Abstract
Serratiopeptidase is a bacterial protease that has been used medicinally in variety of applications. Though, some drawbacks like sensitivity to environmental conditions and low penetration into cells limited its usage as a potent pharmaceutical agent. This study aimed to produce four novel truncated serratiopeptidase analogs with different lengths and possessing one disulfide bridge, in order to enhance protease activity and thermal stability of this enzyme. Mutagenesis and truncation were performed using specific primers by conventional and overlap PCR. The recombinant proteins were expressed in E. coli cells then purified and their protease activity and stability were checked at different pH and temperatures in comparison to the native form of the enzyme, Serra473. Enzyme activity assay showed that T306 [12–302 ss] was not further active which could be due to the large truncation. However, T344 [8–339 ss], T380 [8–339 ss] and T380 [12–302 ss] proteins showed higher proteolytic activity comparing to Serra473. These analogs were active at temperatures of 25–90 °C and pH 6–9.5. Interestingly, remaining enzyme activity of T344 [8–339 ss], T380 [8–339 ss] and T380 [12–302 ss] forms at 90 °C calculated as 87, 83 and 86 percent, respectively, comparing to the activity at room temperature. However, residual activity at the same conditions was 50% for the full length enzyme. Formation of disulfide bond in engineered serratiopeptidases could be the main reason for higher thermal stability compared to Serra473. Thermostability of T344 [8–339 ss], as the most thermostable designed serratiopeptidase, was additionally confirmed using differential scanning calorimetry.
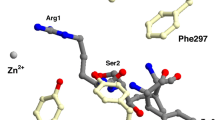
Graphical abstract







Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Anil CS, Kashinath MA (2013) Production, Characterization and optimization of potent protease (Serratiopeptidase) from Serratia marcescens E 15. Int Res J Pharm App Sci 3(4):95–98. https://doi.org/10.1007/s11515-017-1461-3
Bhagat S, Agarwal M, Roy V (2013) Serratiopeptidase: a systematic review of the existing evidence. Int J Surg 11(2013):209–217. https://doi.org/10.1016/j.ijsu.2013.01.010
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Charlton A, Zachariou M (2008) Immobilized metal ion affinity chromatography of histidine-tagged fusion proteins. Humana Press, In Affinity Chromatography, pp 137–150
Coêlho DF, Saturnino TP, Fernandes FF, Mazzola PG, Silveira E, Tambourgi EB (2016) Azocasein Substrate for Determination of Proteolytic Activity: Reexamining a Traditional Method Using Bromelain Samples. Biomed Res Int. 2016:8409183. doi: https://doi.org/10.1155/2016/8409183. Epub 2016 Jan 27. PMID: 26925415; PMCID: PMC4748065.
Cupp-Enyard C (2008) Sigma’s non-specific protease activity assay - Casein as a aubstrate. J vis Exp 19:899. https://doi.org/10.3791/899.PMID:19066538;PMCID:PMC2872977
Esch PM, Gerngross H, Fabian A (1989) Reduction of postoperative swelling. Objective measurement of swelling of the upper ankle joint in treatment with serrapeptase–a prospective study. Fortschr Med 107(4):71–72
Ethiraj S, Gopinath S (2017) Production, purification, characterization, immobilization, and application of Serrapeptase: a review. Front Biol 12(5):333–348. https://doi.org/10.1007/s11515-017-1461-3
Gallagher SR (2012) SDS-polyacrylamide gel electrophoresis (SDS-PAGE). Current Protocols Essential Laboratory Techniques. https://doi.org/10.1002/9780470089941.et0703s06
