Abstract
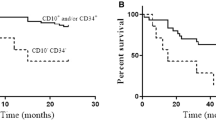
The risk stratification of B-acute lymphoblastic leukemia (B-ALL) is based on clinical and biological factors. However, B-ALL has significant biological and clinical heterogeneity and 50% of B-ALL patients do not have defined prognostic markers. In this sense, the identification of new prognostic biomarkers is necessary. Considering different cohorts of childhood B-ALL patients, gene (DPP4/CD38/ENTPD1/NT5E) and protein (CD38/CD39/CD73) expressions of ectonucleotidases were analyzed in silico and ex vivo and the association with prognosis was established. In univariate analyses, expression of NT5E was significantly associated with worse progression-free survival (PFS) in bone marrow (BM) samples. In multivariate analyses, Kaplan–Meier analysis, and log-rank test, higher NT5E expression predicted unfavorable PFS in BM samples. Considering minimal residual disease (MRD), higher levels of cellularity were associated with the high NT5E expression at day 8 of induction therapy. In addition, we observed that white blood cells (WBC) of childhood B-ALL patients had more CD38 compared to the same cell population of healthy donors (HD). In fact, MRD > 0.1% patients had higher CD38 protein expression on WBC in comparison to HD. Noteworthy, we observed higher CD38 expression on WBC than blasts in MRD > 0.1% patients. We suggest that NT5E gene and CD38 protein expression, of the ectonucleotidases family, could provide interesting prognostic biomarkers for childhood B-ALL.






Similar content being viewed by others
Data Availability
All data are available upon reasonable request.
References
Carroll W, Loh M, Biondi A, Willman C (2011) Childhood leukemia. 29–62. https://doi.org/10.1007/978-3-642-13781-5
Moorman AV (2016) New and emerging prognostic and predictive genetic biomarkers in B-cell precursor acute lymphoblastic leukemia. Haematologica 101:407–416. https://doi.org/10.3324/haematol.2015.141101
Roberts KG, Mullighan CG (2015) Genomics in acute lymphoblastic leukaemia: insights and treatment implications. Nat Rev Clin Oncol 12:344–357. https://doi.org/10.1038/nrclinonc.2015.38
Zhang X, Rastogi P, Shah B, Zhang L (2017) B lymphoblastic leukemia/lymphoma: new insights into genetics, molecular aberrations, subclassification and targeted therapy. Oncotarget 8:66728–66741. https://doi.org/10.18632/oncotarget.19271
Hann I, Vora A, Richards S et al (2000) Benefit of intensified treatment for all children with acute lymphoblastic leukaemia: results from MRC UKALL XI and MRC ALL97 randomised trials. Leukemia 14:356–363. https://doi.org/10.1038/sj.leu.2401704
Schultz KR, Pullen DJ, Sather HN et al (2007) Risk- and response-based classification of childhood B-precursor acute lymphoblastic leukemia: a combined analysis of prognostic markers from the Pediatric Oncology Group (POG) and Children’s Cancer Group (CCG). Blood 109:926–935. https://doi.org/10.1182/blood-2006-01-024729
Carroll WL, Raetz EA (2012) Clinical and laboratory biology of childhood acute lymphoblastic leukemia. J Pediatr 160:10–18. https://doi.org/10.1016/j.jpeds.2011.08.006
Bartram J, Patel B, Fielding AK (2020) Monitoring MRD in ALL: methodologies, technical aspects and optimal time points for measurement. Semin Hematol 57:142–148. https://doi.org/10.1053/j.seminhematol.2020.06.003
Schuurhuis GJ, Heuser M, Freeman S et al (2018) Minimal/measurable residual disease in AML: a consensus document from the European LeukemiaNet MRD Working Party. Blood 131:1275–1291. https://doi.org/10.1182/blood-2017-09-801498
Garza-Veloz I, Martinez-Fierro ML, Jaime-Perez JC et al (2015) Identification of differentially expressed genes associated with prognosis of B acute lymphoblastic leukemia. Dis Markers 2015:1–11. https://doi.org/10.1155/2015/828145
Bungaro S, Dell’Orto MC, Zangrando A et al (2009) Integration of genomic and gene expression data of childhood ALL without known aberrations identifies subgroups with specific genetic hallmarks. Genes Chromosom Cancer 48:22–38. https://doi.org/10.1002/gcc.20616
Dawson AJ, Yanofsky R, Vallente R et al (2011) Array comparative genomic hybridization and cytogenetic analysis in pediatric acute leukemias. Curr Oncol 18:e210. https://doi.org/10.3747/co.v18i5.770
