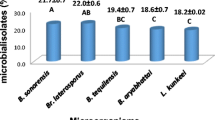
Abstract
Beekeeping is an important agricultural and commercial activity globally practiced. Honey bee is attacked by certain infectious pathogens. Most important brood diseases are bacterial including American Foulbrood (AFB), caused by Paenibacillus larvae (P. larvae), and European Foulbrood (EFB) by Melissococcus plutonius (M. plutonius) in addition of secondary invaders, e.g. Paenibacillus alvei (P. alvei) and Paenibacillus dendritiformis (P. dendritiformis). These bacteria cause the death of larvae in honey bee colonies. In this work, antibacterial activities of extracts, fractions, and isolated certain compounds (nominated 1–3) all originated from moss, Dicranum polysetum Sw. ( D. polysetum), were tested against some honey bee bacterial pathogens. Minimum inhibitory concentration, minimum bactericidal concentration, and sporicidal values of methanol extract, ethyl acetate, and n-hexane fractions ranged between 10.4 and 18.98, 83.4-303.75 & 5.86–18.98 µg/mL against P. larvae, respectively. Antimicrobial activities of the ethyl acetate sub-fractions (fraction) and the isolated compounds (1–3) were tested against AFB- and EFB-causing bacteria. Bio-guided chromatographic separation of ethyl acetate fraction, a crude methanolic extract obtained from aerial parts of D. polysetum resulted in three natural compounds: a novel one, i.e. glycer-2-yl hexadeca-4-yne-7Z,10Z,13Z-trienoate (1, dicrapolysetoate; given as trivial name), in addition to two known triterpenoids poriferasterol (2), and γ-taraxasterol (3). Minimum inhibitory concentration ranges were 1.4-60.75, 8.12-65.0, 2.09–33.44 & 1.8-28.75 µg/mL for sub-fractions, compounds 1, 2, and 3, respectively.

Similar content being viewed by others
References
Aggarwal D, Gautam D, Sharma M, Singla S (2016) Bergenin attenuates renal injury by reversing mitochondrial dysfunction in ethylene glycol induced hyperoxaluric rat model. Eur J Pharmacol 791:611–621. https://doi.org/10.1016/j.ejphar.2016.10.002
Akmaz Ö (2001) The prevalence of american foulbrood disease in Adana region. Pendik Vet Mikrobiyol 32(1–2):55–60
Alippi AM, López AC, Reynaldi FJ, Grasso DH, Aguilar OM (2007) Evidence for plasmid-mediated tetracycline resistance in Paenibacillus larvae, the causal agent of American foulbrood (AFB) disease in honey bees. Vet Microbiol 125(3–4):290–303. https://doi.org/10.1016/j.vetmic.2007.05.018
Alonso-Salces RM, Cugnata NM, Guaspari E, Pellegrini MC, Aubone I, De Piano FG, Antunez K, Fuselli SR (2017) Natural strategies for the control of Paenibacillus larvae, the causative agent of American foulbrood in honey bees: a review. Apidologie 48(3):387–400. https://doi.org/10.1007/s13592-016-0483-1
Alpay Karaoğlu Ş, Yayli N, Erik İ, Korkmaz B, Akpinar R, Bozdeveci A, Suyabatmaz Ş, Batan N, Yeşilyurt A, Kaya S, Nisbet C, Güler A (2022) Biological activity and phytochemical analysis of Dicranum scoparium against the bacterial disease for honey bee. Chem Biodivers 19(7):e202100887. https://doi.org/10.1002/cbdv.202100887
Altuner EM, Canli K, Akata I (2014) Antimicrobial screening of Calliergonella cuspidata, Dicranum polysetum and Hypnum cupressiforme. J Pure Appl Microbiol 8(1):539–545
Arai R, Tominaga K, Wu M, Okura M, Ito K, Okamura N, Onishi H, Osaki M, Sugimura Y, Yoshiyama M, Takamatsu D (2012) Diversity of Melissococcus plutonius from honeybee larvae in Japan and experimental reproduction of european foulbrood with cultured atypical isolates. PLoS ONE 7(3):e33708. https://doi.org/10.1371/journal.pone.0033708
Asakawa Y (2007) Biologically active compounds from bryophytes. Pure Appl Chem 79(4):557–580. https://doi.org/10.1351/pac200779040557
