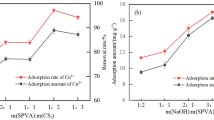
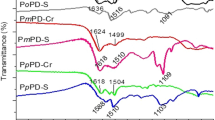
Abstract
The direct and efficient removal of multiple heavy metal ions from acidic wastewater is a challenge. In this study, an efficient adsorbent of polyvinyl alcohol with abundant phosphate functional group (P-PVA) was designed and prepared for the simultaneous removal of multiple heavy metal ions (i.e., Cd2+, Cu2+, and Pb2+) from acidic wastewater. The characterization results indicated that abundant phosphate functional groups can be grafted with polyvinyl alcohol. Under acidic conditions (pH = 3), the adsorption results of P-PVA adsorbent show rapid removal of Cd2+, Cu2+, and Pb2+. The maximum adsorption capacities of 210.21 mg/g, 140.76 mg/g, and 235 mg/g were obtained for Cd2+, Cu2+, and Pb2+ ions, respectively. The adsorbent also shows high adsorption capacity in the presence of humic acid or coexisting cations (i.e., K+, Ca2+, and Mg2+). Competitive adsorption behavior under various systems showed binding strength of P-PVA in the order Pb2+ > Cd2+ > Cu2+. The adsorption mechanism of heavy metal ions was mainly governed by the complexation reaction between the phosphate functional group of P-PVA and heavy metal ions. This new adsorbent has a very high potential for practical application in the treatment of acidic and complexly polluted wastewater.
Graphical Abstract














Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Abdollahi, N., Moussavi, G., & Giannakis, S. (2022). A review of heavy metals’ removal from aqueous matrices by Metal-Organic Frameworks (MOFs): State-of-the art and recent advances. Journal of Environmental Chemical Engineering, 10(3), 107394. https://doi.org/10.1016/j.jece.2022.107394
Azad, H., Mohsennia, M., Cheng, C., & Amini, A. (2021). Facile fabrication of PVB-PVA blend polymer nanocomposite for simultaneous removal of heavy metal ions from aqueous solutions: Kinetic, equilibrium, reusability and adsorption mechanism. Journal of Environmental Chemical Engineering, 9(5). https://doi.org/10.1016/j.jece.2021.106214
Bora, A. J., & Dutta, R. K. (2019). Removal of metals (Pb, Cd, Cu, Cr, Ni, and Co) from drinking water by oxidation-coagulation-absorption at optimized pH. Journal of Water Process Engineering, 31. https://doi.org/10.1016/j.jwpe.2019.100839
Castro-Gonzalez, N. P., Calderon-Sanchez, F., Moreno-Rojas, R., Moreno-Ortega, A., & Tamariz-Flores, J. V. (2017). Health risks in rural populations due to heavy metals found in agricultural soils irrigated with wastewater in the Alto Balsas sub-basin in Tlaxcala and Puebla Mexico. International Journal of Environmental Health Research, 27(6), 476–486. https://doi.org/10.1080/09603123.2017.1386767
Chen, L., Zhou, M., Wang, J., Zhiqin, Z., Chengjiao, D., Xiangxiang, W., et al. (2022). A global meta-analysis of heavy metal(loid)s pollution in soils near copper mines: Evaluation of pollution level and probabilistic health risks. Science of the Total Environment. https://doi.org/10.1016/j.scitotenv.2022.155441
Choi, J., Ide, A., Truong, Y. B., Kyratzis, I. L., & Caruso, R. A. (2013). High surface area mesoporous titanium–zirconium oxide nanofibrous web: a heavy metal ion adsorbent. Journal of Materials Chemistry A, 1(19). https://doi.org/10.1039/c3ta00030c
