Abstract
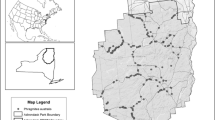
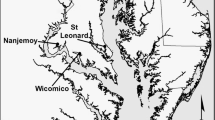
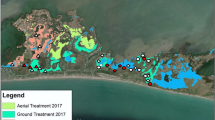
Complete eradication of invasive plants is often infeasible but in some cases it is possible to reduce the invader to low levels with reduced ongoing management costs, referred to as functional eradication. Non-native Phragmites australis has challenged land managers across North America but functional eradication may yet be possible in some scenarios. Here we present data from Chesapeake Bay brackish tidal wetlands where two approaches to Phragmites management (long-term, continuous management and short-term, non-continuous management) were used. We demonstrate that the application of herbicides will lead to the establishment of native species, but long-term, continuous management is required to facilitate functional eradication of Phragmites by keeping it at low levels of occurrence and restoring native plant communities. Ultimately, historical data from sites that were sampled as part of this study indicate that if management is successful and results in functional eradication of Phragmites, then the recovering vegetation will include native species that were present before the sites were invaded by Phragmites.




Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Able KW, Hagan SM (2003) Impact of Common Reed, Phragmites australis, on essential fish habitat: influence on reproduction, embryological development, and larval abundance of mummichog (Fundulus heteroclitus). Estuaries 26(1):40–50. http://www.jstor.org/stable/1353190
Amsberry L, Baker MA, Ewanchuk PJ et al (2000) Clonal integration and the expansion of Phragmites australis. Ecol Appl 10:1110–1118
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting Linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Blanca MJ, Alarcón R, Arnau J, Bono R et al (2017) Non-normal data: is ANOVA still a valid option? Psicothema 29(4):552–557. https://doi.org/10.7334/psicothema2016.383
Boone J, Furbish E, Turner k et al (1988) Clear plastic: A non-chemical herbicide. Restor Manage Notes 6(2):94–95
Bonello J, Judd K (2019) Plant community recovery after herbicide management to remove Phragmites australis in Great Lakes coastal wetlands. Restor Ecol 28:215–221. https://doi.org/10.1111/rec.13062
Chambers RM, Meyerson LA, Saltonstall K (1999) Expansion of Phragmites australis into tidal wetlands of North America. Aquat Bot 64(3):261–273. https://doi.org/10.1016/S0304-3770(99)00055-8
Delacre M, Lakens D, Leys C (2017) Why psychologists should by default use Welch’s t-test instead of Student’s t-test. Int Rev Social Psychol. https://doi.org/10.5334/irsp.82
Dawson FG, Hallows HB (1983) Practical applications of a shading material for macrophyte control in watercourses. Aq Bot 17:301–308
Drake BG (2014) Rising sea level, temperature, and precipitation impact plant and ecosystem responses to elevated CO2 on a Chesapeake Bay wetland: review of a 28-year study. Glob Change Biol 20(11):3329–3343. https://doi.org/10.1111/gcb.12631
Elgersma KJ, Martina JP, Goldberg DF et al (2017) Effectiveness of cattail (Typha spp.) management techniques depends on exogenous nitrogen inputs. Elem Sci Anth 5: 19. https://doi.org/10.1525/elementa.147
Epanchin-Niell RS, Hufford MB, Aslan CE et al (2010) Controlling invasive species in complex social landscapes. Front Ecol Environ 8(4):210–216. https://doi.org/10.1890/090029
Faison EK, Elkins G, Kitka K et al (2020) Response of a wetland plant community to management of Phragmites australis (Poaceae) in southwestern Connecticut. Rhodora 122(989):48–52. https://doi.org/10.3119/19-05
Farnsworth EJ, Meyerson LA (1999) Species composition and inter-annual dynamics of a freshwater tidal plant community following removal of the invasive grass, Phragmites australis. Biol Invasions 1(2):115–127. https://doi.org/10.1023/A:1010068607630
Furman BT, Leone EH, Bell S et al (2018) Braun-Blanquet data in ANOVA designs: comparisons with percent cover and transformations using simulated data. Mar Ecol Prog Ser. https://doi.org/10.3354/meps12604
Gratton C, Denno RF (2005) Restoration of arthropod assemblages in a Spartina salt marsh following removal of the invasive plant Phragmites australis. Restor Ecol 13(2):358–372. https://doi.org/10.1111/j.1526-100X.2005.00045.x
