Abstract
Purpose
Tomato is an important source of lycopene, a carotenoid that has been emerging as a natural preventive agent for prostate disease. Moreover, tomato contains other components with a wide range of physiological properties, but their potential beneficial effects on prostatic hyperplasia (PH) during obesity have not been completely established. In this study, we compared the effect of a lipidic extract of tomato saladette (STE) with Serenoa repens (SR) on obese rats with PH.
Methods
Forty-eight Wistar rats were divided in Control (C) and Obese (Ob) treated without (n = 12) and with (n = 36) testosterone enanthate (TE), once a week for 8 weeks to induce PH. After 4 weeks, SR and STE were administered. Biochemical parameters, oxidative stress markers and inflammatory cytokines production were determined.
Results
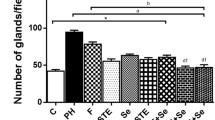
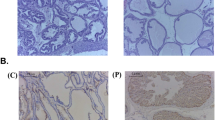
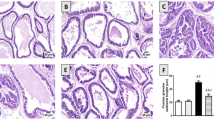
TE increased prostate weight and caused prostatic hyperplasia in C group, and these effects were exacerbated by obesity. SR and STE reverted the increase in prostate weight and hyperplasia caused by TE in C and Ob groups. Obesity increased LDL, TGs, NOx and MAD, but decreased HDLc, GSx, SOD and CAT. SR reverted the effects of obesity, but these were significantly reduced and HDLc increased with STE. Obesity and TE increased TNFα, IL-1β and IL-6 levels, but these were partially reverted by STE compared with SR.
Conclusions
Excess of fat tissue increases the alterations by PH. STE diminishes these alterations compared with SR, suggesting its beneficial effect to improve prostate function. Whole tomato lipid extract could serve as sole therapy or as an adjunct to pharmacological treatment for PH.



Similar content being viewed by others
References
Mraz M, Haluzik M (2014) The role of adipose tissue immune cells in obesity and low-grade inflammation. J Endocrinol 222(3):R113–R127. https://doi.org/10.1530/JOE-14-0283
Foster CS (2000) Pathology of benign prostatic hyperplasia. Prostate Suppl 9:4–14. https://doi.org/10.1002/1097-0045(2000)45:9+%3c4::aid-pros3%3e3.0.co;2-q
García-Flórez M, Oliveira CA, Carvalho HF (2005) Early effects of estrogen on the rat ventral prostate. Braz J Med Biol Res 38(4):487–497. https://doi.org/10.1590/s0100-879x2005000400002
Chughtai B, Forde JC, Thomas DD et al (2016) Benign prostatic hyperplasia. Nat Rev Dis Primers 2:16031. https://doi.org/10.1038/nrdp.2016.31
Langan RC (2019) Benign prostatic hyperplasia. Prim Care 46(2):223–232. https://doi.org/10.1016/j.pop.2019.02.003
Cicero AFG, Allkanjari O, Busetto GM et al (2019) Nutraceutical treatment and prevention of benign prostatic hyperplasia and prostate cancer. Arch Ital Urol Androl. https://doi.org/10.4081/aiua.2019.3.139
van Breemen RB, Pajkovic N (2008) Multitargeted therapy of cancer by lycopene. Cancer Lett 269(2):339–351. https://doi.org/10.1016/j.canlet.2008.05.016
Basu A, Imrhan V (2007) Tomatoes versus lycopene in oxidative stress and carcinogenesis: conclusions from clinical trials. Eur J Clin Nutr 61(3):295–303. https://doi.org/10.1038/sj.ejcn.1602510
Chen J, Song Y, Zhang L (2013) Lycopene/tomato consumption and the risk of prostate cancer: a systematic review and meta-analysis of prospective studies. J Nutr Sci Vitaminol (Tokyo) 59(3):213–223. https://doi.org/10.3177/jnsv.59.213
Clinton SK (2005) Tomatoes or lycopene: a role in prostate carcinogenesis? J Nutr 135(8):2057S-2059S. https://doi.org/10.1093/jn/135.8.2057S
