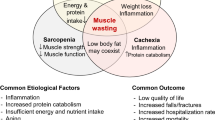
Abstract
The word sarcopenia derives from the Greek terms “sarx” for meat and “penia” for loss, thus being used to define reductions in muscle mass, muscle strength, and lower physical performance that compromise, mainly, the elderly population. Its high negative impact on patients’ quality of life encourages the production and publication of new studies that seek to find methods to prevent and reverse cases of loss of muscle mass and strength. Furthermore, the high prevalence of sarcopenia in patients with chronic kidney disease (CKD) is closely related to its pathophysiology, which consists of a state of increased protein catabolism and decreased muscle tissue synthesis. Also considering the inflammatory nature of CKD and sarcopenia, the purinergic system has been an important target of studies, which seek to relate it to the two previous conditions. This system achieves anti-inflammatory action by inhibiting, through adenosine, pro-inflammatory factors such as interleukin-12 (IL-12), tumor necrosis factor alpha (TNF-α), and nitric oxide (NO), as well as by releasing anti-inflammatory substances such as interleukin-10 (IL-10). Simultaneously, the purinergic system presents pro-inflammatory activity, signaled by adenosine triphosphate (ATP), which occurs through the activation of T cells and the release of pro-inflammatory factors such as those mentioned above. Therefore, the ability of this system to act on inflammatory processes can promote positive and negative changes in the clinical aspect of patients with CKD and/or sarcopenia. Furthermore, it appears that there is a correlation between the practice of repeated physical exercise with the clinical improvement and in the quality of life of these patients, presenting a decrease in the levels of C-reactive protein (CRP), NTPDase, and the pro-inflammatory cytokine IL-6, such as increases in IL-10 resulting from modulation of the purinergic system. In this way, the present article seeks to evaluate the effect of physical exercise as a modulator of the purinergic system in the control of sarcopenia in patients with CKD on hemodialysis, in order to trace a relationship that can bring benefits both for biological markers and for quality of life of these patients.





Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Moorthi RN, Avin KG (2017) Clinical relevance of sarcopenia in chronic kidney disease. Curr Opin Nephrol Hypertens. https://doi.org/10.1097/mnh.0000000000000318
Watanabe H, Enoki Y, Taruyama T (2019) Sarcopenia in chronic kidney disease: factors, mechanisms, and therapeutic interventions. Biol Pharm Bull. https://doi.org/10.1248/bpb.b19-00513
Yabuuchi J et al (2020) Association of advanced glycation end products with sarcopenia and frailty in chronic kidney disease. Sci Rep. https://doi.org/10.1038/s41598-020-74673-x
Dwyer KM, Kishore BK, Robson SC (2020) Conversion of extracellular ATP into adenosine: a master switch in renal health and disease. Nat Rev Nephrology. https://doi.org/10.1038/s41581-020-0304-7
Yang D, He Y, Muñoz-Planillo R, Liu Q, Núñez G (2015) Caspase-11 requires the pannexin-1 channel and the purinergic P2X7 pore to mediate pyroptosis and endotoxic shock. Immunity 43(5):923–32. https://doi.org/10.1016/j.immuni.2015.10.009
Mathur N, Pedersen BK (2008) Exercise as a mean to control low-grade systemic inflammation. Mediat Inflamm. https://doi.org/10.1155/2008/109502