Basile A, Vuotto M, Ielpo M, Moscatiello V, Ricciardi L, Giordano S, Cobianchi RC (1998) Antibacterial activity in Rhynchostegium riparioides (hedw.) card. extract (bryophyta). Phytother Res 12(S1):S146–S148. https://doi.org/10.1002/(SICI)1099-1573(1998)12:1+<S146::AID-PTR278>3.0.CO;2-4
Baş Y, Karaoğlu SA (2015) Characterization and antimicrobial susceptibility of spore-forming bacilli isolated from honeycomb. Recep Tayyip Erdogan University, Graduate School of Natural and Applied Sciences, Department of Biology, Rize, p 123
Beims H, Bunk B, Erler S, Mohr KI, Spröer C, Pradella S, Günther G, Rohde M, von der Ohe W, Steinert M (2020) Discovery of Paenibacillus larvae ERIC V: phenotypic and genomic comparison to genotypes ERIC I-IV reveal different inventories of virulence factors which correlate with epidemiological prevalences of american Foulbrood. Int J Med Microbiol 310(2):151394. https://doi.org/10.1016/j.ijmm.2020.151394
Beuchat LR (1989) Antimicrobials occurring naturally in foods. Food Technol 134–142
Boligon AA, de Brum TF, Zadra M, Piana M, dos Santos Alves CF, Fausto VP, Júnior VdSB, de Almeida Vaucher R, Santos RCV, Athayde ML (2013) Antimicrobial activity of Scutia buxifolia against the honey bee pathogen Paenibacillus larvae. J Invertebr Pathol 112(2):105–107. https://doi.org/10.1016/j.jip.2012.11.009
Borel C, Welti DH, Fernandez I, Colmenares M (1993) Dicranin, an antimicrobial and 15-lipoxygenase inhibitor from the moss Dicranum scoparium. J Nat Prod 56(7):1071–1077. https://doi.org/10.1021/np50097a010
Brødsgaard CJ, Ritter W, Hansen H (1998) Response of in vitro reared honey bee larvae to various doses of Paenibacillus larvae larvae spores. Apidologie 29(6):569–578. https://doi.org/10.1051/apido:19980609
Chaimanee V, Thongtue U, Sornmai N, Songsri S, Pettis JS (2017) Antimicrobial activity of plant extracts against the honeybee pathogens, Paenibacillus larvae and Ascosphaera apis and their topical toxicity to Apis mellifera adults. J Appl Microbiol 123, 1160–1167. https://doi.org/10.1111/jam.13579
Cheng X, Xiao Y, Wang X, Wang P, Li H, Yan H, Liu Q (2012) Anti-tumor and pro-apoptotic activity of ethanolic extract and its various fractions from Polytrichum commune L. ex Hedw in L1210 cells. J Ethnopharmacol 143(1):49–56. https://doi.org/10.1016/j.jep.2012.05.054
CLSI (2015) (Clinical and Laboratory Standards Institute) Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically; Approved Standard-Tenth Edition. CLSI document M07-A10 (ISBN 1-56238-987-4 [Print] (2015)
De Graaf DC, Alippi AM, Antunez K, Aronstein KA, Budge G, De Koker D, De Smet L, Dingman DW, Evans JD, Foster LJ, Fünfhaus A, Garcia-Gonzalez E, Gregore A, Human H, Murray KD, Nguyen BK, Poppinga L, Spivak M, Engelsdorp D, Wilkins S, Genersch E (2013) Standard methods for american foulbrood research. J Apicult Res 52(1):1–26. https://doi.org/10.3896/IBRA.1.52.1.11
Demirci M, Sevim E, Demir İ, Sevim A (2013) Culturable bacterial microbiota of Plagiodera versicolora (L.) (Coleoptera: Chrysomelidae) and virulence of the isolated strains. Folia microbiol 58(3):201–210. https://doi.org/10.1007/s12223-012-0199-1
Dingman DW, Stahly DP (1983) Medium promoting sporulation of Bacillus larvae and metabolism of medium components. Appl Environ Microbiol 46(4):860–869
Djukic M, Becker D, Poehlein A, Voget S, Daniel R (2012) Genome sequence of Paenibacillus alvei DSM 29, a secondary invader during european foulbrood outbreaks. J Bacteriol 194(22):6365. https://doi.org/10.1128/JB.01698-12
Ellis JD, Munn PA (2005) The worldwide health status of honey bees. Bee World 86(4):88–101. https://doi.org/10.1080/0005772X.2005.11417323
Erata H, Batan N (2020) New and remarkable bryophyte records from Turkey and South-West Asia. Plant Biosyst 154(3):376–383. https://doi.org/10.1080/11263504.2019.1635219