Yeoh E, Ross ME, Shurtleff SA et al (2002) Classification, subtype discovery, and prediction of outcome in pediatric acute lymphoblastic leukemia by gene expression profiling. Cancer Cell 1:133–143. https://doi.org/10.1016/S1535-6108(02)00032-6
Burnstock G (2007) Purine and pyrimidine receptors. Cell Mol Life Sci 64:1471–1483. https://doi.org/10.1007/s00018-007-6497-0
Careta O, Cuevas E, Muñoz-Esquerre M et al (2019) Imbalance in the expression of genes associated with purinergic signalling in the lung and systemic arteries of COPD patients. Sci Rep 9:1–9. https://doi.org/10.1038/s41598-019-39233-y
Chang CC, Liu CZ, Cleveland RP (2003) Relative importance of CD38 expression over myeloid-associated markers expression in predicting the clinical course of B-CLL patients. Leuk Lymphoma 44:977–982. https://doi.org/10.1080/1042819031000076990
Zimmermann H (1996) Biochemistry, localization and functional roles of ecto-nucleotidases in the nervous system. Prog Neurobiol 49:589–618. https://doi.org/10.1016/0301-0082(96)00026-3
Yegutkin GG, Henttinen T, Samburski SS et al (2002) The evidence for two opposite, ATP-generating and ATP-consuming, extracellular pathways on endothelial and lymphoid cells. Biochem J 367:121–128. https://doi.org/10.1042/BJ20020439
Horenstein AL, Chillemi A, Zaccarello G, et al (2013) A CD38/CD203a/CD73 ectoenzymatic pathway independent of CD39 drives a novel adenosinergic loop in human T lymphocytes. Oncoimmunology 2https://doi.org/10.4161/ONCI.26246
Di Virgilio F, Boeynaems J-M, Robson SC (2009) Extracellular nucleotides as negative modulators of immunity. Curr Opin Pharmacol 9:507–513. https://doi.org/10.1016/j.coph.2009.06.021
Abousamra NK, Salah El-Din M, Hamza Elzahaf E, Esmael ME (2015) Ectonucleoside triphosphate diphosphohydrolase-1 (E-NTPDase1/CD39) as a new prognostic marker in chronic lymphocytic leukemia. Leuk Lymphoma 56:113–119. https://doi.org/10.3109/10428194.2014.907893
Lu X, Chen Y, Feng B et al (2013) Expression and clinical significance of CD73 and hypoxia-inducible factor-1α in gastric carcinoma. World J Gastroenterol 19:1912. https://doi.org/10.3748/WJG.V19.I12.1912
Zhang B, Song B, Wang X et al (2015) The expression and clinical significance of CD73 molecule in human rectal adenocarcinoma. Tumor Biol 36:5459–5466. https://doi.org/10.1007/s13277-015-3212-x
Liang P, Yeh B, Li W et al (2017) DPP4/CD26 overexpression in urothelial carcinoma confers an independent prognostic impact and correlates with intrinsic biological aggressiveness. Oncotarget 8:2995–3008. https://doi.org/10.18632/oncotarget.13820
Keyhani A, Huh YOO, Jendiroba D et al (2000) Increased CD38 expression is associated with favorable prognosis in adult acute leukemia. Leuk Res 24:153–159. https://doi.org/10.1016/S0145-2126(99)00147-2
Kalina T, Flores-Montero J, Van Der Velden VHJ et al (2012) EuroFlow standardization of flow cytometer instrument settings and immunophenotyping protocols. Leukemia 26:1986–2010. https://doi.org/10.1038/leu.2012.122
Therneau TM, Lumley T (2020) Survival analysis; [R package “survival” version 3.1–12]. In: Compr. R Arch. Netw. https://cran.r-project.org/package=survival. Accessed 28 Jul 2020
Iqbal J, Zaidi M (2006) Extracellular NAD+ metabolism modulates osteoclastogenesis. Biochem Biophys Res Commun 349:533–539. https://doi.org/10.1016/j.bbrc.2006.08.108
Campos-Contreras ADR, Díaz-Muñoz M, Vázquez-Cuevas FG (2020) Purinergic signaling in the hallmarks of cancer. Cells 9:1612. https://doi.org/10.3390/cells9071612
Allard B, Beavis PA, Darcy PK, Stagg J (2016) Immunosuppressive activities of adenosine in cancer. Curr Opin Pharmacol 29:7–16. https://doi.org/10.1016/j.coph.2016.04.001
de Andrade MP, Coutinho-Silva R, Savio LEB (2017) Multifaceted effects of extracellular adenosine triphosphate and adenosine in the tumor–host interaction and therapeutic perspectives. Front Immunol 8:1526. https://doi.org/10.3389/fimmu.2017.01526
Jiang T, Xu X, Qiao M et al (2018) (2018) Comprehensive evaluation of NT5E/CD73 expression and its prognostic significance in distinct types of cancers. BMC Cancer 181(18):1–10. https://doi.org/10.1186/S12885-018-4073-7
Tripathi A, Lin E, Xie W, et al (2020) Prognostic significance and immune correlates of CD73 expression in renal cell carcinoma. J Immunother Cancer 8https://doi.org/10.1136/jitc-2020-001467
Jeong YM, Cho H, Kim T-M et al (2020) CD73 Overexpression promotes progression and recurrence of papillary thyroid carcinoma. Cancers (Basel) 12:3042. https://doi.org/10.3390/cancers12103042