Nunes RB, Alves JP, Kessler LP, Lago PD (2013) Aerobic exercise improves the inflammatory profile correlated with cardiac remodeling and function in chronic heart failure rats. Clinics. https://doi.org/10.6061/clinics/2013
Greaney JL, Wenner MM, Farquhar WB (2015) Exaggerated increases in blood pressure during isometric muscle contraction in hypertension: role for purinergic receptors. Auton Neurosci. https://doi.org/10.1016/j.autneu.2014.12.003
Miron V, Bottari N, Assmann C, Stefanello N, Costa P, Pelinson L, Reichert K, Silva A, Lopes T, Cruz I, Sévigny J, Morsch V, Schetinger M, Cardoso A (2019) Physical exercise prevents alterations in purinergic system and oxidative status in lipopolysaccharide-induced sepsis in rats. J Cell Biochem. https://doi.org/10.1002/jcb.27590
Sabatino A, Cuppari L, Stenvinke P, Lindholm B, Avesani CM (2021) Sarcopenia in chronic kidney disease: what have we learned so far? J Nephrol 34:1347–1372. https://doi.org/10.1007/s40620-020-00840-y
Wang X, Mitch W (2014) Mechanisms of muscle wasting in chronic kidney disease. Nat Rev Nephrol 10:504–516. https://doi.org/10.1038/nrneph.2014.112
Kamijo Y, Kanda E, Ishibashi Y, Yoshida M (2017). Sarcopenia and frailty in PD: impact on mortality, malnutrition, and inflammation. Perit Dial Int: J Int Soc Perit Dial https://doi.org/10.3747/pdi.2017.00271
Hirano T, Akira S, Taga T, Kishimoto T (1990) Biological and clinical aspects of interleukin 6. Immunol Today. https://doi.org/10.1016/0167-5699(90)90173-7
Tuttle C, Thang L, Maier A (2020) Markers of inflammation and their association with muscle strength and mass: a systematic review and meta-analysis. Ageing Res Rev. https://doi.org/10.1016/j.arr.2020.101185
Can B, Kara O, Kizilarslanoglu M, Arik G, Aycicek G, Sumer F, Civelek R, Demirtas UC, Ulger Z (2020) Serum markers of inflammation and oxidative stress in sarcopenia. Aging Clin Exp Res. https://doi.org/10.1007/s40520-016-0626-2
Chu W (2012) Tumor necrosis factor. Cancer Lett. https://doi.org/10.1016/j.canlet.2012.10.014
Malafrina V, Uriz-Otano F, Iniesta R, Gil-Guerrero L (2012) Sarcopenia in the elderly: diagnosis, physiopathology and treatment. Maturitas 71(2):109–114. https://doi.org/10.1016/j.maturitas.2011.11.012
Longo D Kasper D Harrison T (eds.) (2018). Harrison’s principles of internal medicine. New York, NY
Derbré F, Gratas-delamarche A, Ratas-delamarche A, Gómez-Cabrera M, Mari C, Vinã J (2012) Inactivity-induced oxidative stress: a central role in age-related sarcopenia? Eur J Sport Sci. https://doi.org/10.1080/17461391.2011.654268
Haskó G, Linden J, Cronstein B, Pacher P (2008) Adenosine receptors: therapeutic aspects for inflammatory and immune diseases. Nat Rev Drug Discov. https://doi.org/10.1038/nrd2638
Zhou Y, Hellberg M, Hellmark T, Höglund P, Clyne N (2021) Muscle mass and plasma myostatin after exercise training: a substudy of renal exercise (RENEXC)—a randomized controlled trial. Nephrol Dial Transplant. https://doi.org/10.1093/ndt/gfz210
Moon S, Kim T, Yoon S, Chung J, Hwang H (2015) Relationship between stage of chronic kidney disease and sarcopenia in Korean aged 40 years and older using the Korea National Health and Nutrition Examination Surveys. Plos One. https://doi.org/10.1371/journal.pone.0130740
Avin K, Moorthi R (2015) Bone is not alone: the effects of skeletal muscle dysfunction in chronic kidney disease. Curr Osteoporos. https://doi.org/10.1007/s11914-015-0261-4
Sawant A, Garland S, House A, Overend T (2011) Morphological, electrophysiological, and metabolic characteristics of skeletal muscle in people with end-stage renal disease: a critical review. Physiothe. https://doi.org/10.3138/ptc.2010-18