Erik İ, Coşkunçelebi K, Makbul S, Yayli N (2021a) New chlorogenic acid derivatives and triterpenoids from Scorzonera aucheriana. Turk J Chem 45:199–209. https://doi.org/10.39 06/kim-2009-17
Erik İ, Yaylı N, Coşkunçelebi K, Makbul S, Karaoğlu ŞA (2021b) Three new dihydroisocoumarin glycosides with antimicrobial activities from Scorzonera aucheriana. Phytochem Lett 43:45–52. https://doi.org/10.1016/j.phytol.2021.02.010
Erler S, Lewkowski O, Poehlein A, Forsgren E (2018) The curious case of Achromobacter eurydice, a Gram-variable pleomorphic bacterium associated with european Foulbrood disease in honeybees. Microb Ecol 75(1):1–6. https://doi.org/10.1007/s00248-017-1007
Evans JD (2003) Diverse origins of tetracycline resistance in the honey bee bacterial pathogen Paenibacillus larvae. J Invertebr Pathol 83(1):46–50. https://doi.org/10.1016/s0022-2011(03)00039-9
Evans JD, Schwarz RS (2011) Bees brought to their knees: microbes affecting honey bee health. Trends Microbiol 19(12):614–620. https://doi.org/10.1016/j.tim.2011.09.003
Forsgren E (2010) European foulbrood in honey bees. J Invertebr Pathol 103(l):5–9. https://doi.org/10.1016/j.jip.2009.06.016
Forsgren E, Budge GE, Charrière JD, Hornitzky MA (2013) Standard methods for european foulbrood research. J Apic Res 52(1):1–14. https://doi.org/10.3896/IBRA.1.52.1.12
Forsgren E, Locke B, Sircoulomb F, Schäfer MO (2018) Bacterial diseases in honeybees. Curr Clin Microbiol Rep 5:18–25. https://doi.org/10.1007/s40588-018-0083-0
Gaggia F, Baffoni L, Stenico V, Alberoni D, Buglione E, Lilli A, Di Gioia D, Porrini C (2015) Microbial investigation on honey bee larvae showing atypical symptoms of european foulbrood. Bull Insectol 68(2):321–327
Gahtori D, Chaturvedi P, Singh S (2011) Using bryophytes as a tool to cure european foulbrood disease of honey bee: an eco-friendly approach. Curr Sci 101(3):420–423
Genersch E (2010) American Foulbrood in honeybees and its causative agent, Paenibacillus larvae. J Invertebr Pathol 103:10–19. https://doi.org/10.1016/j.jip.2009.06.015
Giménez-Martínez P, Cugnata N, Alonso-Salces RM, Arredondo D, Antunez K, De Castro R, Fuselli SR (2019) Natural molecules for the control of Paenibacillus larvae, causal agent of american foulbrood in honey bees (Apis mellifera L). Span J Agric Res 17(3). https://doi.org/10.5424/sjar/2019173-14740
Grabley S, Thiericke R (1999) Bioactive agents from natural sources: trends in discovery and application. Adv Biochem Engin/Biotechnol 64:101–154. https://doi.org/10.1007/3-540-49811-7-4
Hitchcock J, Stoner A, Wilson W, Menapace D (1979) Pathogenicity of Bacillus pulvifaciens to honey bee larvae of various ages (Hymenoptera: Apidae). J Kans Entomol Soc 238–246. https://www.jstor.org/stable/25083900
Hornitzky MA, Wilson S (1989) A system for the diagnosis of the major bacterial brood diseases of honeybees. J Apic Res 28(4):191–195. https://doi.org/10.1080/00218839.1989.11101183
Ichikawa T (1984) New cyclopentenonyl fatty acids from Japanese mosses (Proceedings of the World Conference of Bryology, Tokyo, Japan, May 23–28, 1983-2-) (Chemistry). J Hattori Bot Lab 56:209–213
Kaya Z, Raynal DJ (2001) Biodiversity and conservation of turkish forests. Biol Conserv 97(2):131–141
Klavina L, Springe G, Nikolajeva V, Martsinkevich I, Nakurte I, Dzabijeva D, Steinberga I (2015) Chemical composition analysis, antimicrobial activity and cytotoxicity screening of moss extracts (moss phytochemistry). Molecules 20(9):17221–17243. https://doi.org/10.3390/molecules200917221
Krzaczkowski L, Wright M, Rebérioux D, Massiot G, Etiévant C, Gairin JE (2009) Pharmacological screening of bryophyte extracts that inhibit growth and induce abnormal phenotypes in human HeLa cancer cells. Fundam Clin Pharmacol 23(4):473–482. https://doi.org/10.1111/j.1472-8206.2009.00698.x
