Abstract
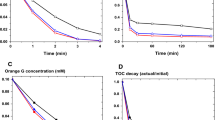
In this work, the advanced oxidation technology was investigated through the degradation of a solution composed of four textile dyes, more complex than the solutions used in similar researches. For this purpose, heterogeneous photo-Fenton process was applied, using the mineral pyrite as iron catalyst and sunlight as a radiation source. This research aimed to identify and determine the most appropriate experimental conditions to conduct the treatment, revealing the influence of the variables pH and iron and H2O2 concentrations on degradation efficiency. The highest degradation (99.74%) and organic matter conversion (95.03%) were observed at pH 3, using 100 mg·L−1 of H2O2 and 0.5 g·L−1 of pyrite for 60 min. Under these conditions, non-linear pseudo-first order and linear pseudo-first order and pseudo-second order kinetic models described the reaction kinetics the treatment (accuracy > 91%). The toxicity analysis revealed a possible formation of toxic intermediates, as the natural development of seed species and the mollusc Biomphalaria glabrata was affected. In general, the adequacy of the proposed treatment and the good use of pyrite in Fenton’s heterogeneous systems were evidenced.







Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
Code Availability
Not applicable.
References
Behnajady, M. A., Modirshahla, N., & Ghanbary, F. (2007). A kinetic model for the decolorization of CI Acid Yellow 23 by Fenton process. Journal of Hazardous Materials, 148, 98–102. https://doi.org/10.1016/j.jhazmat.2007.02.003
Boczkaj, G., & Fernandes, A. (2017). Wastewater treatment by means of advanced oxidation processes at basis pH conditions: A review. Chemical Engineering Journal, 320, 608–633. https://doi.org/10.1016/j.cej.2017.03.084
Couto, C. F., Lange, L. C., & Amaral, M. C. S. (2018). A critical review on membrane separation processes applied to remove pharmaceutically active compounds from water and wastewater. J Water Process Eng., 26, 156–175. https://doi.org/10.1016/j.jwpe.2018.10.010
Cuevas-García, R., Téllez-Romero, J. G., Ramírez, J., Sarabia-Bañuelos, P., Puente-Lee, I., Salcedo-Luna, C., Hernandez-Gonzalez, S., & Nolasco-Arizmendi, V. A. (2021). Effect of the preparation method on particle size and reaction selectivity on naphthalene hydrogenation over Ni/H-MOR catalysts. Catalysis Today, 360, 63–71. https://doi.org/10.1016/j.cattod.2019.08.044
Diao, Z. H., Liu, J. J., Hu, Y. X., Kong, L. J., Jiang, D., & Xu, X. R. (2017). Comparative study of Rhodamine B degradation by the systems pyrite/H2O2 and pyrite/persulfate: Reactivity, stability, products and mechanism. Separation and Purification Technology, 184, 374–383. https://doi.org/10.1016/j.seppur.2017.05.016
Emídio, E. S., Hammer, P., & Nogueira, R. F. P. (2020). Simultaneous degradation of the anticancer drugs 5-fluorouracil and cyclophosphamide using a heterogeneous photo-Fenton process based on copper-containing magnetites (Fe3-xCuxO4). Chemosphere, 241, 124990. https://doi.org/10.1016/j.chemosphere.2019.124990
Farshchi, M. E., Aghdasinia, H., & Khataee, A. (2019). Heterogeneous Fenton reaction for elimination of Acid Yellow 36 in both fluidized-bed and stirred-tank reactors: Computational fluid dynamics versus experiments. Water Research, 151, 203–214. https://doi.org/10.1016/j.watres.2018.12.011