Choudhary, M., Kumar, R., & Neogi, S. (2020). Activated biochar derived from Opuntia ficus-indica for the efficient adsorption of malachite green dye, Cu(+2) and Ni(+2) from water. Journal of Hazardous Materials, 392, 122441. https://doi.org/10.1016/j.jhazmat.2020.122441
Du, X.-D., Yi, X.-H., Wang, P., Zheng, W., Deng, J., & Wang, C.-C. (2019). Robust photocatalytic reduction of Cr(VI) on UiO-66-NH2(Zr/Hf) metal-organic framework membrane under sunlight irradiation. Chemical Engineering Journal, 356, 393–399. https://doi.org/10.1016/j.cej.2018.09.084
Es-saidi, I., Oulguidoum, A., El Bekkali, C., Bouyarmane, H., Laghzizil, A., & Nunzi, J.-M. (2021). Characterization and valorization of natural phosphate in removing of heavy metals and toxic organic species from water. Journal of African Earth Sciences, 173. https://doi.org/10.1016/j.jafrearsci.2020.104022
Fan, J.-P., Luo, J.-J., Zhang, X.-H., Zhen, B., Dong, C.-Y., Li, Y.-C., et al. (2019). A novel electrospun β-CD/CS/PVA nanofiber membrane for simultaneous and rapid removal of organic micropollutants and heavy metal ions from water. Chemical Engineering Journal, 378. https://doi.org/10.1016/j.cej.2019.122232
Fu, L., Wang, S., Lin, G., Zhang, L., Liu, Q., Fang, J., et al. (2019). Post-functionalization of UiO-66-NH2 by 2,5-Dimercapto-1,3,4-thiadiazole for the high efficient removal of Hg(II) in water. Journal of Hazardous Materials, 368, 42–51. https://doi.org/10.1016/j.jhazmat.2019.01.025
Huang, Z., Zhao, M., Wang, S., Dai, L., Zhang, L., & Wang, C. (2020). Selective recovery of gold ions in aqueous solutions by a novel trithiocyanuric-Zr based MOFs adsorbent. Journal of Molecular Liquids, 298, 112090. https://doi.org/10.1016/j.molliq.2019.112090
Hughes, D. L., Afsar, A., Laventine, D. M., Shaw, E. J., Harwood, L. M., & Hodson, M. E. (2018). Metal removal from soil leachates using DTPA-functionalised maghemite nanoparticles, a potential soil washing technology. Chemosphere, 209, 480–488. https://doi.org/10.1016/j.chemosphere.2018.06.121
Kapoor, D., & Singh, M. P. (2021). 10 - Heavy metal contamination in water and its possible sources. In V. Kumar, A. Sharma, & A. Cerdà (Eds.), Heavy Metals in the Environment (pp. 179–189). Elsevier. https://doi.org/10.1016/B978-0-12-821656-9.00010-9
Kumar, A., Cabral-Pinto, M., Kumar, A., Kumar, M., & Dinis, P. A. (2020). Estimation of Risk to the Eco-Environment and Human Health of Using Heavy Metals in the Uttarakhand Himalaya, India. Applied Sciences, 10(20). https://doi.org/10.3390/app10207078
Li, H., Xiao, D., He, H., Lin, R., & Zuo, P. (2013). Adsorption behavior and adsorption mechanism of Cu(II) ions on amino-functionalized magnetic nanoparticles. Transactions of Nonferrous Metals Society of China, 23(9), 2657–2665. https://doi.org/10.1016/S1003-6326(13)62782-X
Li, C., Yan, Y., Zhang, Q., Zhang, Z., Huang, L., Zhang, J., et al. (2019). Adsorption of Cd(2+) and Ni(2+) from Aqueous Single-Metal Solutions on Graphene Oxide-Chitosan-Poly(vinyl alcohol) Hydrogels. Langmuir, 35(13), 4481–4490https://doi.org/10.1021/acs.langmuir.8b04189
Li, Q., Wang, Y., Li, Y., Li, L., Tang, M., Hu, W., et al. (2022). Speciation of heavy metals in soils and their immobilization at micro-scale interfaces among diverse soil components. Science of the Total Environment, 825, 153862. https://doi.org/10.1016/j.scitotenv.2022.153862
Li, Y.-X., Han, Y.-C., & Wang, C.-C. (2021). Fabrication strategies and Cr(VI) elimination activities of the MOF-derivatives and their composites. Chemical Engineering Journal, 405. https://doi.org/10.1016/j.cej.2020.126648
