Abstract
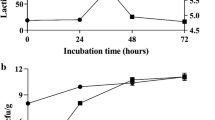
Corn germ meal (CGM) is one of the major byproducts of corn starch extraction. Although CGM has rich fiber content, it lacks good protein content and amino acid balance, and therefore cannot be fully utilized as animal feed. In this study, we investigated the processing effect of cellulase synergized with Bacillus velezensis on the nutritional value of pretreated CGM (PCGM) in two-stage solid-state fermentation (SSF). High-throughput sequencing technology was used to explore the dynamic changes in microbial diversity. The results showed that compared with four combinations of B. velezensis + Lactiplantibacillus plantarum (PCGM-BL), cellulase + L. plantarum (PCGM-CL),control group (PCGM-CK), and cellulase + B. velezensis + L. plantarum (PCGM-BCL), the fourth combination of PCGM-BCL significantly improved the nutritional characteristics of PCGM. After two-stage SSF (48 h), viable bacterial count and contents of crude protein (CP) and trichloroacetic acid-soluble protein (TCA-SP) all were increased in PCGM-BCL (p < 0.05), while the pH was reduced to 4.38 ± 0.02. In addition, compared with PCGM-BL, the cellulose degradation rate increased from 5.02 to 50.74%, increasing the amounts of short-chain fatty acids (216.61 ± 2.74 to 1727.55 ± 23.00 µg/g) and total amino acids (18.60 to 21.02%) in PCGM-BCL. Furthermore, high-throughput sequencing analysis revealed significant dynamic changes in microbial diversity. In the first stage of PCGM-BCL fermentation, Bacillus was the dominant genus (99.87%), which after 24 h of anaerobic fermentation changed to lactobacillus (37.45%). Kyoto Encylopaedia of Genes and Genomes (KEGG) metabolic pathway analysis revealed that the pathways related to the metabolism of carbohydrates, amino acids, cofactors, and vitamins accounted for more than 10% of the enriched pathways throughout the fermentation period. Concisely, we show that cellulase can effectively improve the nutritional value of PCGM when synergized with B. velezensis in two-stage SSF.




Similar content being viewed by others
Data availability
All data analyzed during this study are included in this manuscript and additional material.
References
Agama-Acevedo E, Rendón-Villalobos R, Tovar J, Trejo-Estrada SR, Bello-Pérez LA (2005) Effect of storage time on in vitro digestion rate and resistant starch content of tortillas elaborated from commercial corn masas. Arch Latinoam Nutr 55(1):86–92
Arevalo-Villena M, Briones-Perez A, Corbo MR, Sinigaglia M, Bevilacqua A (2017) Biotechnological application of yeasts in food science: starter cultures, probiotics and enzyme production. J Appl Microbiol 123(6):1360–1372. https://doi.org/10.1111/jam.13548
Bolyen E, Rideout JR, Dillon MR, Bokulich NA, Abnet CC, Al-Ghalith GA, Alexander H, Alm EJ, Arumugam M, Asnicar F, Bai Y, Bisanz JE, Bittinger K, Brejnrod A, Brislawn CJ, Brown CT, Callahan BJ, Caraballo-Rodríguez AM, Chase J, Cope EK, Da Silva R, Diener C, Dorrestein PC, Douglas GM, Durall DM, Duvallet C, Edwardson CF, Ernst M, Estaki M, Fouquier J, Gauglitz JM, Gibbons SM, Gibson DL, Gonzalez A, Gorlick K, Guo J, Hillmann B, Holmes S, Holste H, Huttenhower C, Huttley GA, Janssen S, Jarmusch AK, Jiang L, Kaehler BD, Kang KB, Keefe CR, Keim P, Kelley ST, Knights D, Koester I, Kosciolek T, Kreps J, Langille MGI, Lee J, Ley R, Liu YX, Loftfield E, Lozupone C, Maher M, Marotz C, Martin BD, McDonald D, McIver LJ, Melnik AV, Metcalf JL, Morgan SC, Morton JT, Naimey AT, Navas-Molina JA, Nothias LF, Orchanian SB, Pearson T, Peoples SL, Petras D, Preuss ML, Pruesse E, Rasmussen LB, Rivers A, Robeson MS 2nd, Rosenthal P, Segata N, Shaffer M, Shiffer A, Sinha R, Song SJ, Spear JR, Swafford AD, Thompson LR, Torres PJ, Trinh P, Tripathi A, Turnbaugh PJ, Ul-Hasan S, van der Hooft JJJ, Vargas F, Vázquez-Baeza Y, Vogtmann E, von Hippel M, Walters W, Wan Y, Wang M, Warren J, Weber KC, Williamson CHD, Willis AD, Xu ZZ, Zaneveld JR, Zhang Y, Zhu Q, Knight R, Caporaso JG (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat Biotechnol 37(8):852–857. https://doi.org/10.1038/s41587-019-0209-9