Gil-Lozano, C., Davila, A. F., Losa-Adams, E., Fairen, A. G., & Gago-Duport, L. (2017). Quantifying Fenton reaction pathways driven by self-generated H2O2 on pyrite surfaces. Science and Reports, 7, 43703. https://doi.org/10.1038/srep43703
Gomes Júnior, O., Batista, L. L., Ueira-Vieira, C., Sousa, R. M., Starling, M. C. V., & Trovó, A. G. (2020). Degradation mechanism of fipronil and its transformation products, matrix effects and toxicity during the solar/photo-Fenton process using ferric citrate complex. Journal of Environmental Management, 269, 110756. https://doi.org/10.1016/j.jenvman.2020.110756
Guo, X., Wang, K., & Xu, Y. (2019). Tartaric acid enhanced CuFe2O4-catalyzed heterogeneous photo-Fenton-like degradation of methylene blue. Materials Science and Engineering B, 245, 75–84. https://doi.org/10.1016/j.mseb.2019.05.015
Hayati, F., Isari, A. A., Anvaripour, B., Fattahi, M., & Kakavandi, B. (2020). Ultrasound-assisted photocatalytic degradation of sulfadiazine using MgO@CNT heterojunction composite: Effective factors, pathway and biodegradability studies. Chemical Engineering Journal, 381, 122636. https://doi.org/10.1016/j.cej.2019.122636
Huang, S., Zhang, Q., Liu, P., Ma, S., Xie, B., Yang, K., & Zhao, Y. (2020). Novel up-conversion carbon quantum dots/α-FeOOH nanohybrids eliminate tetracycline and its related drug resistance in visible-light responsive Fenton system. Applied Catalysis, b: Environmental, 263, 118336. https://doi.org/10.1016/j.apcatb.2019.118336
Kalantary RR, Moradi M, Pirsaheb M, Esrafili A, Jafari AJ, Gholami M, Vasseghian Y, Antolini E, Dragoi EN (2019). Enhanced photocatalytic inactivation of E. coli by natural pyrite in presence of citrate and EDTA as effective chelating agents: Experimental evaluation and kinetic and ANN models. The Journal of Environmental Chemical Engineering, 7, 102906. https://doi.org/10.1016/j.jece.2019.102906.
Khataee, A., Gholami, P., & Sheydaei, M. (2016). Heterogeneous Fenton process by natural pyrite for removal of a textile dye from water: Effect of parameters and intermediate identification. Journal of the Taiwan Institute of Chemical Engineers, 58, 366–373. https://doi.org/10.1016/j.jtice.2015.06.015
Kollias, K., Mylona, E., Adam, K., Chrysochoou, M., Papassiopi, N., & Xenidis, A. (2019). Characterization of phosphate coating formed on pyrite surface to prevent oxidation. Journal of Applied Geochemistry, 110, 104435. https://doi.org/10.1016/j.apgeochem.2019.104435
Kumar, M. S., Sonawane, S. H., Bhanvase, B. A., & Bethi, B. (2018). Treatment of ternary dye wastewater by hydrodynamic cavitation combined with other advanced oxidation processes (AOP’s). J Water Process Eng., 23, 250–256. https://doi.org/10.1016/j.jwpe.2018.04.004
Kumar, A., Rana, A., Sharma, G., Naushad, M., Dhiman, P., Kumari, A., & Stadler, F. J. (2019). Solar active nano-Zn1-xMgxFe2O4 as a magnetically separable sustainable photocatalyst for degradation of sulfadiazine antibiotic. Journal of Molecular Liquids, 290, 111177. https://doi.org/10.1016/j.molliq.2019.111574
Le, T. X. H., Van Nguyen, T., Yacouba, Z. A., Zoungrana, L., Avril, F., Nguyen, D. L., Petit, E., Mendret, J., Bonniol, V., Bechelany, M., et al. (2017). Correlation between degradation pathway and toxicity of acetaminophen and its by-products by using the electro-Fenton process in aqueous media. Chemosphere, 172, 1–9. https://doi.org/10.1016/j.chemosphere.2016.12.060
Nguyen, V. H., Smith, S. M., Wantala, K., & Kajitvichyanukul, P. (2020). Photocatalytic remediation of persistent organic pollutants (POPs): A review. Arabian Journal of Chemistry, 13, 8309–8337. https://doi.org/10.1016/j.arabjc.2020.04.028