Lin, S., Bediako, J. K., Cho, C.-W., Song, M.-H., Zhao, Y., Kim, J.-A., et al. (2018). Selective adsorption of Pd(II) over interfering metal ions (Co(II), Ni(II), Pt(IV)) from acidic aqueous phase by metal-organic frameworks. Chemical Engineering Journal, 345, 337–344. https://doi.org/10.1016/j.cej.2018.03.173
Liu, G., Liao, L., Dai, Z., Qi, Q., Wu, J., Ma, L. Q., et al. (2020). Organic adsorbents modified with citric acid and Fe3O4 enhance the removal of Cd and Pb in contaminated solutions. Chemical Engineering Journal, 395. https://doi.org/10.1016/j.cej.2020.125108
Liu, Z., & Zhang, F. S. (2009). Removal of lead from water using biochars prepared from hydrothermal liquefaction of biomass. Journal of Hazardous Materials, 167(1–3), 933–939. https://doi.org/10.1016/j.jhazmat.2009.01.085
Mai, D. D., Bui, T. H., Huan Pham, V., Bui, T. H., Pham, T. K., Nguyen, D. C., & Nguyen, T. L. (2022). Simultaneous adsorption of heavy metals on mesoporous reduced graphene oxide/γ-Fe2O3 nanocomposites. Journal of Porous Materials, 29(6), 1947–1956. https://doi.org/10.1007/s10934-022-01306-1
Meng, Z., Huang, S., Xu, T., Lin, Z., & Wu, J. (2022a). Competitive adsorption, immobilization, and desorption risks of Cd, Ni, and Cu in saturated-unsaturated soils by biochar under combined aging. Journal of Hazardous Materials, 434, 128903. https://doi.org/10.1016/j.jhazmat.2022.128903
Meng, Z., Xu, T., Huang, S., Ge, H., Mu, W., & Lin, Z. (2022b). Effects of competitive adsorption with Ni(II) and Cu(II) on the adsorption of Cd(II) by modified biochar co-aged with acidic soil. Chemosphere, 293, 133621. https://doi.org/10.1016/j.chemosphere.2022.133621
Pan, Y., Liu, Z., Wang, W., Peng, C., Shi, K., & Ji, X. (2016). Highly efficient macroporous adsorbents for toxic metal ions in water systems based on polyvinyl alcohol–formaldehyde sponges. Journal of Materials Chemistry A, 4(7), 2537–2549. https://doi.org/10.1039/c5ta09295g
Peng, W., Li, H., Liu, Y., & Song, S. (2017). A review on heavy metal ions adsorption from water by graphene oxide and its composites. Journal of Molecular Liquids, 230, 496–504. https://doi.org/10.1016/j.molliq.2017.01.064
Qin, G., Niu, Z., Yu, J., Li, Z., Ma, J., & Xiang, P. (2021). Soil heavy metal pollution and food safety in China: Effects, sources and removing technology. Chemosphere, 267, 129205. https://doi.org/10.1016/j.chemosphere.2020.129205
Regazzoni, A. E. (2020). Adsorption kinetics at solid/aqueous solution interfaces: On the boundaries of the pseudo-second order rate equation. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 585. https://doi.org/10.1016/j.colsurfa.2019.124093
Rego, R. M., Sriram, G., Ajeya, K. V., Jung, H. Y., Kurkuri, M. D., & Kigga, M. (2021). Cerium based UiO-66 MOF as a multipollutant adsorbent for universal water purification. Journal of Hazardous Materials, 416, 125941. https://doi.org/10.1016/j.jhazmat.2021.125941
Sekhavat Pour, Z., & Ghaemy, M. (2016). Preparation of poly(vinylalcohol)/poly(acrylamide-co-vinyl imidazole)/γ-Fe2O3semi-IPN nanocomposite and their application for removal of heavy metal ions from water. Polymers for Advanced Technologies, 27(12), 1557–1568. https://doi.org/10.1002/pat.3830