Green SJ, Grosholz ED (2021) Functional eradication as a framework for invasive species control. Front Ecol Environ 19(2):98–107. https://doi.org/10.1002/fee.2277
Hallinger KD, Shisler JK (2009) Seed bank colonization in tidal wetlands following Phragmites control (New Jersey). Ecol Restor 27(1):16. https://doi.org/10.3368/er.27.1.16
Hazelton ELG, Mozdzer TJ, Burdick DM et al (2014) Phragmites australis management in the United States: 40 years of methods and outcomes. AoB Plants. https://doi.org/10.1093/aobpla/plu001
Hazelton ELG, Downard R, Kettenring KM et al (2018) Spatial and temporal variation in brackish wetland seedbanks: implications for wetland restoration following Phragmites control. Estuaries Coasts 41(1):68–84. https://doi.org/10.1007/s12237-017-0289-z
Holdredge C, Bertness MD (2011) Litter legacy increases the competitive advantage of invasive Phragmites australis in New England wetlands. Biol Invasions 13(2):423–433. https://doi.org/10.1007/s10530-010-9836-2
Jacobson S, Brooks H, Baldwin A et al (2023) Transplanting native species to expedite succession following Phragmites australis removal. Final report. Maryland Sea Grant Program, Annapolis, MD
Kettenring KM, Blois S, Hauber D (2012) Moving from a regional to a continental perspective of Phragmites australis invasion in North America. AoB Plants. https://doi.org/10.1093/aobpla/pls040
Kettenring KM, Whigham DF, Hazelton ELG et al (2015) Biotic resistance, disturbance, and mode of colonization impact the invasion of a widespread, introduced wetland grass. Ecol Appl 25(2):66–480. http://www.jstor.org/stable/24432316
Kiviat E (2006) Phragmites management sourcebook for the tidal Hudson River. Report to the Hudson River Foundation. New York, New York
Kuznetsova A, Brockhoff PB, Christensen RHB (2017) lmerTest Package: tests in Linear mixed effects models. J Stat Softw 82(13):1–26. https://doi.org/10.18637/jss.v082.i13
Lambert AM, Saltonstall K, Long R et al (2016) Biogeography of Phragmites australis lineages in the southwestern United States. Biol Invasions 18(9):2597–2617. https://doi.org/10.1007/s10530-016-1164-8
Levene H (1960) Robust tests for equality of variances. In: Olkin I (ed) Contributions to Probabiulity and statistics. Stanford University Press, Redwood City, California, pp 278–292
Lombard KB, Tomassi D, Ebersole J (2012) Long-term management of an invasive plant: lessons from seven years of Phragmites australis control. Northeastern Naturalist 19:181–193. http://www.jstor.org/stable/41495846
Lu M, Herbert ER, Langley JA et al (2019) Nitrogen status regulates morphological adaptation of marsh plants to elevated CO2. Nat Clim Change 9(10):764–768. https://doi.org/10.1038/s41558-019-0582-x
Marks M, Lapin B, Randall J (1994) Phragmites australis (P. communis): threats, management, and monitoring. Nat Areas J 14(4):285–294
Martin LJ, Blossey B (2013) The runaway weed: costs and failures of Phragmites australis management in the USA. Estuaries Coasts 36(3):626–632. https://doi.org/10.1007/s12237-013-9593-4
McCormick J, Somes HH Jr (1982) The Coastal wetlands of Maryland. Maryland Department of Natural Resource, Coastal Zone Management Program. Chevy Chase, Maryland
McCormick MK, Kettenring KM, Baron HM et al (2010) Extent and reproductive mechanisms of Phragmites australis spread in brackish wetlands in Chesapeake Bay, Maryland (USA). Wetlands 30(1):67–74. https://doi.org/10.1007/s13157-009-0007-0
Meyerson LA, Saltonstall K, Windham L et al (2000) A comparison of Phragmites australis in freshwater and brackish marsh environments in North America. Wetlands Ecol Manage 8(2):89–103. https://doi.org/10.1023/A:1008432200133
Meyerson LA, Lambert AM, Saltonstall K (2010) A tale of three lineages: expansion of common reed (Phragmites australis) in the U.S. Southwest and Gulf Coast. Invasive Plant Sci Manage 3(4):515–520. https://doi.org/10.1614/IPSM-D-09-00052.1
Oksanen J (2022) Package ‘vegan’. http://vegan.r-forge.r-project.org/. https://cran.r-project.org/web/packages/vegan/vegan.pdf
Quirion B, Simek Z, Dávalos et al (2018) Management of invasive Phragmites australis in the Adirondacks: a cautionary tale about prospects of eradication. Biol Invasions 20(1):59–73. https://doi.org/10.1007/s10530-017-1513-2
