Abstract
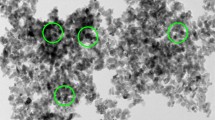
The magnetic nanocomposite TiO2/Fe3O4 coupled with persulfate (PS) under UV irradiation for methyl orange (MO) degradation in aqueous solution was investigated in this study. The features of TiO2/Fe3O4 were characterized via SEM, XPS, UV–Vis, and MPMS. The effect of several operating parameters including PS concentration, TiO2/Fe3O4 dosage, MO concentration, and pH was assessed in UV/TiO2/Fe3O4/PS system. The removal of MO via UV/TiO2/Fe3O4/PS reached 99.5% under the following conditions: [PS] = 60 mM; [TiO2/Fe3O4] = 0.4 g/L; [MO] = 20 mg/L; pH = 3.0; T = 25 ℃; UV = 300 W; and t = 150 min. In addition, possible mechanisms of TiO2/Fe3O4 photocatalysis, PS activation, and MO degradation were analyzed. Moreover, TiO2/Fe3O4 was easily separated from the reacted solution by magnets and had relatively stable recycling performance. Overall, the application of UV/TiO2/Fe3O4/PS can be introduced as an effective method for MO and even dyes removal.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Abbas, M., Rao, B. P., Reddy, V., & Kim, C. (2014). Fe3O4/TiO2 core/shell nanocubes: Single-batch surfactantless synthesis, characterization and efficient catalysts for methylene blue degradation. Ceramics International, 40, 11177–11186. https://doi.org/10.1016/j.ceramint.2014.03.148
Adegoke, K. A., & Bello, O. S. (2015). Dye sequestration using agricultural wastes as adsorbents. Water Resources and Industry, 12, 8–24. https://doi.org/10.1016/j.wri.2015.09.002
Chen, Y., Yuan, T., Wang, F., Hu, J., & Tu, W. (2016). Magnetically separable Fe3O4@TiO2 nanospheres: Preparation and photocatalytic activity. Journal Materials Science-Materials El., 27, 9983–9988. https://doi.org/10.1007/s10854-016-5330-7
Chiou, C. H., Wu, C. Y., & Juang, R. S. (2008). Influence of operating parameters on photocatalytic degradation of phenol in UV/TiO2 process. Chemical Engineering Journal, 139, 322–329. https://doi.org/10.1016/j.cej.2007.08.002
Chong, M. N., Jin, B., Chow, C. W., & Saint, C. (2010). Recent developments in photocatalytic water treatment technology: A review. Water Research, 44, 2997–3027. https://doi.org/10.1016/j.watres.2010.02.039
Chu, A. C., Sahu, R. S., Chou, T. H., & Shih, Y. H. (2021). Magnetic Fe3O4@TiO2 nanocomposites to degrade bisphenol A, one emerging contaminant, under visible and long wavelength UV light irradiation. Journal of Environmental Chemical Engineering, 9, 105539. https://doi.org/10.1016/j.jece.2021.105539
De Luca, A., He, X., Dionysiou, D. D., Dantas, R. F., & Esplugas, S. (2017). Effects of bromide on the degradation of organic contaminants with UV and Fe2+ activated persulfate. Chemical Engineering Journal, 318, 206–213. https://doi.org/10.1016/j.cej.2016.06.066
Dubey, M., Kumar, R., Srivastava, S. K., & Joshi, M. (2021). Visible light induced photodegradation of chlorinated organic pollutants using highly efficient magnetic Fe3O4/TiO2 nanocomposite. Optik, 243, 167309. https://doi.org/10.1016/j.ijleo.2021.167309
El Harfi, S., & El Harfi, A. (2017). Classifications, properties and applications of textile dyes: A review Applied Journal Environment Engineering Science, 3, 311–320.