Shakya, A., & Agarwal, T. (2019). Removal of Cr(VI) from water using pineapple peel derived biochars: Adsorption potential and re-usability assessment. Journal of Molecular Liquids, 293. https://doi.org/10.1016/j.molliq.2019.111497
Shi, W., Chu, Y., Xia, M., Wang, F., & Fu, C. (2021). The adsorption performance and micro-mechanism of MoS2/montmorillonite composite to atenolol and acebutolol: Adsorption experiments and a novel visual study of interaction. Ecotoxicology and Environmental Safety, 213, 111993. https://doi.org/10.1016/j.ecoenv.2021.111993
Sitko, R., Turek, E., Zawisza, B., Malicka, E., Talik, E., Heimann, J., et al. (2013). Adsorption of divalent metal ions from aqueous solutions using graphene oxide. Dalton Transactions, 42(16), 5682–5689. https://doi.org/10.1039/c3dt33097d
Tan, P., Sun, J., Hu, Y., Fang, Z., Bi, Q., Chen, Y., & Cheng, J. (2015). Adsorption of Cu(2+), Cd(2+) and Ni(2+) from aqueous single metal solutions on graphene oxide membranes. Journal of Hazardous Materials, 297, 251–260. https://doi.org/10.1016/j.jhazmat.2015.04.068
Tang, J., Chen, Y., Zhao, M., Wang, S., & Zhang, L. (2021). Phenylthiosemicarbazide-functionalized UiO-66-NH2 as highly efficient adsorbent for the selective removal of lead from aqueous solutions. Journal of Hazardous Materials, 413, 125278. https://doi.org/10.1016/j.jhazmat.2021.125278
Tang, Z., Xu, D., Chen, H., Tang, S., He, C., Ning, S., et al. (2023). Adsorption of Pd(II) from wastewater by a functionalized metal-organic framework with aminopyridine structure: Performance and mechanism. Microporous and Mesoporous Materials, 353, 112519. https://doi.org/10.1016/j.micromeso.2023.112519
Tang, Y., Chen, Z., Wen, Q., Liu, B., & Huang, X. (2022). Magnetic powdery acrylic polymer with ultrahigh adsorption capacity for atenolol removal: Preparation, characterization, and microscopic adsorption mechanism. Chemical Engineering Journal, 446. https://doi.org/10.1016/j.cej.2022.137175
Tassanapukdee, Y., Prayongpan, P., & Songsrirote, K. (2021). Removal of heavy metal ions from an aqueous solution by CS/PVA/PVP composite hydrogel synthesized using microwaved-assisted irradiation. Environmental Technology & Innovation, 24. https://doi.org/10.1016/j.eti.2021.101898
Tchinsa, A., Hossain, M. F., Wang, T., & Zhou, Y. (2021). Removal of organic pollutants from aqueous solution using metal organic frameworks (MOFs)-based adsorbents: A review. Chemosphere, 284, 131393. https://doi.org/10.1016/j.chemosphere.2021.131393
Tran, H. N., Nguyen, D. T., Le, G. T., Tomul, F., Lima, E. C., Woo, S. H., et al. (2019). Adsorption mechanism of hexavalent chromium onto layered double hydroxides-based adsorbents: A systematic in-depth review. Journal of Hazardous Materials, 373, 258–270. https://doi.org/10.1016/j.jhazmat.2019.03.018
Wang, J., & Guo, X. (2023). Adsorption kinetics and isotherm models of heavy metals by various adsorbents: An overview. Critical Reviews in Environmental Science and Technology, 53(21), 1837–1865. https://doi.org/10.1080/10643389.2023.2221157
Xiao, H., & Liu, S. (2018). Zirconium phosphate (ZrP)-based functional materials: Synthesis, properties and applications. Materials & Design, 155, 19–35. https://doi.org/10.1016/j.matdes.2018.05.041
Xu, H., Zhu, S., Xia, M., & Wang, F. (2021). Rapid and efficient removal of diclofenac sodium from aqueous solution via ternary core-shell CS@PANI@LDH composite: Experimental and adsorption mechanism study. Journal of Hazardous Materials, 402, 123815. https://doi.org/10.1016/j.jhazmat.2020.123815