Callahan BJ, McMurdie PJ, Rosen MJ, Han AW, Johnson AJ, Holmes SP (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13(7):581–583. https://doi.org/10.1038/nmeth.3869
Chen L, Gu W, Xu H-y, Yang G-L, Shan X-F, Chen G, Kang Y-h, Wang C-F, Qian A-D (2018a) Comparative genome analysis of Bacillus velezensis reveals a potential for degrading lignocellulosic biomass. 3 Biotech 8(5):253. https://doi.org/10.1007/s13205-018-1270-7
Chen L, Gu W, Xu HY, Yang GL, Shan XF, Chen G, Wang CF, Qian AD (2018b) Complete genome sequence of Bacillus velezensis 157 isolated from Eucommia ulmoides with pathogenic bacteria inhibiting and lignocellulolytic enzymes production by SSF. 3 Biotech 8(2):114. https://doi.org/10.1007/s13205-018-1125-2
Chen L, Zhao Z, Yu W, Zheng L, Li L, Gu W, Xu H, Wei B, Yan X (2021) Nutritional quality improvement of soybean meal by Bacillus velezensis and Lactobacillus plantarum during two-stage solid- state fermentation. AMB Express 11(1):23. https://doi.org/10.1186/s13568-021-01184-x
Chen L, Chen W, Zheng B, Yu W, Zheng L, Qu Z, Yan X, Wei B, Zhao Z (2022) Fermentation of NaHCO3-treated corn germ meal by Bacillus velezensis CL-4 promotes lignocellulose degradation and nutrient utilization. Appl Microbiol Biotechnol 106(18):6077–6094. https://doi.org/10.1007/s00253-022-12130-7
Chen L, Qu Z, Yu W, Zheng L, Qiao H, Wang D, Wei B, Zhao Z (2023) Comparative genomic and transcriptome analysis of Bacillus velezensis CL-4 fermented corn germ meal. AMB Express 13(1):10. https://doi.org/10.1186/s13568-023-01510-5
de Vos WM, Tilg H, Van Hul M, Cani PD (2022) Gut microbiome and health: mechanistic insights. Gut 71(5):1020–1032. https://doi.org/10.1136/gutjnl-2021-326789
Evans NM, Shao S (2022) Mycotoxin metabolism by edible insects. Toxins. https://doi.org/10.3390/toxins14030217
Franklin MA, Mathew AG, Vickers JR, Clift RA (2002) Characterization of microbial populations and volatile fatty acid concentrations in the jejunum, ileum, and cecum of pigs weaned at 17 vs 24 days of age. J Anim Sci 80(11):2904–2910
Frisvad JC, Møller LLH, Larsen TO, Kumar R, Arnau J (2018) Safety of the fungal workhorses of industrial biotechnology: update on the mycotoxin and secondary metabolite potential of aspergillus Niger, aspergillus oryzae, and Trichoderma reesei. Appl Microbiol Biotechnol 102(22):9481–9515. https://doi.org/10.1007/s00253-018-9354-1
Gilbert ER, Wong EA, Webb KE Jr (2008) Peptide absorption and utilization: implications for animal nutrition and health. J Anim Sci 86(9):2135–2155. https://doi.org/10.2527/jas.2007-0826
Hedayati MT, Pasqualotto AC, Warn PA, Bowyer P, Denning DW (2007) Aspergillus flavus: human pathogen, allergen and mycotoxin producer. Microbiol 153:1677–1692. https://doi.org/10.1099/mic.0.2007/007641-0
Huang Q, Su YB, Li DF, Liu L, Huang CF, Zhu ZP, Lai CH (2015) Effects of inclusion levels of wheat bran and body weight on ileal and fecal digestibility in growing pigs. Asian-Australasian J Anim Sci 28(6):847–854. https://doi.org/10.5713/ajas.14.0769