Eskandarian, M. R., Rasoulifard, M. H., Fazli, M., Ghalamchi, L., & Choi, H. (2019). Synergistic decomposition of imidacloprid by TiO2-Fe3O4 nanocomposite conjugated with persulfate in a photovoltaic-powered UV-LED photoreactor. Korean Journal of Chemical Engineering, 36, 965–974. https://doi.org/10.1007/s11814-018-0230-1
Gao, Y., Luo, J., Song, T., & Yu, X. (2021). Research progress on nano-Fe0/PS system for degradation of refractory organics in aqueous solution. Journal of Environmental Chemical Engineering, 9, 105345. https://doi.org/10.1016/j.jece.2021.105345
Hernandez-Maya, R., Solís-López, M., & Th-Th, C. (2018). Photocatalytic degradation of Orange G using TiO2/Fe3O4 nanocomposites. Journal Materials Science-Materials El., 29, 15436–15444. https://doi.org/10.1007/s10854-018-9069-1
Karimi, H. (2020). Removal of RR198 dye by TiO2/Fe3O4/persulfate nanoparticles under UV-LED irradiation and comparison of OFAT and CCD experimental design in RSM modelling. Indian Journal Chemistry Technology, 27, 283–293.
Kermani, M., Kakavandi, B., Farzadkia, M., Esrafili, A., Jokandan, S. F., & Shahsavani, A. (2018). Catalytic ozonation of high concentrations of catechol over TiO2@Fe3O4 magnetic core-shell nanocatalyst: Optimization, toxicity and degradation pathway studies. Journal of Cleaner Production, 192, 597–607. https://doi.org/10.1016/j.jclepro.2018.04.274
Lee, S. Y., & Park, S. J. (2013). TiO2 photocatalyst for water treatment applications. Journal of Industrial and Engineering Chemistry, 19, 1761–1769. https://doi.org/10.1016/j.jiec.2013.07.012
Li, Y. S., Church, J. S., & Woodhead, A. L. (2012). Infrared and Raman spectroscopic studies on iron oxide magnetic nano-particles and their surface modifications. Journal of Magnetism and Magnetic Materials, 324, 1543–1550. https://doi.org/10.1016/j.jmmm.2011.11.065
Liu, C. C., Hsieh, Y. H., Lai, P. F., Li, C. H., & Kao, C. L. (2006). Photodegradation treatment of azo dye wastewater by UV/TiO2 process. Dyes and Pigments., 68, 191–195. https://doi.org/10.1016/j.dyepig.2004.12.002
Liu, Y., Li, Z., Green, M., Just, M., Li, Y. Y., & Chen, X. (2017). Titanium dioxide nanomaterials for photocatalysis. Journal Physics D. Applied Physics, 50, 193003. https://doi.org/10.1088/1361-6463/aa6500
Mercyrani, B., Hernandez-Maya, R., Solís-López, M., Th-Th, C., & Velumani, S. (2018). Photocatalytic degradation of Orange G using TiO2/Fe3O4 nanocomposites. Journal Materials Science-Materials El., 29, 15436–15444. https://doi.org/10.1007/s10854-018-9069-1
Nasirian, M., & Mehrvar, M. (2016). Modification of TiO2 to enhance photocatalytic degradation of organics in aqueous solutions. Journal of Environmental Chemical Engineering, 4, 4072–4082. https://doi.org/10.1016/j.jece.2016.08.008
Oh, W. D., & Lim, T. T. (2019). Design and application of heterogeneous catalysts as peroxydisulfate activator for organics removal: An overview. Chemical Engineering Journal, 358, 110–133. https://doi.org/10.1016/j.cej.2018.09.203
Razip, N. I. M., Lee, K. M., Lai, C. W., & Ong, B. H. (2019). Recoverability of Fe3O4/TiO2 nanocatalyst in methyl orange degradation. Materials Research Express, 6, 075517. https://doi.org/10.1088/2053-1591/ab176e
Rodriguez, S., Vasquez, L., Costa, D., Romero, A., & Santos, A. (2014). Oxidation of Orange G by persulfate activated by Fe (II), Fe (III) and zero valent iron (ZVI). Chemosphere, 101, 86–92. https://doi.org/10.1016/j.chemosphere.2013.12.037
Saqlain, S., Cha, B. J., Kim, S. Y., Ahn, T. K., Park, C., Oh, J. M., Jeong, E. C., Seo, H. O., & Kim, Y. D. (2020). Visible light-responsive Fe-loaded TiO2 photocatalysts for total oxidation of acetaldehyde: Fundamental studies towards large-scale production and applications. Applied Surface Science, 505, 144160. https://doi.org/10.1016/j.apsusc.2019.144160