Gurung N, Ray S, Bose S, Rai V (2013) A broader view: microbial enzymes and their relevance in industries, medicine, and beyond. BioMed Res Int 2013:329121. https://doi.org/10.1155/2013/329121
Ishihara Y, Kitamura S, Takaku F (1983) Experimental studies on distribution of cefotiam, a new beta-lactam antibiotic, in the lung and trachea of rabbits II. Combined effects with serratiopeptidase. Jpn J Antibiot 36(10):2665–2670
Iversen SL, Jørgensen MH (1995) Azocasein assay for alkaline protease in complex fermentation broth. Biotechnol Tech 9:573–576. https://doi.org/10.1007/BF00152446
Jadav SP, Patel NH, Shah TG, Gajera MV, Trivedi HR, Shah BK (2010) Comparison of anti-inflammatory activity of serratiopeptidase and diclofenac in albino rats. J Pharmacol Pharmacother 1(2):116–117. https://doi.org/10.4103/0976-500X.72362
Jadhav SB, Shah N, Rathi A, Rathi V, Rathi A (2020) Serratiopeptidase: insights into the therapeutic applications. Biotechnol Rep (amst) 28:e00544. https://doi.org/10.1016/j.btre.2020.e00544
Joshi KK, Nerurkar RP (2012) Anti-inflammatory effect of the serratiopeptidase-rationale or fashionable: a study in rat paw edema model induced by the carrageenan. Indian J Physiol Pharmacol 56(4):367–374
Khanna A, Khanna M, Aggarwal A (2013) Serratia marcescens-a rare opportunistic nosocomial pathogen and measures to limit its spread in hospitalized patients. J Clin Diagn Res 7(2):243–246. https://doi.org/10.7860/JCDR/2013/5010.2737
Klein G, Kullich W (2000) Short-term treatment of painful osteoarthritis of the knee with oral enzymes: randomised, double blind study versus Diclofenac. Clin Drug Investig 19(1):15–23. https://doi.org/10.2165/00044011-200019010-00003
Kv S, Devi GS, Mathew ST (2008) Liposomal formulations of serratiopeptidase: in vitro studies using PAMPA and Caco-2 models. Mol Pharm. https://doi.org/10.1021/mp700090r
Mahlen SD (2011) Serratia infections: from military experiments to current practice. Clin Microbiol Rev 24(4):755–791. https://doi.org/10.1128/CMR.00017-11
Mahmood T, Yang PC (2012) Western blot: technique, theory, and trouble shooting. N Am J Med Sci 4(9):429–434. https://doi.org/10.4103/1947-2714.100998
Majima Y, Inagaki M, Hirata K, Takeuchi K, Morishita A, Sakakura Y (1988) The effect of an orally administered proteolytic enzyme on the elasticity and viscosity of nasal mucus. Arch Otorhinolaryngol 244(6):355–359. https://doi.org/10.1007/BF00497464
Malshe PC (1998) Orally administered serratiopeptidase: can it work ? J Assoc Physicians India 46(5):492
Malshe PC (2000) A preliminary trial of serratiopeptidase in patients with carpal tunnel Syndrome. J Assoc Physicians India 48(11):1130
Metkar SK, Girigoswami A, Murugesan R, Girigoswami K (2016) In vitro and in vivo insulin amyloid degradation mediated by Serratiopeptidase. Mater Sci Eng C 70(Pt1):728–735. https://doi.org/10.1016/j.msec.2016.09.049
Niu C, Zhu L, Xu X, Li Q (2016) Rational Design of Disulfide Bonds Increases Thermostability of a Mesophilic 1,3–1,4-β-Glucanase from Bacillus terquilensis. PLoS ONE 11(4):e0154036. https://doi.org/10.1371/journal.pone.0154036
Panagariya A, Sharma AK (1999) A preliminary trial of serratiopeptidase in patients with carpal tunnel syndrome. J Assoc Physicians India 47(12):1170–1172. https://doi.org/10.5958/0974-360X
Pansuriya R, Singhal R (2011) Effects of dissolved oxygen and agitation on production of serratiopeptidase by Serratia marcescens NRRL B-23112 in stirred tank bioreactor and its kinetic modeling. J Microbiol Biotechnol 21(4):430–437. https://doi.org/10.4014/jmb.1009.0903
Rawat M, Daharwal SJ, Singh D (2008) An overview of miracle enzyme- Serratiopeptidase. Research J Pharm and Tech 1(3):124–131