Limpens J, Schröder FH, de Ridder CMA et al (2006) Combined lycopene and vitamin E treatment suppresses the growth of PC-346C human prostate cancer cells in nude mice. J Nutr 136(5):1287–1293. https://doi.org/10.1093/jn/136.5.1287
Chen L, Stacewicz-Sapuntzakis M, Duncan C et al (2001) Oxidative DNA damage in prostate cancer patients consuming tomato sauce-based entrees as a whole-food intervention. JNCI J Natl Cancer Inst 93(24):1872–1879. https://doi.org/10.1093/jnci/93.24.1872
Mohanty NK, Saxena S, Singh UP et al (2005) Lycopene as a chemopreventive agent in the treatment of high-grade prostate intraepithelial neoplasia. Urol Oncol 23(6):383–385. https://doi.org/10.1016/j.urolonc.2005.05.012
Arts IC, Hollman PC (2005) Polyphenols and disease risk in epidemiologic studies. Am J Clin Nutr 81(1 Suppl):317S-325S. https://doi.org/10.1093/ajcn/81.1.317S
Huxley RR, Neil HAW (2003) The relation between dietary flavonol intake and coronary heart disease mortality: a meta-analysis of prospective cohort studies. Eur J Clin Nutr 57(8):904–908. https://doi.org/10.1038/sj.ejcn.1601624
Yang XB, Zhao Y, Yang Y, Ruan Y (2008) Isolation and characterization of immunostimulatory polysaccharide from and herb tea, Gynostemma pentaphyllum Makino. J Agric Food Chem 56:6905–6909. https://doi.org/10.1021/jf801101u
Paubert-Braquet M, Cousse H, Raynaud JP et al (1998) Effect of the lipidosterolic extract of Serenoa repens (Permixon) and its major components on basic fibroblast growth factor-induced proliferation of cultures of human prostate biopsies. Eur Urol 33(3):340–347. https://doi.org/10.1159/000019570
Silvestri I, Cattarino S, Aglianò A et al (2013) Effect of Serenoa repens ( Permixon ®) on the expression of inflammation-related genes : analysis in primary cell cultures of human prostate carcinoma. J Inflamm 10:11. https://doi.org/10.1186/1476-9255-10-11
Perry R, Milligan G, Anderson P et al (2012) Real-world use of Permixon® in benign prostatic hyperplasia–determining appropriate monotherapy and combination treatment. Adv Ther 29(6):538–550. https://doi.org/10.1007/s12325-012-0024-x
Allkanjari O, Vitalone A (2015) What do we know about phytotherapy of benign prostatic hyperplasia? Life Sci 126:42–56. https://doi.org/10.1016/j.lfs.2015.01.023
Levin RM, Das AK (2000) A scientific basis for the therapeutic effects of Pygeum africanum and Serenoa repens. Urol Res 28(3):201–209. https://doi.org/10.1007/s002409900098
Governa P, Giachetti D, Biagi M et al (2016) Hypothesis on Serenoa Repens (Bartram) small extract inhibition of prostatic 5α-reductase x-ray structure. Peer J 4:e2698. https://doi.org/10.7717/peerj.2698
Colado-Velázquez JI, Mailloux-Salinas P, Medina-Contreras J et al (2015) Effect of Serenoa Repens on oxidative stress, inflammatory and growth factors in obese Wistar rats with benign prostatic hyperplasia. Phytother Res 10:1525–1531. https://doi.org/10.1002/ptr.5406
Minutoli L, Bitto A, Squadrito F et al (2013) Serenoa Repens, lycopene and selenium: a triple therapeutic approach to manage benign prostatic hyperplasia. Curr Med Chem 20(10):1306–1312. https://doi.org/10.2174/0929867311320100007
Di Silverio F, Bosman C, Salvatori M et al (2005) Combination therapy with rofecoxib and finasteride in the treatment of men with lower urinary tract symptoms (LUTS) and benign prostatic hyperplasia (BPH). Eur Urol 47(1):72–78. https://doi.org/10.1016/j.eururo.2004.08.024