Mabona U, Viljoen A, Shikanga E, Marston A, Van Vuuren S (2013) Antimicrobial activity of southern african medicinal plants with dermatological relevance: from an ethnopharmacological screening approach, to combination studies and the isolation of a bioactive compound. J Ethnopharmacol 148(1):45–55. https://doi.org/10.1016/j.jep.2013.03.056
Mărghitaş L, Dezmirean D, Chirilă F, Fiţ N, Bobiş O (2011) Antibacterial activity of different plant extracts and phenolic phytochemicals tested on Paenibacillus larvae bacteria. J Anim Sci Biotechnol 44(2):94–99
Masood F, Thebeau JM, Cloet A, Kozii IV, Zabrodski MW, Biganski S, Liang J, Marta Guarna M, Simko E, Ruzzini A, Wood SC (2022) Evaluating approved and alternative treatments against an oxytetracycline-resistant bacterium responsible for european foulbrood disease in honey bees. Sci Rep 12(1):5906. https://doi.org/10.1038/s41598-022-09796-4
Matin Yekta M, Alavi SHR (2008) New triterpenoids from Peucedanum ruthenicum. Iran J Pharm Sci 4(4):289–294
Mejias E (2020) American Foulbrood and the Risk in the Use of Antibiotics as a Treatment. Modern Beekeeping - Bases for Sustainable Production https://doi.org/10.5772/intechopen.90303
Miyagi T, Peng CY, Chuang RY, Mussen EC, Spivak MS (2000) Verification of oxytetracycline-resistant american foulbrood pathogen Paenibacillus larvae in the United States. J Invertebr Pathol 75(1):95–96. https://doi.org/10.1006/jipa.1999.4888
Narayan GR, Viswas K, Pathak M, Singh PS, Gupta A (2010) Antibacterial activities of ethanolic extracts of plants used in folk medicine. Int J Ayurveda Res (IJRAP) 1(2):529–535
Nikolajeva V, Liepina L, Petrina Z, Krumina G, Grube M, Muiznieks I (2012) Antibacterial activity of extracts from some bryophytes. Adv Microbiol 2(03):345. https://doi.org/10.4236/aim.2012
Nordström S, Fries I (1995) A comparison of media and cultural conditions for identification of Bacillus larvae in honey. J Apic Res 34(2):97–103
Oldroyd B, Goodman R, Hornitzky M, Chandler D (1989) The effect on american foulbrood of standard oxytetracycline hydrochloride treatments for the control of european foulbrood of honeybees (Apis mellifera). Aust J Agric Res 40(3):691–697
Okayama A, Sákogawa T, Nakajima C, Hayama T (1997) Sporicidal activities of disinfectants on Paenibacillus larvae. J Vet Med Sci 59(10):953–954. https://doi.org/10.1292/jvms.59.953
Özdemir T, Batan N (2017) The bryophyte checklist of Trabzon province of Turkey. Arctoa 26(1):58–67. https://doi.org/10.15298/arctoa.26.05
Özkırım A, Keskin N, Kürkçüoğlu M, Başer KHC (2012) Evaluation of some essential oils as alternative antibiotics against american foulbrood agent Paenibacillus larvae on honey bees Apis mellifera L. J Essent Oil Res 24(5):465–470. https://doi.org/10.1080/10412905.2012.703504
Paul VJ, Fenical W (1980) Palisadins A, B and related monocyclofarnesol-derived sesquiterpenoids from the red marine alga Laurencia cf. palisada. Tetrahedron Lett. 21(29):2787–2790. https://doi.org/10.1016/S0040-4039(00)78607-2
Perez C, Pauli M, Bazerque P (1990) An antibiotic assay by agar well diffusion method. Acta Biol Med Ger 15:113–115
Pınarbaş M, Karaoğlu A (2017) Characterization and antibiotic sensitiveness of Gram positive bacteria isolated from american foulbrood suspect bee (Apis mellifera) and bee products, with genetic diversity of Paenibacillus larvae isolates. Recep Tayyip Erdogan University, Graduate School of Natural and Applied Sciences, Department of Biology, p 117
Richards ED, Tell LA, Davis JL, Baynes RE, Lin Z, Maunsell FP, Riviere JE, Jaberi-Douraki M, Martin KL, Davidson G (2021) Honey bee medicine for veterinarians and guidance for avoiding violative chemical residues in honey. J Am Vet Med Assoc 259(8):860–873. https://doi.org/10.2460/javma.259.8.860