Ziaaldini M, Marzetti E, Picca A, Murlasitis Z (2017) Biochemical pathways of sarcopenia and their modulation by physical exercise: a narrative review. Front Med (Lausanne) 4:167. https://doi.org/10.3389/fmed.2017.00167
Fahrner C, Hackney A (1998) Effects of endurance exercise on free testosterone concentration and the binding affinity of sex hormone binding globulin. Sports Med. https://doi.org/10.1055/s-2007.971872
Kraemer W, Ratamess A (2005) Hormonal responses and adaptations to resistance exercise and training. Sports Med 35:339–361. https://doi.org/10.2165/00007256-200535040-00004
Virgilio F, Vuerich M (2015) Purinergic signaling in the immune system. Auton Neurosci. https://doi.org/10.1016/j.autneu.2015.04.011
Zyma M, Pawliczak R (2020) Characteristics and the role of purinergic receptors in pathophysiology with focus on immune response: purinergic receptors are widely expressed in immune cells and regulate high variety of their functions, taking part in pathogenesis of many diseases. Int Rev Immunol. https://doi.org/10.1080/088301.2020.1723582
Sharif K, Watad A, Bragazzi NL, Lichtbroun M, Amital H, Shoenfeld Y (2018) Physical activity and autoimmune diseases: get moving and manage the disease. Autoimmun Rev. https://doi.org/10.1016/j.autrev.2017.11.010
Schmidt T, van Mackelenbergh M, Wesch D, Mundhenke C (2017) Physical activity influences the immune system of breast cancer patients. J Cancer Res Ther. https://doi.org/10.4103/0973-1482.150356
Da Silveira MP, da Silva Fagundes KK, Bizuti MR, Starck E, Rossi RC, de Resende e, Silva DT, (2021) Physical exercise as a tool to help the immune system against COVID-19: an integrative review of the current literature. Clin Exp Med 21:15–28. https://doi.org/10.1007/s10238-020-00650-3
Vianna HR, Soares CMBM, Tavares MS, Teixeira MM, Silva ACS (2011) Inflamação na doença renal crônica: papel de citocinas. J Bras Nefrol. https://doi.org/10.1590/s0101-28002011000300012
Da Luz Scheffer D, Latini A (2020) Exercise-induced immune system response: anti-inflammatory status on peripheral and central organs. Biochimica et Biophysica Acta (BBA) - Mol Basis Dis https://doi.org/10.1016/j.bbadis.2020.165823.
Svensson M, Lexell J, Deierborg T (2015) Effects of physical exercise on neuroinflammation, neuroplasticity, neurodegeneration, and behavior: what we can learn from animal models in clinical settings. Neurorehabil Neural Repair 29(6):577–589. https://doi.org/10.1177/1545968314562108
Acanfora D, Scicchitano P, Casucci G, Lanzillo B, Capuano N, Furgi G.Ciccone M M. (2016) Exercise training effects on elderly and middle-aged patients with chronic heart failure after acute decompensation: a randomized, controlled trial. Int J Cardiol https://doi.org/10.1016/j.ijcard.2016.10.026
Cardoso AM, Silvério MNO, de Oliveira Maciel SFV (2021) Purinergic signaling as a new mechanism underlying physical exercise benefits: a narrative review. Purinergic Signal 17(4):649–679. https://doi.org/10.1007/s11302-021-09816-4
Cardoso A, Abdalla F, Bagatini M, Curry M, Zanini D, Roberta S, Augusto J, Rosa D, Morsch V, Chitolina M (2015) Swimming training prevents alterations in ecto-NTPDase and adenosine deaminase activities in lymphocytes from Nω-nitro-L-arginine methyl ester hydrochloride induced hypertension rats. J Hypertens 33(4):763–772. https://doi.org/10.1097/HJH.0000000000000468