Yan, Y., Chu, Y., Khan, M. A., Xia, M., Shi, M., Zhu, S., et al. (2022). Facile immobilization of ethylenediamine tetramethylene-phosphonic acid into UiO-66 for toxic divalent heavy metal ions removal: An experimental and theoretical exploration. Science of the Total Environment, 806, 150652. https://doi.org/10.1016/j.scitotenv.2021.150652
Yang, S., Feng, W., Wang, S., Chen, L., Zheng, X., Li, X., & Zhou, D. (2021). Farmland heavy metals can migrate to deep soil at a regional scale: A case study on a wastewater-irrigated area in China. Environmental Pollution, 281, 116977. https://doi.org/10.1016/j.envpol.2021.116977
Yu, N., Wu, K., & Tao, L. (2021). Synchronous reduction-fixation of reducible heavy metals from aqueous solutions: Application of novel mesoporous MFT/SBA-15 composite materials. Chemosphere, 276, 130112. https://doi.org/10.1016/j.chemosphere.2021.130112
Yu, Y., An, Q., Jin, L., Luo, N., Li, Z., & Jiang, J. (2020). Unraveling sorption of Cr (VI) from aqueous solution by FeCl3 and ZnCl2-modified corn stalks biochar: Implicit mechanism and application. Bioresource Technology, 297. https://doi.org/10.1016/j.biortech.2019.122466
Zamora-Ledezma, C., Negrete-Bolagay, D., Figueroa, F., Zamora-Ledezma, E., Ni, M., Alexis, F., & Guerrero, V. H. (2021). Heavy metal water pollution: A fresh look about hazards, novel and conventional remediation methods. Environmental Technology & Innovation, 22. https://doi.org/10.1016/j.eti.2021.101504
Zeng, X., Zhang, G., Wen, J., Li, X., Zhu, J., & Wu, Z. (2023). Simultaneous removal of aqueous same ionic type heavy metals and dyes by a magnetic chitosan/polyethyleneimine embedded hydrophobic sodium alginate composite: Performance, interaction and mechanism. Chemosphere, 318, 137869. https://doi.org/10.1016/j.chemosphere.2023.137869
Zhang, P., Zou, K., Yuan, L., Liu, J., Liu, B., Qing, T.-P., & Feng, B. (2022). A biomass resource strategy for alginate-polyvinyl alcohol double network hydrogels and their adsorption to heavy metals. Separation and Purification Technology, 301, 122050. https://doi.org/10.1016/j.seppur.2022.122050
Zhao, G., Zhang, H., Fan, Q., Ren, X., Li, J., Chen, Y., & Wang, X. (2010). Sorption of copper(II) onto super-adsorbent of bentonite-polyacrylamide composites. Journal of Hazardous Materials, 173(1–3), 661–668. https://doi.org/10.1016/j.jhazmat.2009.08.135
Acknowledgements
This work was funded by the basic scientific research of central universities (2232020A-10) (China), and the Joint Foundation of Iron and Steel, National Natural Science Foundation of China (U1660107).
Author information
Authors and Affiliations
Contributions
Xi Wang: Methodology, Investigation, Writing-original draft. Asif Hussain: Supervision, Writing-Reviewing & Editing. Hongyi Zhu: material characterization and investigation. Yongkuan Li: investigation. Xing Wang: investigation. Dengxin Li: Key concepts and Supervision.
Corresponding author
Ethics declarations
Competing Interest
The authors declare no competing or financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Hussain, A., Zhu, H. et al. Simultaneous Adsorption of Multi-Heavy Metal Ions from Wastewater via Grafted Phosphate Functionality on Polyvinyl Alcohol. Water Air Soil Pollut 234, 638 (2023). https://doi.org/10.1007/s11270-023-06666-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-023-06666-1