Abstract
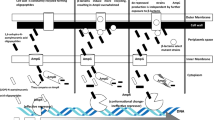
Shiga toxin-producing Escherichia coli (STEC) are zoonotic pathogens and may induce severe diarrheagenic diseases in humans and other animals. Non-O157 STEC have been emerging as important pathogens causing outbreaks worldwide. Bacterial resistance to antimicrobials has become a global public health problem, which involves different ecological spheres, including animals. This study aimed to characterize the resistance to antimicrobials, plasmids and virulence, as well as the serotypes and phylogenetic groups in E. coli isolated from sheep in Brazil. A total of 57 isolates were obtained and showed different antimicrobial resistance profiles. Nineteen isolates presented acquired antimicrobial resistance genes (ARGs) (blaCTX-M-Gp9, qnrB, qnrS, oqxB, oqxA, tetA, tetB, tetC, sul1 and sul2) and plasmid families (F, FIA, FIB, I1, K, HI1 and ColE-like). The stx1, stx2 and ehxA virulence genes were detected by PCR, being 50 isolates (87.7%) classified as STEC. A great diversity of serotypes was detected, being O176:HNM the most predominant. Phylogenetic group E was the most prevalent, followed by B1, A and B2. To the best of our knowledge, this is the first report in the world of blaCTX-M-Gp9 (O75, O114, O100, O128ac and O176 serogroups), qnrB and oqxB genes in non-O157 STEC in healthy sheep. The results obtained in the present study call attention to the monitoring of antimicrobial-resistant non-O157 STEC harboring acquired ARGs worldwide and indicate a zoonotic risk due to the profile of virulence, resistance and serotype found.
Similar content being viewed by others
References
Amézquita-López BA, Quiñones B, Soto-Beltrán M, Lee BG, Yambao JC, Lugo-Melchor OY, Chaidez C (2016) Antimicrobial resistance profiles of Shiga toxin-producing Escherichia coli O157 and Non-O157 recovered from domestic farm animals in rural communities in Northwestern Mexico. Antimicrob Resist Infect Control 5:1. https://doi.org/10.1186/s13756-015-0100-5
Aranda K, Fabbricotti SH, Fagundes-Neto U, Scaletsky ICA (2007) Single multiplex assay to identify simultaneously enteropathogenic, enteroaggregative, enterotoxigenic, enteroinvasive and Shiga toxin producing Escherichia coli strains in Brazilian children. FEMS Microbiol Lett 267:145. https://doi.org/10.1111/j.1574-6968.2006.00580.x
Bai L, Hurley D, Li J, Meng Q, Wang J, Fanning S, Xiong Y (2016) Characterisation of multidrug-resistant Shiga toxin-producing Escherichia coli cultured from pigs in China: co-occurrence of extended-spectrum β-lactamase- and mcr-1-encoding genes on plasmids. Int J Antimicrob Agents 48:445–448. https://doi.org/10.1016/j.ijantimicag.2016.06.021
Cantón R, González-Alba JM, Galán JC (2012) CTX-M enzymes: origin and diffusion. Front Microbiol 3:110. https://doi.org/10.3389/fmicb.2012.00110
Carattoli A (2013) Plasmids and the spread of resistance. Int J Med Microbiol 303(6–7):298–304. https://doi.org/10.1016/j.ijmm.2013.02.001
Carattoli A, Bertini A, Villa L, Falbo V, Hopkins KL, Threlfall EJ (2005) Identification of plasmids by PCR-based replicon typing. J Microbiol Methods 63:219–228. https://doi.org/10.1016/j.mimet.2005.03.018
Carlos C, Pires MM, Stoppe NC, Hachich EM, Sato MI, Gomes TA, Amaral LA, Ottoboni LM (2010) Escherichia coli phylogenetic group determination and its application in the identification of the major animal source of fecal contamination. BMC Microbiol 10:161. https://doi.org/10.1186/1471-2180-10-161
Cattoir V, Poirel L, Rotimi V, Soussy CJ, Nordmann P (2007) Multiplex PCR for detection of plasmid-mediated quinolone resistance qnr genes in ESBL-producing enterobacterial isolates. J Antimicrob Chemother 60:394–397. https://doi.org/10.1093/jac/dkm204
Centers for Disease Control and Prevention (CDC). National STEC Surveillance Annual Report, 2015. Atlanta, Georgia: US Department of Health and Human Services, CDC, 2017. Retrieved from https://www.cdc.gov/nationalsurveillance/pdfs/STEC_Annual_Summary_2015-508c.pdf. Accessed 10 Jan 2019.