Ibrahim S, Al-Saryi N, Al-Kadmy IMS, Aziz SN (2021) Multidrug-resistant Acinetobacter baumannii as an emerging concern in hospitals. Mol Biol Rep 48(10):6987–6998. https://doi.org/10.1007/s11033-021-06690-6
Jaworski NW, Lærke HN, Bach Knudsen KE, Stein HH (2015) Carbohydrate composition and in vitro digestibility of dry matter and nonstarch polysaccharides in corn, sorghum, and wheat and coproducts from these grains1. J Anim Sci 93(3):1103–1113. https://doi.org/10.2527/jas.2014-8147
Ji F, Li H, Xu J, Shi J (2011) Enzyme-linked immunosorbent-assay for Deoxynivalenol (DON). Toxins 3(8):968–978. https://doi.org/10.3390/toxins3080968
Kaeuffer C, Baldacini M, Ruge T, Ruch Y, Zhu YJ, De Cian M, Philouze G, Bachellier P, Denis J, Lefebvre N, Schneider F, Hansmann Y, Letscher-Bru V, Herbrecht R, Sabou M, Danion F (2022) Fungal Infections caused by Kazachstania spp., Strasbourg, France, 2007–2020. Emerg Infect Dis 28(1):29–34. https://doi.org/10.3201/eid2801.211543
Katoh K, Misawa K, Kuma K, Miyata T (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res 30(14):3059–3066. https://doi.org/10.1093/nar/gkf436
Kechin A, Boyarskikh U, Kel A, Filipenko M (2017) cutPrimers: a new tool for accurate cutting of primers from reads of targeted next generation sequencing. J Comput Biology: J Comput Mol cell Biology 24(11):1138–1143. https://doi.org/10.1089/cmb.2017.0096
Khalid F, Khalid A, Fu Y, Hu Q, Zheng Y, Khan S, Wang Z (2021) Potential of Bacillus velezensis as a probiotic in animal feed: a review. J Microbiol 59(7):627–633. https://doi.org/10.1007/s12275-021-1161-1
Lee JW, McKeith FK, Stein HH (2012) Up to 30% corn germ may be included in diets fed to growing–finishing pigs without affecting pig growth performance, carcass composition, or pork fat quality1. J Anim Sci 90(13):4933–4942. https://doi.org/10.2527/jas.2012-5129
Leong SL, Lantz H, Pettersson OV, Frisvad JC, Thrane U, Heipieper HJ, Dijksterhuis J, Grabherr M, Pettersson M, Tellgren-Roth C, Schnürer J (2015) Genome and physiology of the ascomycete filamentous fungus Xeromyces Bisporus, the most xerophilic organism isolated to date. Environ Microbiol 17(2):496–513. https://doi.org/10.1111/1462-2920.12596
Li Y, Li Z, Liu H, Noblet J, Liu L, Li D, Wang F, Lai C (2018) Net energy content of rice bran, corn germ meal, corn gluten feed, peanut meal, and sunflower meal in growing pigs. Asian-Australasian J Anim Sci 31(9):1481–1490. https://doi.org/10.5713/ajas.17.0829
Liu Y, Song M, Almeida FN, Tilton SL, Cecava MJ, Stein HH (2014) Energy concentration and amino acid digestibility in corn and corn coproducts from the wet-milling industry fed to growing pigs. J Anim Sci 92(10):4557–4565. https://doi.org/10.2527/jas.2014-6747
Liu J, Long Z, Xue L, Li C (2022) First report of Didymella sinensis causing leaf blight on Italian ryegrass in China. Plant Dis. https://doi.org/10.1094/pdis-08-22-1831-pdn
Lymperopoulos A, Suster MS, Borges JI (2022) Short-chain fatty acid receptors and cardiovascular function. Int J Mol Sci. https://doi.org/10.3390/ijms23063303
Mat DJL, Cattenoz T, Souchon I, Michon C, Le Feunteun S (2018) Monitoring protein hydrolysis by pepsin using pH-stat: in vitro gastric digestions in static and dynamic pH conditions. Food Chem 239:268–275. https://doi.org/10.1016/j.foodchem.2017.06.115
Medeiros S, Xie J, Dyce PW, Cai HY, DeLange K, Zhang H, Li J (2018) Isolation of bacteria from fermented food and grass carp intestine and their efficiencies in improving nutrient value of soybean meal in solid state fermentation. J Anim Sci Biotechnol 9:29. https://doi.org/10.1186/s40104-018-0245-1