Carraro JC, Raynaud JP, Koch G et al (1996) Comparison of phytotherapy (Permixon) with finasteride in the treatment of benign prostate hyperplasia: a randomized international study of 1,098 patients. Prostate 29(4):231–240. https://doi.org/10.1002/(SICI)1097-0045(199610)29:4%3c231
Geavlete P, Multescu R, Geavlete B (2011) Serenoa repens extract in the treatment of benign prostatic hyperplasia. Ther Adv Urol 3(4):193–198. https://doi.org/10.1177/1756287211418725
Espinosa-Juárez J, Colado-Velázquez JIII, Mailoux-Salinas P et al (2017) Beneficial effects of lipidic extracts of saladette tomato pomace and Serenoa repens on prostate and bladder health in obese male Wistar rats. J Sci Food Agric 97(13):4451–4458. https://doi.org/10.1002/jsfa.8308
Altavilla D, Bitto A, Polito F et al (2011) The combination of Serenoa Repens, Selenium and Lycopene is more effective than Serenoa repens alone to prevent hormone dependent prostatic growth. J Urol 186:1524–1529. https://doi.org/10.1016/j.juro.2011.05.049
Colado-Velazquez JIII et al. (2021) Extracto de Tomate (Lycopersicum esculentum) para el tratamiento de hiperplasia de próstata en obesidad. (Mexico, Mexico Patent No. 380295). Mexicano de la Protección Industrial. https://www.gob.mx
Fish WW, Perkins-Veazie P, Collins JK (2002) A quantitative assay for lycopene that utilizes reduced volumes of organic solvents. J Food Compos Anal 15(3):309–317. https://doi.org/10.1006/jfca.2002.1069
Miranda KM, Espey MG, Wink DA (2001) A rapid, simple spectrophotometric method for simultaneous detection of nitrate and nitrite. Nitric Oxide 5(1):62–71. https://doi.org/10.1006/niox.2000.0319
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Lee S, Min HG, Choi SH et al (2006) Central obesity as a risk factor for prostatic hyperplasia. Obesity (Silver Spring) 14(1):172–179. https://doi.org/10.1038/oby.2006.21
Kristal AR, Arnold KB, Schenk JM et al (2007) Race/ethnicity, obesity, health related behaviors and the risk of symptomatic benign prostatic hyperplasia: results from the prostate cancer prevention trial. J Urol 177(4):1395–1400. https://doi.org/10.1016/j.juro.2006.11.065
Lokeshwar SD, Harper BT, Webb E et al (2019) Epidemiology and treatment modalities for the management of benign prostatic hyperplasia. Transl Androl Urol 8(5):529–539. https://doi.org/10.21037/tau.2019.10.01
Patel ND, Parsons JK (2014) Epidemiology and etiology of benign prostatic hyperplasia and bladder outlet obstruction. Indian J Urol 30(2):170–176. https://doi.org/10.4103/0970-1591.126900
Lee RK, Chung D, Chughtai B, Te AE, Kaplan SA (2012) Central obesity as measured by waist circumference is predictive of severity of lower urinary tract symptoms. BJU Int 110(4):540–545. https://doi.org/10.1111/j.1464-410X.2011.10819.x
Parsons JK, Carter HB, Partin AW et al (2006) Metabolic factors associated with benign prostatic hyperplasia. J Clin Endocrinol Metab 91(7):2562–2568. https://doi.org/10.1210/jc.2005-2799
Batai K, Phung M, Bell R et al (2021) Correlation between body mass index and prostate volume in benign prostatic hyperplasia patients undergoing holmium enucleation of the prostate surgery. BMC Urol 21(1):88. https://doi.org/10.1186/s12894-020-00753-9
El-Sherbiny M, El-Shafey M, El-Din El-Agawy MS et al (2021) Diacerein ameliorates testosterone-induced benign prostatic hyperplasia in rats: Effect on oxidative stress, inflammation and apoptosis. Int Immunopharmacol 100:108082. https://doi.org/10.1016/j.intimp.2021.108082