Cergole-Novella MC, Pignatari AC, Castanheira M, Guth BE (2011) Molecular typing of antimicrobial-resistant Shiga-toxin-producing Escherichia coli strains (STEC) in Brazil. Res Microbiol 162:117–123. https://doi.org/10.1016/j.resmic.2010.09.022
Cerna JF, Nataro JP, Estrada-Garcia T (2003) Multiplex PCR for detection of three plasmid-borne genes of enteroaggregative Escherichia coli strains. J Clin Microbiol 41:2138–2140
Chen X, Zhang W, Pan W, Yin J, Pan Z, Gao S, Jiao X (2012) Prevalence of qnr, aac(6')-Ib-cr, qepA, and oqxAB in Escherichia coli isolates from humans, animals, and the environment. Antimicrob Agents Chemother 56:3423–3427. https://doi.org/10.1128/AAC.06191-11
Clermont O, Christenson JK, Denamur E, Gordon DM (2013) The Clermont Escherichia coli phylo-typing method revisited: improvement of specificity and detection of new phylo-groups. Environ Microbiol Rep 5:58–65. https://doi.org/10.1111/1758-2229.12019
CLSI (2017) Performance standards for antimicrobial susceptibility testing: twenty-seventh informational supplement. CLSI document M100–S27. Clinical and Laboratory Standards Institute, Wayne, PA
Dallenne C, Costa A, Decré D, Favier C, Arlet G (2010) Development of a set of multiplex PCR assays for the detection of genes enconding importante beta-lactamases in Enterobacteriaceae. J Antimicrob Chemother 65:490–495. https://doi.org/10.1093/jac/dkp498
Etcheverría AI, Padola NL (2013) Shiga toxin-producing Escherichia coli: factors involved in virulence and cattle colonization. Virulence 4:366–372. https://doi.org/10.4161/viru.24642
Ewers C, Stamm I, Stolle I, Guenther S, Kopp PA, Fruth A, Wieler LH, Scheufen S, Bauerfeind R, Bethe A, Prenger-Berninghoff E (2014) Detection of Shiga toxin- and extended-spectrum β-lactamase-producing Escherichia coli O145:NM and Ont:NM from calves with diarrhoea. J Antimicrob Chemother 69:2005–2007. https://doi.org/10.1093/jac/dku042
Ferdous M, Friedrich AW, Grundmann H, de Boer RF, Croughs PD, Islam MA, Kluytmans-van den Bergh MF, Kooistra-Smid AM, Rossen JW (2016) Molecular characterization and phylogeny of Shiga toxin-producing Escherichia coli isolates obtained from two Dutch regions using whole genome sequencing. Clin Microbiol Infect 22:642.e1–9. https://doi.org/10.1016/j.cmi.2016.03.028
Ferens WA, Hovde CJ (2011) Escherichia coli O157:H7: animal reservoir and sources of human infection. Foodborne Pathog Dis 8:465–487. https://doi.org/10.1089/fpd.2010.0673
Folster JP, Pecic G, Taylor E, Whichard J (2011) Characterization of Isolates from an outbreak of multidrug-resistant, Shiga toxin-producing Escherichia coli O145 in the United States. Antimicrob Agents Chemother 55:5955–5956. https://doi.org/10.1128/AAC.05545-11
García-Fernández A, Fortini D, Veldman K, Mevius D, Carattoli A (2009) Characterization of plasmids harbouring qnrS1, qnrB2 and qnrB19 genes in Salmonella. J Antimicrob Chemother 63:274–281. https://doi.org/10.1093/jac/dkn470
Ghanbarpour R, Kiani M (2013) Characterization of non-O157 shiga toxin-producing Escherichia coli isolates from healthy fat-tailed sheep in southeastern of Iran. Trop Anim Health Prod 45:641–648. https://doi.org/10.1007/s11250-012-0271-5
