Abstract
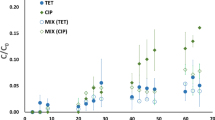
Ciprofloxacin, griseofulvin, and rifampicin are three human antibiotics that are also widely used in the shrimp culture of Cangio coastal wetland (Vietnam, 10° 24’ 38” N, 106° 57’ 17” E). They have been detected in shrimp larvae pond and receiving water bodies. However, the environmental fate of these antibiotics in coastal wetland milieu is currently unknown. The aim of this study was to determine the degradation potential of these antibiotics in water and sediments from Cangio coastal wetlands. The effects of light, microbial activities, and presence of sediments on the degradation of all three antibiotics were investigated in “water-only” and “water–sediment” experiments. Results indicate that the environmental fate of those antibiotics was quite complex. Photodegradation seemed to play a major role in “water-only” system, since shorter t 1/2 was observed for ciprofloxacin, griseofulvin, and rifampicin, with light than in the dark, for both sterile and non-sterile conditions. Biodegradation played a minor role in the disappearance of the antibiotics and was overlaid by photodegradation. In addition, sorption to sediment was of major importance for antibiotics, especially for ciprofloxacin and rifampicin. The t 1/2 of these antibiotics in aqueous phase of “water–sediment” system was higher than for “water-only” experiments, indicating that a part of antibiotics were adsorbed by sediment. The biodegradation did not play a major role on sediment sorption of CIP and RIF, since no statistically significant differences between non-sterile and sterile conditions were observed. Only for GRI, the impact of the biodegradation to the sediment sorption could be found and led to the weak affinity to sediment sorption of this antibiotic. All three antibiotics were more sensitive to photodegradation than to biodegradation; however, the degradation rate was low. In addition, the sorption by sediment occurred also with a slow rate, so these antibiotics could recalcitrant persist in the coastal wetland environment.



Similar content being viewed by others
References
Ali, J., Ali, N., Sultana, Y., Baboota, S., & Faiyaz, S. (2007). Development and validation of stability indicating HPLC method for analysis of antitubercular drugs. Acta Chromatographica, 18, 168–179.
Cardoza, L. A., Knapp, C. W., Larive, C. K., Belden, J. B., Lydy, M., & Graham, D. W. (2005). Factors affecting the fate of ciprofloxacin in aquatic field systems. Water, Air, and Soil Pollution, 161, 383–398.
Cordova-Kreylos, A. L., & Scow, K. M. (2007). Effects of ciprofloxacin on salt marsh sediment microbial communities. ISME Journal, 1, 585–595.
Dekker, T. G. (2005). Active Pharmaceutical Ingredients (APIs). Workshop on GMP and Quality Assurance of Multisource Tuberculosis Medicines. Kuala Lumpur, Malaysia 21–25 February 2005.
Ferdig, M., Kaleta, A., & Buchberger, W. (2005). Improved liquid chromatographic determination of nine currently used fluoroquinolones with fluorescence and mass spectrometric detection for environmental samples. Journal of Separation Science, 28, 1448–1456.
Häggblom, M. M., & Bosset, I. D. (2004). Halogenated organic compounds—A global perspectives. In M. M. Häggblom & I. D. Bosset (Eds.), Dehalogenation (pp. 3–29). New York: Springer.
Kümmerer, K. (2009). Antibiotics in the aquatic environment—a review—Part I. Chemosphere, 75, 417–434.
Li, B., & Zhang, T. (2010). Biodegradation and adsorption of antibiotics in the activated sludge process. Environment Science & Technology, 44(9), 3468–3473.
Li, X., Chen, J., & Nie, X. (2010). Degradation and residue of ciprofloxacin in different simulated water bodies. Acta Scientiarum Naturalium Universitatis Sunyatseni, 49(3), 102–106.
Liu, J., Sun, J., Zhang, W., Gao, K., & He, Z. (2008). HPLC determination of rifampicin and related compounds in pharmaceuticals using monolithic column. Journal of Pharmaceutical and Biomedical Analysis, 46(2), 405–409.
Lunestad, B. T., Samuelsen, O. B., Fjelde, S., & Ervik, A. (1995). Photostability of eight antibacterial agents in seawater. Aquaculture, 134, 217–225.
OECD Test 308 (2002). Aerobic and anaerobic transformation in aquatic sediment systems. In OECD Guidelines for the Testing of Chemicals; Organization for Economic Cooperation and Development: Paris, 2002.