Sakhinia E, Farahangpour M, Tholouli E et al (2006) Comparison of gene-expression profiles in parallel bone marrow and peripheral blood samples in acute myeloid leukaemia by real-time polymerase chain reaction. J Clin Pathol 59:1059–1065. https://doi.org/10.1136/jcp.2005.031161
Flotho C, Coustan-Smith E, Pei D et al (2006) Genes contributing to minimal residual disease in childhood acute lymphoblastic leukemia: prognostic significance of CASP8AP2. Blood 108:1050–1057. https://doi.org/10.1182/blood-2006-01-0322
Wieten E, van der Linden-Schrever MBE, Sonneveld E et al (2011) CD73 (5′-nucleotidase) expression has no prognostic value in children with acute lymphoblastic leukemia. Leukemia. https://doi.org/10.1038/leu.2011.174
Wang W, Gao L, Li Y et al (2016) The application of CD73 in minimal residual disease monitoring using flow cytometry in B-cell acute lymphoblastic leukemia. Leuk Lymphoma 57:1174–1181. https://doi.org/10.3109/10428194.2015.1070153
Koehler M, Behm F, Hancock M, Pui CH (1993) Expression of activation antigens CD38 and CD71 is not clinically important in childhood acute lymphoblastic leukemia. Leukemia 7(1):41–5 (PMID: 8418378)
C L, DY S, XJ X, et al (2020) High CD38 expression in childhood T-cell acute lymphoblastic leukemia is not associated with prognosis. Cancer Biomark 27:277–284https://doi.org/10.3233/CBM-190946
Morandi F, Horenstein AL, Costa F et al (2018) CD38: a target for immunotherapeutic approaches in multiple myeloma. Front Immunol 0:2722. https://doi.org/10.3389/FIMMU.2018.02722
Sek K, Mølck C, Stewart G et al (2018) Targeting adenosine receptor signaling in cancer immunotherapy. Int J Mol Sci 19:3837. https://doi.org/10.3390/ijms19123837
Hammami A, Allard D, Allard B, Stagg J (2019) Targeting the adenosine pathway for cancer immunotherapy. Semin Immunol 42:101304. https://doi.org/10.1016/J.SMIM.2019.101304
Malavasi F, Deaglio S, Funaro A et al (2008) Evolution and function of the ADP ribosyl cyclase/CD38 gene family in physiology and pathology. Physiol Rev 88:841–886. https://doi.org/10.1152/physrev.00035.2007
Tembhare R, Sriram H, Khanka T et al (2020) Flow cytometric evaluation of CD38 expression levels in the newly diagnosed T-cell acute lymphoblastic leukemia and the effect of chemotherapy on its expression in measurable residual disease, refractory disease and relapsed disease: an implication for anti-CD38 immunotherapy. J Immunother Cancer 8:630. https://doi.org/10.1136/jitc-2020-000630
Silverman LB, Sallan SE (2003) Newly diagnosed childhood acute lymphoblastic leukemia: update on prognostic factors and treatment. Curr Opin Hematol 10:290–296. https://doi.org/10.1097/00062752-200307000-00007
Pui C-H, Campana D, Evans WE (2001) Childhood acute lymphoblastic leukaemia – current status and future perspectives. Lancet Oncol 2:597–607. https://doi.org/10.1016/S1470-2045(01)00516-2
Funding
This study was supported by CNPq (PQ no. 302879/2017–0, AMO Battastini grant), FAPERGS (FAPERGS/PQG grant no. 19/2551–0001783-9; ARD grant no. 19/2551–0001269-1 and PRONEX no. 16–2551-0000467–6), and INCT/CNPq/CAPES/ FAPERGS grant no. 465671/2014–4. Fundo de Incentivo a Pesquisa do Hospital de Clínicas de Porto Alegre (project no.: 2018–0401; CAAE no.: 93973218110015327). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior-Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Vitória Brum da Silva Nunes, Camila Kehl Dias, Marco Antônio De Bastiani, and Fabrício Figueiró. The first draft of the manuscript was written by Vitória Brum da Silva Nunes and all authors commented on later versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
The study was conducted in accordance with the 1964 Helsinki Declaration, and the protocol was approved by the Ethics Committee of the Hospital de Clínicas de Porto Alegre (Project number: 2018–0401; CAAE number: 93973218110015327). All guardians of the children provided informed consent.
Conflicts of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Silva Nunes, V.B., Dias, C.K., De Bastiani, M.A. et al. NT5E gene and CD38 protein as potential prognostic biomarkers for childhood B-acute lymphoblastic leukemia. Purinergic Signalling 18, 211–222 (2022). https://doi.org/10.1007/s11302-022-09841-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-022-09841-x