Karczmarczyk M, Abbott Y, Walsh C, Leonard N, Fanning S (2011) Characterization of multidrug-resistant Escherichia coli isolates from animals presenting at a university veterinary hospital. Appl Environ Microbiol 77:7104–7112. https://doi.org/10.1128/AEM.00599-11
Kerrn MB, Klemmensen T, Frimodt-Mǿller N, Espersen F (2002) Susceptibility of Danish Escherichia coli strains isolated from urinary tract infections and bacteraemia, and distribution of sul genes conferring sulphonamide resistance. J Antimicrob Chemother 50:513–516. https://doi.org/10.1093/jac/dkf164
Kumar A, Taneja N, Kumar Y, Sharma M (2012) Detection of Shiga toxin variants among Shiga toxin-forming Escherichia coli isolates from animal stool, meat and human stool samples in India. J Appl Microbiol 113:1208–1216. https://doi.org/10.1111/j.1365-2672.2012.05415.x
Kusumoto M, Hikoda Y, Fujii Y, Murata M, Miyoshi H, Ogura Y, Gotoh Y, Iwata T, Hayashi T, Akiba M (2016) Emergence of a multidrug-resistant Shiga toxin-producing enterotoxigenic Escherichia coli lineage in diseased swine in Japan. J Clin Microbiol 54:1074–1081. https://doi.org/10.1128/JCM.03141-15
Lima IF, Boisen N, Quetz Jda S, Havt A, de Carvalho EB, Soares AM, Lima NL, Mota RM, Nataro JP, Guerrant RL, Lima AÂ (2013) Prevalence of enteroaggregative Escherichia coli and its virulence-related genes in a case-control study among children from north-eastern Brazil. J Med Microbiol 62:683–693. https://doi.org/10.1099/jmm.0.054262-0
Lyimo B, Buza J, Subbiah M, Temba S, Kipasika H, Smith W, Call DR (2016) IncF plasmids are commonly carried by antibiotic resistant Escherichia coli isolated from drinking water sources in Northern Tanzania. J Med Microbiol 2016:3103672. https://doi.org/10.1155/2016/3103672
Martin MJ, Thottathil SE, Newman TB (2015) Antibiotics overuse in animal agriculture: a call to action for health care providers. Am J Public Health 105:2409–2410. https://doi.org/10.2105/AJPH.2015.302870
Mora A, López C, Dhabi G, López-Beceiro AM, Fidalgo LE, Díaz EA, Martínez-Carrasco C, Mamani R, Herrera A, Blanco JE, Blanco M, Blanco J (2012) Seropathotypes, phylogroups, Stx subtypes, and intimin types of wildlife-carried, shiga toxin-producing Escherichia coli strains with the same characteristics as human-pathogenic isolates. Appl Environ Microbiol 78:2578–2585. https://doi.org/10.1128/AEM.07520-11
Mukherjee S, Mosci RE, Anderson CM, Snyder BA, Collins J, Rudrik JT, Manning SD (2017) Antimicrobial drug-resistant Shiga toxin-producing Escherichia coli infections, Michigan, USA. Emerg Infect Dis 23:1609–1611. https://doi.org/10.3201/eid2309.170523
Ng LK, Martin I, Alfa M, Mulvey M (2001) Multiplex PCR for the detection of tetracycline resistant genes. Mol Cell Probes 15:209–215. https://doi.org/10.1006/mcpr.2001.0363
Noppe-Leclercq I, Wallet F, Haentjens S, Courcol R, Simonet M (1999) PCR detection of aminoglycoside resistance genes: a rapid molecular typing method for Acinetobacter baumannii. Res Microbiol 150:317–322
Orskov F, Orskov I (1984) Serotyping of Escherichia coli. Methods Microbiol 14:43–112
Pallecchi L, Riccobono E, Sennati S, Mantella A, Bartalesi F, Trigoso C, Gotuzzo E, Bartoloni A, Rossolini GM (2010) Characterization of small ColE-like plasmids mediating widespread dissemination of the qnrB19 gene in commensal enterobacteria. Antimicrob Agents Chemother 54:678–682. https://doi.org/10.1128/AAC.01160-09