Núñez, J., Yeber, M., Cisternas, N., Thibaut, R., Medina, P., & Carrasco, C. (2019). Application of electrocoagulation for the efficient pollutants removal to reuse the treated wastewater in the dyeing process of the textile industry. Journal of Hazardous Materials, 371, 705–711. https://doi.org/10.1016/j.jhazmat.2019.03.030
Oliveira, C. M., Müller, T. G., André, R. A., Oliveira, E. M., Oliveira, E. M., Peterson, M., & Raupp-Pereira, F. (2019). Pyrite from coal mining: High-energy milling and analysis of the electrical and optical properties. Materials Letters, 253, 339–342. https://doi.org/10.1016/j.matlet.2019.07.003
Pham, A. L. T., Sedlak, D. L., & Doyle, F. M. (2012). Dissolution of mesoporous silica supports in aqueous solutions: Implications for mesoporous silica-based water treatment processes. Applied Catalysis, b: Environmental, 126, 258–264. https://doi.org/10.1016/j.apcatb.2012.07.018
Rede, D., Santos, L. H., Ramos, S., Oliva-Teles, F., Antão, C., Sousa, S. R., & Delerue-Matos, C. (2016). Ecotoxicological impact of two soil remediation treatments in Lactuca sativa seeds. Chemosphere, 159, 193–198. https://doi.org/10.1016/j.chemosphere.2016.06.002
Ribeiro, J. P., & Nunes, M. I. (2021). Recent trends and developments in Fenton processes for industrial wastewater treatment - A critical review. Environmental Research, 197, 110957. https://doi.org/10.1016/j.envres.2021.110957
Rubeena, K. K., Reddy, P. H. P., Laiju, A. R., & Nidheesh, P. V. (2018). Iron impregnated biochars as heterogeneous Fenton catalyst for the degradation of acid red 1 dye. Journal of Environmental Management, 226, 320–328. https://doi.org/10.1016/j.jenvman.2018.08.055
Santana, R. M. R., Nascimento, G. E., Napoleão, D. C., & Duarte, M. M. B. (2017). Degradation and kinetic study of Reactive blue BF-5G and Remazol red RB 133% dyes using Fenton and photo-Fenton process. Revista Eletrônica em Gestão, Educação e Tecnologia Ambiental, 21, 104–118. https://doi.org/10.5902/2236117027358
Santana, R. M. R., Napoleão, D. C., Santos Júnior, S. G., Gomes, R. K. M., Moraes, N. F., Zaidan, L. E. M. C., Elihimas, D. R. M., Nascimento, G. E., & Duarte, M. M. B. (2021). Photo-Fenton process under sunlight irradiation for textile wastewater degradation: Monitoring of residual hydrogen peroxide by spectrophotometric method and modeling artificial neural network models to predict treatment. Chemical Papers, 75, 2305–2316. https://doi.org/10.1007/s11696-020-01449-y
Santos, M. M. M., Duarte, M. M. M. B., Nascimento, G. E., Souza, N. B. G. D., & Rocha, O. R. S. D. (2019). Use of TiO2 photocatalyst supported on residues of polystyrene packaging and its applicability on the removal of food dyes. Environmental Technology, 40, 1494–1507. https://doi.org/10.1080/09593330.2017.1423396
Shaida, M. A., Sen, A. K., & Dutta, R. K. (2018). Alternate use of sulphur rich coals as solar photo-Fenton agent for degradation of toxic azo dyes. Journal of Cleaner Production, 195, 1003–1014. https://doi.org/10.1016/j.jclepro.2018.05.286
Shi, X., Tian, A., You, J., Yang, H., Wang, Y., & Xue, X. (2018). Degradation of organic dyes by a new heterogeneous Fenton reagent-Fe2GeS4 nanoparticle. Journal of Hazardous Materials, 353, 182–189. https://doi.org/10.1016/j.jhazmat.2018.04.018