Ros RM, Mazimpaka V, Abou-Salama U, Aleffi M, Blockeel TL, Brugués M, Cros RM, Dia MG, Dirkse GM, Draper I (2013) Mosses of the Mediterranean, an annotated checklist. Cryptogamie Bryologie 34(2):99–283. https://doi.org/10.7872/cryb.v34.iss2.2013.99
Sevim E, Bozdeveci A, Pınarbaş Çetin M, Kekeçoğlu M, Akpınar R, Keskin M, Kolaylı S, Alpay Karaoğlu Ş (2021) Antibacterial effects of Anatolian propolis on Paenibacillus larvae. U Bee J 21(2):177–186. https://doi.org/10.31467/uluaricilik.976536
Stephan JG, Lamei S, Pettis JS, Riesbeck K, de Miranda JR, Forsgren E (2019) Honeybee-specific lactic acid bacterium supplements have no effect on American foulbrood-infected honeybee colonies. Appl Environ Microbiol 85(13):e00606-e00619. https://doi.org/10.1128/AEM.00606-19
Smith AJE (1980) The moss flora of Britain and Ireland. Cambridge university press
Thompson HM, Brown MA (2001) Is contact colony treatment with antibiotics an effective control for european foulbrood? Bee World 82(3):130–138. https://doi.org/10.1080/0005772X.2001.11099515
Üçüncü O, Cansu TB, Özdemir T, Karaoğlu A, Yaylı N (2010) Chemical composition and antimicrobial activity of the essential oils of mosses (Tortula muralis Hedw., Homalothecium lutescens (Hedw.) H. Rob., Hypnum cupressiforme Hedw., and Pohlia nutans (Hedw.) Lindb.) from Turkey. Turk J Chem 34(5):825–834. https://doi.org/10.3906/kim-1002-62
Wang XN, Yu WT, Lou HX (2005) Antifungal constituents from the chinese moss Homalia trichomanoides. Chem Biodivers 2(1):139–145. https://doi.org/10.1002/cbdv.200490165
Wiese N, Fischer J, Heidler J, Lewkowski O, Degenhardt J, Erler S (2018) The terpenes of leaves, pollen, and nectar of thyme (Thymus vulgaris) inhibit growth of bee disease-associated microbes. Sci Rep 8(1):14634. https://doi.org/10.1038/s41598-018-32849-6
Yekta SS, Hedenström M, Stehr JE, Dario M, Hertkorn N, Björn A (2018) Pretreatment of anaerobic digester samples by hydrochloric acid for solution-state 1H and 13 C NMR spectroscopic characterization of organic matter. Chemosphere 199:201–209. https://doi.org/10.1016/j.chemosphere.2018.02.015
You Y, Yoo S, Yoon HG, Park J, Lee YH, Kim S, Oh KT, Lee J, Cho HY, Jun W (2010) In vitro and in vivo hepatoprotective effects of the aqueous extract from Taraxacum officinale (dandelion) root against alcohol-induced oxidative stress. Food Chem Toxicol 48(6):1632–1637. https://doi.org/10.1016/j.fct.2010.03.037
Záveská Drábková L, Dobrev PI, Motyka V (2015) Phytohormone profiling across the Bryophytes. PLoS ONE 10(5):e0125411. https://doi.org/10.1371/journal.pone.0125411
Zhang X, Xiong H, Liu L (2012) Effects of taraxasterol on inflammatory responses in lipopolysaccharide-induced RAW 264.7 macrophages. J Ethnopharmacol 141(1):206–211. https://doi.org/10.1016/j.jep.2012.02.020
Funding
Author Ş.A.K. has received research support from Recep Tayyip Erdogan University Research Fund (RTEÜ-FBA-2017-807) and Scientific and Technological Research Council of Turkey (TUBITAK-TOVAG-118O415). Thanks for their financial supports.
Author information
Authors and Affiliations
Contributions
All authors contributed significantly to this article. Conceptualization; ŞAK, NY, RA, AG, Methodology; ŞAK, RA, NY, AB, NB, ŞS, İE, BK, SK, CN, and AG, Draft-manuscript; ŞAK, NY, AB, İE, CN, Funding Acquisition; ŞAK, Writing–Final Version; All authors have reviewed, discussed, edited, and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Ethical approval
Not applicable, because the research was not on humans or animals.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karaoğlu, Ş.A., Yayli, N., Akpinar, R. et al. Phytochemicals, antimicrobial, and sporicidal activities of moss, Dicranum polysetum Sw., against certain honey bee bacterial pathogens. Vet Res Commun 47, 1445–1455 (2023). https://doi.org/10.1007/s11259-023-10094-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10094-1