Metges CC (2000) Contribution of microbial amino acids to amino acid homeostasis of the host. J Nutr. https://doi.org/10.1093/jn/130.7.1857S
Morrison DJ, Preston T (2016) Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes 7(3):189–200. https://doi.org/10.1080/19490976.2015.1134082
Okuma TA, Huynh TP, Hellberg RS (2018) Use of enzyme-linked immunosorbent assay to screen for aflatoxins, ochratoxin A, and deoxynivalenol in dry pet foods. Mycotoxin Res 34(1):69–75. https://doi.org/10.1007/s12550-017-0300-3
Olukomaiya O, Fernando C, Mereddy R, Li X, Sultanbawa Y (2019) Solid-state fermented plant protein sources in the diets of broiler chickens: a review. Anim Nutr 5(4):319–330. https://doi.org/10.1016/j.aninu.2019.05.005
Parada Venegas D, De la Fuente MK, Landskron G, González MJ, Quera R, Dijkstra G, Harmsen HJM, Faber KN, Hermoso MA (2019) Short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel Diseases. Front Immunol 10:277. https://doi.org/10.3389/fimmu.2019.00277
Rigatti SJ (2017) Random Forest. J Insur Med 47(1):31–39. https://doi.org/10.17849/insm-47-01-31-39.1
Sakamoto K, Asano T, Furuya A, Takahashi S (1980) Estimation of in vivo digestibility with the laying hen by an in vitro method using the intestinal fluid of the pig. Br J Nutr 43(2):389–391. https://doi.org/10.1079/bjn19800103
Sánchez ÓJ, Ospina DA, Montoya S (2017) Compost supplementation with nutrients and microorganisms in composting process. Waste Manag 69:136–153. https://doi.org/10.1016/j.wasman.2017.08.012
Shi C, He J, Yu J, Yu B, Mao X, Zheng P, Huang Z, Chen D (2015) Amino acid, phosphorus, and energy digestibility of Aspergillus Niger fermented rapeseed meal fed to growing pigs. J Anim Sci 93(6):2916–2925. https://doi.org/10.2527/jas.2014-8326
Shi C, Zhang Y, Lu Z, Wang Y (2017) Solid-state fermentation of corn-soybean meal mixed feed with Bacillus subtilis and Enterococcus faecium for degrading antinutritional factors and enhancing nutritional value. J Anim Sci Biotechnol 8:50. https://doi.org/10.1186/s40104-017-0184-2
Shi M, Liu Z, Wang H, Shi C, Liu L, Wang J, Li D, Zhang S (2019) Determination and prediction of the digestible and metabolizable energy contents of corn germ meal in growing pigs. Asian-Australasian J Anim Sci 32(3):405–412. https://doi.org/10.5713/ajas.17.0891
Shin NR, Whon TW, Bae JW (2015) Proteobacteria: microbial signature of dysbiosis in gut microbiota. Trends Biotechnol 33(9):496–503. https://doi.org/10.1016/j.tibtech.2015.06.011
Stevenson A, Hamill PG, Dijksterhuis J, Hallsworth JE (2017) Water-, pH- and temperature relations of germination for the extreme xerophiles Xeromyces Bisporus (FRR 0025), aspergillus penicillioides (JH06THJ) and Eurotium Halophilicum (FRR 2471). Microb Biotechnol 10(2):330–340. https://doi.org/10.1111/1751-7915.12406
Stokes JL, Gunness M (1946) The amino acid composition of microorganisms. J Bacteriol 51:570
Su W, Jiang Z, Hao L, Li W, Gong T, Zhang Y, Du S, Wang C, Lu Z, Jin M, Wang Y (2021) Variations of soybean meal and corn mixed substrates in physicochemical characteristics and microbiota during two-stage solid-state fermentation. Front Microbiol 12:688839. https://doi.org/10.3389/fmicb.2021.688839
Su W, Jiang Z, Wang C, Xu B, Lu Z, Wang F, Zong X, Jin M, Wang Y (2022) Dynamics of defatted rice bran in physicochemical characteristics, microbiota and metabolic functions during two-stage co-fermentation. Int J Food Microbiol 362:109489. https://doi.org/10.1016/j.ijfoodmicro.2021.109489
Urriola PE, Shurson GC, Stein HH (2010) Digestibility of dietary fiber in distillers coproducts fed to growing pigs. J Anim Sci 88(7):2373–2381. https://doi.org/10.2527/jas.2009-2227