Wang L, Hou Y, Wang R et al (2021) Inhibitory effect of astaxanthin on testosterone-induced benign prostatic hyperplasia in rats. Mar Drugs 19(12):652. https://doi.org/10.3390/md19120652
Chen Z, Miao L, Gao X, Wang G, Xu Y (2015) Effect of obesity and hyperglycemia on benign prostatic hyperplasia in elderly patients with newly diagnosed type 2 diabetes. Int J Clin Exp Med 8(7):11289–11294
Shih HJ, Huang CJ, Lin JA et al (2018) Hyperlipidemia is associated with an increased risk of clinical benign prostatic hyperplasia. Prostate 78(2):113–120. https://doi.org/10.1002/pros.23451
Jerde TJ, Bushman W (2009) IL-1 induces IGF-dependent epithelial proliferation in prostate development and reactive hyperplasia. Sci Signal 2(86):ra49. https://doi.org/10.1126/scisignal.2000338
Corona G, Vignozzi L, Rastrelli G et al (2014) Benign prostatic hyperplasia: a new metabolic disease of the aging male and its correlation with sexual dysfunctions. Int J Endocrinol. https://doi.org/10.1155/2014/329456
Nandy PR, Saha S (2016) Association between components of metabolic syndrome and prostatic enlargement: an Indian perspective. Med J Armed Forces India 72(4):350–355. https://doi.org/10.1016/j.mjafi.2016.07.005
Wan X, Yang Z, Ji H et al (2021) Effects of lycopene on abdominal fat deposition, serum lipids levels and hepatic lipid metabolism-related enzymes in broiler chickens. Anim Biosci 34(3):385–392. https://doi.org/10.5713/ajas.20.0432
Hosseini-Vashan SJ, Golian A, Yaghobfar A (2016) Growth, immune, antioxidant, and bone responses of heat stress-exposed broilers fed diets supplemented with tomato pomace. Int J Biometeorol 60:1183–1192. https://doi.org/10.1007/s00484-015-1112-9
Silva SA, Gobbo MG, Pinto-Fochi ME et al (2015) Prostate hyperplasia caused by long-term obesity is characterized by high deposition of extracellular matrix and increased content of MMP-9 and VEGF. Int J Exp Pathol 96(1):21–30. https://doi.org/10.1111/iep.12107
Timms BG, Hofkamp LE (2011) Prostate development and growth in benign prostatic hyperplasia. Differentiation 82(4–5):173–183. https://doi.org/10.1016/j.diff.2011.08.002
Ribeiro DL, Pinto ME, Maeda SY et al (2012) High fat-induced obesity associated with insulin-resistance increases FGF-2 content and causes stromal hyperplasia in rat ventral prostate. Cell Tissue Res 349(2):577–588. https://doi.org/10.1007/s00441-012-1420-x
Cohen PG (2009) Benign prostatic hyperplasia: the hypogonadal-obesity-prostate connection. Med Hypotheses 73(2):142–143. https://doi.org/10.1016/j.mehy.2009.03.013
Ribeiro DL, Pinto ME, Rafacho A et al (2012) High-fat diet obesity associated with insulin resistance increases cell proliferation, estrogen receptor, and PI3K proteins in rat ventral prostate. J Androl 33(5):854–865. https://doi.org/10.2164/jandrol.111.016089
Koerner A, Kratzsch J, Kiess W (2005) Adipocytokines: leptin–the classical, resistin–the controversical, adiponectin–the promising, and more to come. Best Pract Res Clin Endocrinol Metab 19(4):525–546. https://doi.org/10.1016/j.beem.2005.07.008
Leze E, Alves-Pereira JL, Colli S et al (2012) Leptin regulates proliferation and apoptosis in human prostate. Sci World J 2012:842301. https://doi.org/10.1100/2012/842301
Vikram A, Kushwaha S, Jena GB (2011) Relative influence of testosterone and insulin in the regulation of prostatic cell proliferation and growth. Steroids 76(4):416–423. https://doi.org/10.1016/j.steroids.2010.12.014
Takeda M, Tang R, Shapiro E et al (1995) Effects of nitric oxide on human and canine prostates. Urology 45(3):440–446. https://doi.org/10.1016/S0090-4295(99)80013-2