Pedersen BK (2013) Muscle as a secretory organ. Compr Physiol 3. https://doi.org/10.1002/cphy.c120033
Wackerhage H, Schoenfeld BJ, Hamilton DL, Lehti M, Hulmi JJ (2019) Stimuli and sensors that initiate skeletal muscle hypertrophy following resistance exercise. J Appl Physiol 126(1):30–43. https://doi.org/10.1152/japplphysiol.00685.2018
Lee JH, Jun HS (2019) (2019) Role of myokines in regulating skeletal muscle mass and function. Front Physiol 30:10. https://doi.org/10.3389/fphys.2019.00042
Burnstock G (2018) The therapeutic potential of purinergic signalling. Biochem Pharmacol. https://doi.org/10.1016/j.bcp.2017.07.016
Domingos LB (2020) O envolvimento do sistema purinérgico na formação e extinção da memória aversiva condicionada em camundongos. Universidade de São Paulo, Dissertação
Cardoso A, Bagatini M, Martins C, Martins C, Abdlla F, Zanini D, Gutierris R, Pimentel V, Thome G, Leal C, Vieira J, Stefanello N, Fiorin F, Baldissareli J, Royes L, Morsch V, Schetinger M (2012) Exercise training prevents ecto-nucleotidases alterations in platelets of hypertensive rats. Mol Cell Biochem. https://doi.org/10.1007/s11010-012-1431-7
Monaghan et al (2021) Biochem Pharmacol 187:114389. https://doi.org/10.1016/j.bcp.2020.114389
Di Virgilio F, Sarti AC, Coutinho-Silva R (2020) Purinergic signaling, DAMPs, and inflammation. Am J Physiol Cell Physiol. https://doi.org/10.1152/ajpcell.00053.2020
Lammers MD, Anéli NM, de Oliveira GG, Maciel SFVO, Zanini D, Manica A, de Resende e Silva DT, Bagatini MD, Sévigny J, de Sá CA, Manfredi LH, Cardoso AM (2020) The anti-inflammatory effect of resistance training in hypertensive women: the role of purinergic signaling. J Hypertens 38(12):2490–2500. https://doi.org/10.1097/HJH.0000000000002578
Ferrari D, McNamee E, Idzko M, Gamabari R, Etlzschig (2016) Purinergic signaling during immune cell trafficking. Trends Immunol. https://doi.org/10.1016/j.it.2016.04.004
Menzies R, Unwin R, Bailey A (2014) Renal P2 receptors and hypertension. Acta Physiol 213:232–241. https://doi.org/10.1111/apha.12412
Bailey M, Hilman K, Unwin R (2000) P2 receptors in the kidney. J Auton Nervous System 1–3:264–270. https://doi.org/10.1016/S0165-1838(00)00125-9
Menzies R, Tam F, Unwin R, Bailey A (2016) Purinergic signalling in kidney disease. Kidney Int 91:2-315–323 https://doi.org/10.1016/j.kint.2016.08.029
Wilkinson T, Shur N, Smith A (2016) Exercise as medicine in chronic kidney disease. Scandinavian J Med Sci Sports. https://doi.org/10.1111/sms.12714
Funding
FAPESC-Research Support Foundation of the State of Santa Catarina.
Author information
Authors and Affiliations
Contributions
G.V.S.S., M.R.B., and D.T.R.S. contributed to the conception, project administration, and supervision. N.R.O., L.Z.M.L., V.C.G.S.A., A.C.G.Z., C.Z., and J.G.P. contributed to the formal analysis, investigation, and methodology. D.T.R.S. supervised the methodology. G.V.S.S., M.R.B., and F.B.H. contributed to the writing and translation of the original draft. M.R.B. and D.T.R.S. revised the final work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflicts of interest
The authors declare no competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Resende e Silva, D.T., Bizuti, M.R., de Oliveira, N.R. et al. Physical exercise as a modulator of the purinergic system in the control of sarcopenia in individuals with chronic kidney disease on hemodialysis. Purinergic Signalling (2023). https://doi.org/10.1007/s11302-023-09950-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11302-023-09950-1