Vu, A. T., Xuan, T. N., & Lee, C. H. (2019). Preparation of mesoporous Fe2O3·SiO2 composite from rice husk as an efficient heterogeneous Fenton-like catalyst for degradation of organic dyes. Journal of Water Process Engineering, 28, 169–180. https://doi.org/10.1016/j.jwpe.2019.01.019
Wang, H., Chen, T., Chen, D., Zou, X., Li, M., Huang, F., Sun, F., Wang, C., Shu, D., & Liu, H. (2020). Sulfurized olitic hematite as a heterogeneous Fenton-like catalyst for tetracycline antibiotic degradation. Applied Catalysis, B: Environmental, 260, 118203. https://doi.org/10.1016/j.apcatb.2019.118203
Wang, F. P., Zhao, X. J., Wahid, F., Zhao, X. Q., Qin, X. T., Bai, H., & Jia, S. R. (2021). Sustainable, superhydrophobic membranes based on bacterial cellulose for gravity-driven oil/water separation. Carbohydrate Polymers, 253, 117220. https://doi.org/10.1016/j.carbpol.2020.117220
Wu, D. L., Chen, Y. F., Zhang, Y. L., Feng, Y., & Shih, K. M. (2015). Ferric iron enhanced chloramphenicol oxidation in pyrite (FeS2) induced Fenton-like reactions. Separation and Purification Technology, 154, 60–67. https://doi.org/10.1016/j.seppur.2015.09.016
Yang, Q., Ma, Y., Chen, F., Yao, F., Sun, J., Wang, S., Yi, K., Hou, L., Li, X., & Wang, D. (2019). Recent advances in photo-activated sulfate radical-advanced oxidation process (SR-AOP) for refractory organic pollutants removal in water. Chemical Engineering Journal, 378, 122149. https://doi.org/10.1016/j.cej.2019.122149
Young, B. J., Riera, N. I., Beily, M. E., Bres, P. A., Crespo, D. C., & Ronco, A. E. (2012). Toxicity of the effluent from an anaerobic bioreactor treating cereal residues on Lactuca sativa. Ecotoxicology and Environmental Safety, 76, 182–186. https://doi.org/10.1016/j.ecoenv.2011.09.019
Zeng, L., Gong, J., Dan, J., Li, S., Zhang, J., Pu, W., & Yang, C. (2019). Novel visible light enhanced Pyrite-Fenton system toward ultrarapid oxidation of p-nitrophenol: Catalytic activity, characterization and mechanism. Chemosphere, 228, 232–240. https://doi.org/10.1016/j.chemosphere.2019.04.103
Zhang, H., Niu, J., Yin, X., Guo, Y., & Cheng, F. (2020). Role of inherent pyrite in coal on physicochemical structure of activated carbon and adsorption capacity. Fuel, 262, 116527. https://doi.org/10.1016/j.fuel.2019.116527
Zheng, Z., Wang, Z., Wang, X., & Liu, D. (2019). Blue light-triggered chemical reactions underlie phosphate deficiency-induced inhibition of root elongation of arabidopsis seedlings grown in Petri dishes. Molecular Plant, 12, 1515–1523. https://doi.org/10.1016/j.molp.2019.08.001
Zhu, X., Li, J., Xie, B., Feng, D., & Li, Y. (2020). Accelerating effects of biochar for pyrite-catalyzed Fenton-like oxidation of herbicide 2,4-D. Chemical Engineering Journal, 391, 123605. https://doi.org/10.1016/j.cej.2019.123605
Acknowledgements
Authors thank to Fundação de Amparo à Ciência e Tecnologia de Pernambuco, Núcleo de Química Analítica Avançada de Pernambuco—NUQAAPE (FACEPE, APQ-0346-1.06/14 process), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – Brasil (CAPES) and Fundação de Apoio ao Desenvolvimento (FADE/UFPE).
Funding
This study was financed in part by the Coordenação e Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate and for Publication
All authors agree.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Rocha Santana, R.M., Napoleão, D.C., Gonzaga dos Santos Júnior, S. et al. Sunlight Irradiated Pyrite-Fenton System for Advanced Oxidative Treatment of Textile Dyes Mixture. Water Air Soil Pollut 233, 170 (2022). https://doi.org/10.1007/s11270-022-05629-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05629-2