Rohal CB, Cranney C, Hazelton E et al (2019a) Invasive Phragmites australis management outcomes and native plant recovery are context dependent. Ecol Evol. https://doi.org/10.1002/ece3.5820
Rohal CB, Cranney C, Kettenring KM (2019b) Abiotic and landscape factors constrain restoration outcomes across spatial scales of a widespread invasive plant. Front Plant Sci 10:481. https://doi.org/10.3389/fpls.2019.00481
Rohal CB, Reinhardt Adams C, Reynolds LK et al (2021) Do common assumptions about the wetland seed bank following invasive plant removal hold true? Divergent outcomes following multi-year Phragmites australis management. Appl Veg Sci 24(4):e12626. https://doi.org/10.1111/avsc.12626
Rohal CB, Hazelton ELG, McFarland EK et al (2023) Landscape and site factors drive invasive Phragmites management and native plant recovery across Chesapeake Bay wetlands. Ecosphere 14(1):e4392. https://doi.org/10.1002/ecs2.4392
Royston JP (1982) An extension of Shapiro and Wilk’s W Test for normality to large samples. J Royal Stat Soc Ser C (Applied Statistics) 31(2):115–124. https://doi.org/10.2307/2347973
Shapiro SS, Wilk MB (1965) An analysis of variance test for normality (complete samples). Biometrika 52(3/4):591–611. https://doi.org/10.2307/2333709
Uddin MDN, Robinson RW (2017) Responses of plant species diversity and soil physical-chemical-microbial properties to Phragmites australis invasion along a density gradient. Sci Rep 7(1):11007. https://doi.org/10.1038/s41598-017-11205-0
Uddin MN, Robinson RW, Buultjens A et al (2017) Role of allelopathy of Phragmites australis in its invasion processes. J Exp Mar Biol Ecol 486:237–244. https://doi.org/10.1016/j.jembe.2016.10.016
Warren RS, Fell PE, Rozsa R et al (2002) Salt marsh restoration in Connecticut: 20 years of science and management. Restor Ecol 10(3):497–513. https://doi.org/10.1046/j.1526-100X.2002.01031.x
Willcox JD (2013) Response of Phragmites australis to black plastic treatment. Master's Theses. 444. https://opencommons.uconn.edu/gs_theses/444
Whyte R, Trexel-Kroll D, Klarer D et al (2008) The invasion and spread of Phragmites australis during a period of low water in a Lake Erie coastal wetland. J Coastal Res 55:111–120. https://doi.org/10.2112/SI55-19.1
Zimmerman CL, Shirer RR, Corbin JD (2018) Native plant recovery following three years of common reed (Phragmites australis) control. Invasive Plant Sci Manage 11(4):175–180. https://doi.org/10.1017/inp.2018.24
Acknowledgements
We thank Cathy Oliver and Cyrene Simmons for providing access to their property. Building the database for the Short-term Management sites was made possible by the help of Chris Carroll (Anne Arundel County Department of Parks and Recreation). Sampling sites that were part of the Continuous Management sites was made possible by staff of the Chesapeake Wildlife Heritage (Phillips Boyd, Mary Henle, Ned Gerber). We especially thank Phillips for travelling to the sites with us. Access to the data at the Monitoring sites was provided by Christine Rohal. We thank Jack Olson and Matt Ogburn for their efforts in collecting data for the Monitored dataset in 2020. Equipment and vehicles needed for transportation were provided by the Smithsonian Environmental Research Center.
Funding
DW received support from Maryland Sea Grant under award R/EH-3 from the National Oceanic and Atmospheric Administation, U.S. Department of Commerce. SJ was supported by a Maryland Sea Grant Research Fellowship. Matching funds were provided by the Smithsonian Institution, University of Maryland, and Utah State University. SJ was funded with a SeaGrant Fellowship.
Author information
Authors and Affiliations
Contributions
All authors were involved in the design and conduct of the study. HB and SJ analyzed the data and prepared the figures and tables. DW and HB drafted the manuscript and all authors participated in the editing and review process.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Brooks, H., Jacobson, S., Baldwin, A.H. et al. Long-term periodic management of Phragmites australis maintains native brackish wetland plant communities. Wetlands Ecol Manage (2024). https://doi.org/10.1007/s11273-024-09984-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11273-024-09984-1