Roybal, J., Walker, C. C., Pfenning, A. P., Turnipseed, S. B., Storey, J. M., Gonzales, S. A., & Hurlbut, J. A. (2002). Concurrent determination of four fluoroquinolones in catfish, shrimp, and salmon by liquid chromatography with fluorescence detection. Journal of AOAC International, 85(6), 1293–1301.
Seifrtová, M., Pena, A., Lino, C. M., & Solich, P. (2008). Determination of fluoroquinolone antibiotics in hospital and municipal wastewaters in Coimbra by liquid chromatography with a monolithic. Analytical and Bioanalytical Chemistry, 391, 799–805.
Thoma, K., & Kubler, N. (1997). New results in the photoinstability of antimycostics. In A. Albini & E. Fasani (Eds.), Drugs, photochemistry and photostability (pp. 129–130). UK: Royal Society of Chemistry.
Thuy, H. T. T., & Loan, T. T. C. (2012). Antibiotic residues from shrimp farming in coastal wetland. Natural Resources and Environment, 2(2012), 61–63 (in Vietnamese).
Thuy, H. T. T., Nga, L. P., & Loan, T. T. C. (2011). Antibiotic contaminants in coastal wetlands from Vietnamese shrimp farming. Environmental Science and Pollution Research, 18, 835–841.
Thuy, H. T. T., Nga, L. P., Loan, T. T. C., Quoc, D. P., & Phuong, T. H. (2012). Fate of fluoroquinolone antibiotics in Vietnamese coastal wetland ecosystem. Wetlands Ecology and Management, 20, 399–408.
Tolls, J. (2001). Sorption of veterinary pharmaceuticals in soils: a review. Environmental Science & Technology, 35(17), 3397–3406.
Tuan, X. L., & Munekage, Y. (2004). Residues of selected antibiotics in water and mud from shrimp ponds in mangrove areas in Vietnam. Marine Pollution Bulletin, 49, 922–929.
Tuan, X. L., Munekage, Y., & Kato, S. (2005). Antibiotic resistance in bacteria from shrimp farming in mangrove areas. Science of the Total Environment, 349, 95–105.
Turiel, E., & Bordin, G. (2005). Determination of quinolones and fluoroquinolones in hospital sewage water by off-line and on-line solid-phase extraction procedures coupled to HPLC-UV. Journal of Separation Science, 28(3), 257–267.
Turiel, E., Martın-Esteban, A., Bordin, G., & Rodrıguez, A. R. (2004). Stability of fluoroquinolone antibiotics in river water samples and in octadecyl silica solid-phase extraction cartridges. Analytical and Bioanalytical Chemistry, 380, 123–128.
Vasconcelos, T. G., Henriques, D. M., König, A., Martins, A. F., & Kümmerer, K. (2009). Photo-degradation of the antimicrobial ciprofloxacin at high pH: identification and biodegradability assessment of the primary by-products. Chemosphere, 76, 487–493.
Wei, B., Liang, D., & Bates, T. R. (2008). Development and validation of a HPLC method to determine griseofulvin in rat plasma: application to pharmacokinetic studies. Analytical Chemistry Insights, 2008(3), 103–109.
Xia, L., Chen, J., Nie, X., Lin, J., & Zhou, X. (2010). Degradation and residue of ciprofloxacin in different simulated water bodies. Acta Scientiarum Naturalium, 49(3), 102–106.
Zhang, C.-Y., Zhou, X.-Z., Chen, J.-F., & Nie, X.-P. (2009). Dynamics and fate of ciprofloxacin in a simulated aquatic system. Journal of Ecology and Rural Environment, 25(3), 73–78.
Zhang, C., Guo, X., Li, B., & Wang, Y. (2012). Biodegradation of ciprofloxacin in soil. Journal of Molecular Liquids, 173, 184–186.
Acknowledgments
This research is funded by Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 105.09-2011.02. The authors thank MSc. Truong Lam Son Hai for laboratory assistance, Prof. Lewis Hinchman and Dr. Paul Truong for English editing and two anonymous reviewers for providing input that greatly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thuy, H.T.T., Loan, T.T.C. Degradation of Selected Pharmaceuticals in Coastal Wetland Water and Sediments. Water Air Soil Pollut 225, 1940 (2014). https://doi.org/10.1007/s11270-014-1940-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-014-1940-y