Paton AW, Paton JC (1998) Detection and characterization of Shiga toxigenic Escherichia coli by using multiplex PCR assays for stx1, stx2, eaeA, enterohemorrhagic E. coli hlyA, rfbO111, and rfbO157. J Clin Microbiol 36:598–602
Perreten V, Boerlin P (2003) A new sulfonamide resistance gene (sul3) in Escherichia coli is widespread in the pig population of Switzerland. Antimicrob Agents Chemother 47:1169–1172
Salyers A, Shoemaker NB (2006) Reservoirs of antibiotic resistance genes. Anim Biotechnol 17:137–146. https://doi.org/10.1080/10495390600957076
Scheutz F, Cheasty T, Woodward D, Smith HR (2004) Designation of O174 and O175 to temporary O groups OX3 and OX7, and six new E. coli O groups that include Verocytotoxin-producing E. coli (VTEC): O176, O177, O178, O179, O180 and O181. APMIS 112:569–584. https://doi.org/10.1111/j.1600-0463.2004.apm1120903.x
Schmidt H, Knop C, Franke S, Aleksic S, Heesemann J, Karch H (1995) Development of PCR for screening of enteroaggregative Escherichia coli. J Clin Microbiol 33:701–705
Srinivasa TR, Gill JPS, Kumar GVVPS, Ghatak S (2011) Multi drug resistance patterns of Shiga toxin–producing Escherichia coli (STEC) and non–STEC isolates from meats, RTE meat foods, drinking water and human diarrhoeic samples of Punjab. India Arch Clin Microbiol 2:3. https://doi.org/10.3823/225
Stoppe NC, Silva JS, Carlos C, Sato MIZ, Saraiva AM, Ottoboni LMM, Torres TT (2017) Worldwide phylogenetic group patterns of Escherichia coli from commensal human and wastewater treatment plant isolates. Front Microbiol 8:2512. https://doi.org/10.3389/fmicb.2017.02512
Toner E, Adalja A, Gronvall GK, Cicero A, Inglesby TV (2015) Antimicrobial resistance is a global health emergency. Health Secur 13:153–155. https://doi.org/10.1089/hs.2014.0088
Tseng M, Sha Q, Rudrik JT, Collins J, Henderson T, Funk JA, Manning SD (2016) Increasing incidence of non-O157 Shiga toxin-producing Escherichia coli (STEC) in Michigan and association with clinical illness. Epidemiol Infect 144:1394–1405. https://doi.org/10.1017/S0950268815002836
Valat C, Haenni M, Saras E, Auvray F, Forest K, Oswald E, Madec JY (2012) CTX-M-15 extended-spectrum β-lactamase in a shiga toxin-producing Escherichia coli isolate of serotype O111:H8. Appl Environ Microbiol 78:8–9. https://doi.org/10.1128/AEM.06997-11
Weisburg WG, Barns SM, Pelletier BA, Lane DJ (1991) 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 173:697–703. https://doi.org/10.1128/jb.173.2.697-703.1991
Yang QE, Sun J, Li L, Deng H, Liu BT, Fang LX, Liao XP, Liu YH (2015) IncF plasmid diversity in multi-drug resistant Escherichia coli strains from animals in China. Front Microbiol 6:964. https://doi.org/10.3389/fmicb.2015.00964
Acknowledgements
The authors thank the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for fellowship [Grant No. 88882.180855/2018-01].
Funding
This work was supported by São Paulo Research Foundation—FAPESP [Grant No. 2018/19539-0].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Furlan, J.P.R., Gallo, I.F.L., de Campos, A.C.L.P. et al. Characterization of non-O157 Shiga toxin-producing Escherichia coli (STEC) obtained from feces of sheep in Brazil. World J Microbiol Biotechnol 35, 134 (2019). https://doi.org/10.1007/s11274-019-2712-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-019-2712-z