Roumeguère T, Sfeir J, El Rassy E et al (2017) Oxidative stress and prostatic diseases. Mol Clin Oncol 7(5):723–728. https://doi.org/10.3892/mco.2017.1413
Shoieb SM, Esmat A, Khalifa AE, Abdel-Naim AB (2018) Chrysin attenuates testosterone-induced benign prostate hyperplasia in rats. Food Chem Toxicol 111:650–659. https://doi.org/10.1016/j.fct.2017.12.017
Rao AV, Agarwal S (2000) Role of antioxidant lycopene in cancer and heart disease. J Am Coll Nutr 19(5):563–569. https://doi.org/10.1080/07315724.2000.10718953
Story EN, Kopec RE, Schwartz SJ et al (2010) An update on the health effects of tomato lycopene. Annu Rev Food Sci Technol 1:189–210. https://doi.org/10.1146/annurev.food.102308.124120
Wang H, Leung LK (2010) The carotenoid lycopene differentially regulates phase I and II enzymes in dimethylbenz[a]anthracene-induced MCF-7 cells. Nutrition 26(11–12):1181–1187. https://doi.org/10.1016/j.nut.2009.11.013
Cormio L, Calò B, Falagario U, Iezzi M, Lamolinara A, Vitaglione P, Silecchia G, Carrieri G, Fogliano V, Iacobelli S, Natali PG, Piantelli M (2021) Improvement of urinary tract symptoms and quality of life in benign prostate hyperplasia patients associated with consumption of a newly developed whole tomato-based food supplement: a phase II prospective, randomized double-blinded, placebo-controlled study. J Transl Med 19(1):24. https://doi.org/10.1186/s12967-020-02684-3.8
Rho J, Seo CS, Park HS et al (2020) Asteris Radix et Rhizoma suppresses testosterone-induced benign prostatic hyperplasia in rats by regulating apoptosis and inflammation. J Ethnopharmacol 12(255):112779. https://doi.org/10.1016/j.jep.2020.112779
Abdel-Naim AB, Neamatallah T, Eid BG, Esmath A et al (2018) 2-methoxyestradiol attenuates testosterone-induced benign prostate hyperplasia in rats through inhibition of HIF-1α/TGF-β/Smad2 axis. Oxid Med Cell Longev. https://doi.org/10.1155/2018/4389484
Csikós E, Horváth A, Ács K et al (2021) Treatment of benign prostatic hyperplasia by natural drugs. Molecules 26(23):7141. https://doi.org/10.3390/molecules26237141
Riso P, Visioli F, Grande S et al (2006) Effect of a tomato-based drink on markers of inflammation, immunomodulation, and oxidative stress. J Agric Food Chem 54(7):2563–2566. https://doi.org/10.1021/jf053033c
Zhao Q, Yang F, Meng L et al (2020) Lycopene attenuates chronic prostatitis/chronic pelvic pain syndrome by inhibiting oxidative stress and inflammation via the interaction of NF-κB, MAPKs, and Nrf2 signaling pathways in rats. Andrology 3:747–755. https://doi.org/10.1111/andr.12747.ers
Acknowledgements
The research was carried out with partial support by CONACYT postgraduate fellowship #96606.
Author information
Authors and Affiliations
Contributions
G.B., JIIIC.V. and P.M.S contributed to the study design and reviewed the manuscript. P.M.S, J.L.A., D.J.A.CH. and G.N.S. contributed to experimental work and analyzed the data. E.C.E., D.J.A.CH. and N.L.G.V. contributed to discussion and approved the version to be published.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Colado-Velázquez, J., Mailloux-Salinas, P., Arias-Chávez, D.J. et al. Lipidic extract of whole tomato reduces hyperplasia, oxidative stress and inflammation on testosterone-induced BPH in obese rats. Int Urol Nephrol 55, 529–539 (2023). https://doi.org/10.1007/s11255-022-03383-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-022-03383-2