Song, C., Wang, L., Ren, J., Lv, B., Sun, Z., Yan, J., Li, X., & Liu, J. (2016). Comparative study of diethyl phthalate degradation by UV/H2O2 and UV/TiO2: Kinetics, mechanism, and effects of operational parameters. Environment Science Pollution Reasearch, 23, 2640–2650. https://doi.org/10.1007/s11356-015-5481-8
Song, T., Li, R., Li, N., & Gao, Y. (2019). Research progress on the application of nanometer TiO2 photoelectrocatalysis technology in wastewater treatment. Science of Advanced Materials, 11, 158–165. https://doi.org/10.1166/sam.2019.3411
Yao, K. F., Peng, Z., Liao, Z. H., & Chen, J. J. (2009). Preparation and photocatalytic property of TiO2-Fe3O4 core–shell nanoparticles. Journal Nanoscience and Nanotechnology, 9, 1458–1461. https://doi.org/10.1166/jnn.2009.c178
Yao, H., Fan, M., Wang, Y., Luo, G., & Fei, W. (2015). Magnetic titanium dioxide based nanomaterials: Synthesis, characteristics, and photocatalytic application in pollutant degradation. Journal Materials Chemistry a., 3, 17511–17524. https://doi.org/10.1039/C5TA03215F
Youssef, N. A., Shaban, S. A., Ibrahim, F. A., & Mahmoud, A. S. (2016). Degradation of methyl orange using Fenton catalytic reaction. Egypt Journal Petrology, 25, 317–321. https://doi.org/10.1016/j.ejpe.2015.07.017
Yu, X., Lin, X., Feng, W., & Li, W. (2019). Effective removal of tetracycline by using bio-templated synthesis of TiO2/Fe3O4 heterojunctions as a UV–Fenton catalyst. Catalysis Letters, 149, 552–560. https://doi.org/10.1007/s10562-018-2544-8
Yuan, G. E., Qin, Y., Feng, M., Zhang, W., Ru, X., & Zhang, X. (2021). Synergistic activation of persulfate by natural chalcocite and ferrous ions by promoting the cycling of Fe3+/Fe2+ couple for degradation of organic pollutants. Ecotoxicol Environment Safety, 212, 111975. https://doi.org/10.1016/j.ecoenv.2021.111975
Zhang, Y., Yu, X., Jia, Y., Jin, Z., Liu, J., & Huang, X. (2011). A facile approach for the synthesis of Ag-coated Fe3O4@TiO2 core/shell microspheres as highly efficient and recyclable photocatalysts. European Journal of Inorganic Chemistry, 2011, 5096–5104. https://doi.org/10.1002/ejic.201100707
Zhang, B. T., Zhang, Y., Teng, Y., & Fan, M. (2015). Sulfate radical and its application in decontamination technologies. Critical Review Environmrntal Science Tecnilogy, 45, 1756–1800. https://doi.org/10.1080/10643389.2014.970681
Zhou, Z. H., Wang, J., Liu, X., & Chan, H. S. O. (2001). Synthesis of Fe3O4 nanoparticles from emulsions. Journal of Materials Chemistry, 11, 1704–1709. https://doi.org/10.1039/B100758K
Bohra, M., Agarwal, N., Singh, V., 2019. A short review on Verwey transition in nanostructured Fe3O4 materials. Journal Nanomaterials. 2019. https://doi.org/10.1155/2019/8457383.
Duan, X. L., Yuan, C. G., Guo, Q., Niu, S. L., He, K. Q., Xia, G. W., 2021. Preparation of halloysite nanotubes-encapsulated magnetic microspheres for elemental mercury removal from coal-fired flue gas. Journal Hazard Materials 406, 124683. https://doi.org/10.1016/j.jhazmat.2020.124683 Get rights and content
Tahergorabi, M., Esrafili, A., Shirzad-Siboni, M., Kermani, M., 2020. Photodegradation of catechol in water over magnetically separable Fe3O4/TiO2 composite photocatalysts. International Journal Environment An. Ch. 1-19. https://doi.org/10.1080/03067319.2020.1785441
Funding
This work was supported by the Jilin Provincial Department of Science and Technology of China (No. YDZJ202201ZYTS681 and No. YDZJ202201ZYTS630).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Song, T., Gao, Y., Hu, R. et al. Degradation of Methyl Orange in Aqueous Solution via Magnetic TiO2/Fe3O4 Conjugated with Persulfate. Water Air Soil Pollut 234, 508 (2023). https://doi.org/10.1007/s11270-022-05982-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05982-2