Rouhani M, Valizadeh V, Ahangari Cohan R, Norouzian D (2018) Computational design, structure refinement and molecular dynamics simulation of novel engineered serratiopeptidase analogs. J Biomol Struct Dyn 1(11):4171–4180. https://doi.org/10.1080/07391102.2018.1540361
Rouhani M, Valizadeh V, Molahsalehi S, Norouzian D (2020) Production and expression optimization of heterologous serratiopeptidase. Iran J Public Health 49:931–939. https://doi.org/10.18502/ijph.v49i5.3211
Selan L, Papa R, Tilotta M, Vrenna G, Carpentieri A, Amoresano A, Pucci P, Artini M (2015) Serratiopeptidase: a well-known metalloprotease with a new non-proteolytic activity against S. aureus biofilm. BMC Microbiol. https://doi.org/10.1186/s12866-015-0548-8
Srivastava V, Mishra S, Chaudhuri TK (2019) Enhanced production of recombinant serratiopeptidase in Escherichia coli and its characterization as a potential biosimilar to native biotherapeutic counterpart. Microb Cell Fact 18:215–230. https://doi.org/10.1186/s12934-019-1267-x
Tiwari M (2017) The role of serratiopeptidase in the resolution of inflammation. Asian J Pharm Sci 12(3):209–215. https://doi.org/10.1016/j.ajps.2017.01.003
Wen J, Arthur K, Chemmalil L, Muzammil S, Gabrielson J, Jiang Y (2012) Applications of differential scanning calorimetry for thermal stability analysis of proteins: qualification of DSC. J Pharm Sci 101(3):955–964. https://doi.org/10.1002/jps.22820
Wetzel R, Perry LJ, Baase WA, Becktel WJ (1988) Disulfide bonds and thermal stability in T4 lysozyme. Proc Natl Acad Sci U S A 85(2):401–405. https://doi.org/10.1073/pnas.85.2.401
Yamaguchi H, Miyazaki M (2014) Refolding techniques for recovering biologically active recombinant proteins from inclusion bodies. Biomolecules 4(1):235–251. https://doi.org/10.3390/biom4010235
Zavodszky M, Chen C, huang J, Zolkiewski M, Wen L, Krishnamoorthi R, (2001) Disulfide bond effects on protein stability: designed variants of Cucurbita maxima trypsin inhibitor-V. Prot Sci 10:149–160
Zhang L, Conway JF, Thibodeau PH (2012) Calcium-induced folding and stabilization of the Pseudomonas aeruginosa alkaline protease. J Biol Chem 287(6):4311–4322. https://doi.org/10.1074/jbc.M111.310300
Zhang XF, Yang GY, Zhang Y, Xie Y, Withers SG, Feng Y (2016) A general and efficient strategy for generating the stable enzymes. Sci Rep 6:33797. https://doi.org/10.1038/srep33797
Zhang L, Morrison AJ, Thibodeau PH (2015) Interdomain contacts and the stability of serralysin protease from Serratia marcescens. PLoS ONE. https://doi.org/10.1371/journal.pone.0138419
Acknowledgements
The authors wish to express their deep gratitude to all who provided support during the course of this research. This project was financially supported by Pasteur Institute of Iran.
Funding
Pasteur Institute of Iran
Author information
Authors and Affiliations
Contributions
M.R. (Ph.D. student) contributed in the laboratory work, analysis of the data and drafted the paper. A.A., S.P. and S.M. helped with the experimental assays, R.A.C. gave critical suggestions for designing enzyme engineered forms. V.V. designed the work, supervised the study and critically revised the manuscript and N.D. helped with data analysis and critical reading of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rouhani, M., Valizadeh, V., Aghai, A. et al. Design, expression and functional assessment of novel engineered serratiopeptidase analogs with enhanced protease activity and thermal stability. World J Microbiol Biotechnol 38, 17 (2022). https://doi.org/10.1007/s11274-021-03195-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-021-03195-z