R. S., da Silva, M., de Jesus, L. W. O., De Oliveira, J. M., Crispim, A. C., Santos, J. C. C., & de Aquino, T. M. (2023). NMR-based metabolomics applied to ecotoxicology with zebrafish (Danio rerio) as a prominent model for metabolic profiling and biomarker discovery: Overviewing the most recent approaches. Science of The Total Environment, 161737. https://doi.org/10.1016/j.scitotenv.2023.161737.
Richter, K., Mathes, V., Fronius, M., Althaus, M., Hecker, A., Krasteva-Christ, G., & Grau, V. (2016). Phosphocholine–an agonist of metabotropic but not of ionotropic functions of α9-containing nicotinic acetylcholine receptors. Scientific reports, 6(1), 1–13. https://doi.org/10.1038/srep28660.
Schertl, P., Danne, L., & Braun, H. P. (2017). 3-Hydroxyisobutyrate dehydrogenase is involved in both, valine and isoleucine degradation in Arabidopsis thaliana. Plant Physiology, 175(1), 51–61. https://doi.org/10.1104/pp.17.00649.
Schumacher, J. (2017). How light affects the life of Botrytis. Fungal Genetics and Biology, 106, 26–41. https://doi.org/10.1016/j.fgb.2017.06.002.
Selbmann, L., Egidi, E., Isola, D., Onofri, S., Zucconi, L., de Hoog, G. S., Chinaglia, S., Testa, L., Tosi, S., Balestrazzi, A., Lantieri, A., Compagno, R., & Varese, G. C. (2013). Biodiversity, evolution and adaptation of fungi in extreme environments. Plant Biosystems-An International Journal Dealing with all Aspects of Plant Biology, 147(1), 237–246. https://doi.org/10.1080/11263504.2012.753134.
Thabet, S., Simonet, F., Lemaire, M., Guillard, C., & Cotton, P. (2014). Impact of photocatalysis on fungal cells: Depiction of cellular and molecular effects on Saccharomyces cerevisiae. Applied and environmental microbiology, 80(24), 7527–7535. https://doi.org/10.1128/AEM.02416-14.
Veerasamy, R., Rajak, H., Jain, A., Sivadasan, S., Varghese, C. P., & Agrawal, R. K. (2011). Validation of QSAR models-strategies and importance. Int J Drug Des Discov, 3, 511–519.
Vijay, S., Renukadevi, P., Mangammal, P., & Krishnamoorthy, S. V. (2021). Effect of media, temperature and pH on the growth and pycnidial production of Lasiodiplodia theobromae (Pat.), causative of black root rot in mulberry. The Pharma Inn Journal, 10(11), 340–343. https://www.thepharmajournal.com/archives/%3Fyear=2021%26vol=10%26issue=11%26ArticleId=8668.
Worley, B., & Powers, R. (2013). Multivariate analysis in metabolomics. Current metabolomics, 1(1), 92–107.
Yu, Z., & Fischer, R. (2019). Light sensing and responses in fungi. Nature Reviews Microbiology, 17(1), 25–36. https://doi.org/10.1038/s41579-018-0109-x.
Yusef, H. M., & Allam, M. E. (1967). The effect of light on growth and sporulation of certain fungi. Mycopathologia et mycologia applicata, 33(2), 81–89. https://doi.org/10.1007/BF02053439.
Ząbek, A., Klimek-Ochab, M., Jawień, E., & Młynarz, P. (2017). Biodiversity in targeted metabolomics analysis of filamentous fugal pathogens by 1H NMR-based studies. World Journal of Microbiology and Biotechnology, 33, 132. https://doi.org/10.1007/s11274-017-2285-7.
Zaidi, K. U., Ali, A. S., Ali, S. A., & Naaz, I. (2014). Microbial tyrosinases: promising enzymes for pharmaceutical, food bioprocessing, and environmental industry. Biochemistry research international, 2014. https://doi.org/10.1155/2014/854687.
Zhang, Z., Han, X. M., Wei, J. H., Xue, J., Yang, Y., Liang, L., Li, X. J., Guo, Q. M., Xu, Y. H., & Gao, Z. H. (2014). Compositions and antifungal activities of essential oils from agarwood of Aquilaria sinensis (Lour.) Gilg induced by Lasiodiplodia theobromae (Pat.) Griffon. & Maubl. Journal of the Brazilian Chemical Society, 25, 20–26. https://doi.org/10.5935/0103-5053.20130263.
Zhang, H., Wang, G., Yang, Q., Yang, X., Zheng, Y., Liu, Y., & Xing, F. (2021). Effects of Light on the Ochratoxigenic Fungi Aspergillus ochraceus and A. carbonarius. Toxins, 13(4), 251. https://doi.org/10.3390/toxins13040251.
Author information
Authors and Affiliations
Contributions
ACC, SMAC, JRR, RRS constructed experimental design and prepared the manuscript; VAP prepared figures and tables; All authors reviewed and edted the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
This article does not contain any studies involving human or animal models experiment performed by any authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Crispim, A.C., Crispim, S.M.A., Rocha, J.R. et al. Light effects on Lasiodiplodia theobromae metabolome cultured in vitro. Metabolomics 19, 75 (2023). https://doi.org/10.1007/s11306-023-02041-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-023-02041-7