Wang C, Shi C, Zhang Y, Song D, Lu Z, Wang Y (2018) Microbiota in fermented feed and swine gut. Appl Microbiol Biotechnol 102(7):2941–2948. https://doi.org/10.1007/s00253-018-8829-4
Wang C, Shi C, Su W, Jin M, Xu B, Hao L, Zhang Y, Lu Z, Wang F, Wang Y, Du H (2020) Dynamics of the physicochemical characteristics, microbiota, and metabolic functions of soybean meal and corn mixed substrates during two-stage solid-state fermentation. mSystems 5(1):10–1128
Yang Y, Kiarie E, Slominski BA, Brûlé-Babel A, Nyachoti CM (2010) Amino acid and fiber digestibility, intestinal bacterial profile, and enzyme activity in growing pigs fed dried distillers grains with solubles-based diets. J Anim Sci 88(10):3304–3312. https://doi.org/10.2527/jas.2009-2318
Yao Y, Long M (2020) The biological detoxification of deoxynivalenol: a review. Food Chem Toxicol 145:111649. https://doi.org/10.1016/j.fct.2020.111649
Yuan SF, Hsu TC, Wang CA, Jang MF, Kuo YC, Alper HS, Guo GL, Hwang WS (2018) Production of optically pure L(+)-lactic acid from waste plywood chips using an isolated thermotolerant Enterococcus faecalis SI at a pilot scale. J Ind Microbiol Biotechnol 45(11):961–970. https://doi.org/10.1007/s10295-018-2078-5
Zhang W, Li D, Liu L, Zang J, Duan Q, Yang W, Zhang L (2013) The effects of dietary fiber level on nutrient digestibility in growing pigs. J Anim Sci Biotechnol 4(1):17. https://doi.org/10.1186/2049-1891-4-17
Zhang Z, Liu Z, Zhang S, Lai C, Ma D, Huang C (2018) Effect of inclusion level of corn germ meal on the digestible and metabolizable energy and evaluation of ileal AA digestibility of corn germ meal fed to growing pigs1. J Anim Sci 97(2):768–778. https://doi.org/10.1093/jas/sky469
Zheng L, Li D, Li ZL, Kang LN, Jiang YY, Liu XY, Chi YP, Li YQ, Wang JH (2017) Effects of Bacillus fermentation on the protein microstructure and anti-nutritional factors of soybean meal. Lett Appl Microbiol 65(6):520–526. https://doi.org/10.1111/lam.12806
Ziętek M, Celewicz Z, Szczuko M (2021) Short-chain fatty acids, maternal microbiota and metabolism in pregnancy. Nutrients. https://doi.org/10.3390/nu13041244
Acknowledgements
The authors would like to thank all the reviewers who participated in the review, as well as MJEditor (www.mjeditor.com) for providing English editing services during the preparation of this manuscript.
Funding
This research was supported by the Postdoctoral Grant of Jilin Province (JLSB2022001); The Basic scientific research fund project of Jilin Academy of Agricultural Sciences (KYJF2021JQ103); Funding program for High-level scientifc and technological innovation talents introduced by Scientifc research Institutes of Jilin Province.
Author information
Authors and Affiliations
Contributions
LC: Methodology, Resources, Formal analysis, Investigation, Writing–original draft, Writing–review and editing, Funding acquisition, Supervision. YG: Conceptualization, Investigation, Writing—review and editing. XL: Resources, Formal analysis, Data curation. LZ: Resources, Investigation. BW: Project administration, Writing—review and editing, Supervision. ZZ: Project administration, Investigation, Data curation, Writing—review and editing, Supervision.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, L., Guo, Y., Liu, X. et al. Cellulase with Bacillus velezensis improves physicochemical characteristics, microbiota and metabolites of corn germ meal during two-stage co-fermentation. World J Microbiol Biotechnol 40, 59 (2024). https://doi.org/10.1007/s11274-023-03831-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-023-03831-w