Abstract
Purpose
The goal of this study was to externally validate the recently proposed prognostic model for the prediction of estimated glomerular filtration rate (eGFR) < 60 ml/min/1.73 m2 1 year after living donor nephrectomy.
Methods
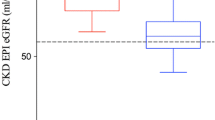
130 living kidney donors (median age at donation 52.3 years, range 24.7–75.6 years) were investigated before and after donation between March 2000 and April 2016. Preoperative eGFR values varied between 61.7 and 148.4 ml/min (mean: 89, median: 88). Observed eGFR 1 year after transplantation (±45 days) ranged between 36.3 and 97.1 ml/min (mean: 55, median: 53). 70.8% of donors displayed eGFR values < 60 ml/min 1 year after donation. Predicted eGFR 1 year after donation was determined using the prognostic model proposed by Benoit et al. (Int Urol Nephrol 49(5):793–801. doi:10.1007/s11255-017-1559-1, 2017): postoperative eGFR ml/min/1.73 m2 = 31.71 + (0.521 × eGFR in ml/min prior to donation −0.314 × Age in years at donation). Pearson correlation and receiver operating characteristics curve (ROC-curve) were used to assess external validity of the proposed prognostic model to predict postoperative eGFR in ml/min and eGFR < 60 ml/min.
Results
The correlation between predicted and observed eGFR 1 year after donation was significant (p < 0.001; R 2 = 0.594). The area under the ROC-curve (AUROC) demonstrated a high sensitivity and specificity for predicted eGFR values < 60 ml/min (AUROC = 0.866).
Conclusions
The proposed prognostic model for the prediction of postoperative eGFR was successfully validated in our cohort. We therefore consider the model as generally applicable.


Similar content being viewed by others
References
Cecka JM (1999) The UNOS scientific renal transplant registry. Clin Transpl 1–21
Opelz G, Wujciak T, Dohler B, Scherer S, Mytilineos J (1999) HLA compatibility and organ transplant survival. Collaborative transplant study. Rev Immunogenet 1(3):334–342
Park YH, Min SK, Lee JN, Lee HH, Jung WK, Lee JS, Lee JH, Lee YD (2004) Comparison of survival probabilities for living-unrelated versus cadaveric renal transplant recipients. Transplant Proc 36(7):2020–2022. doi:10.1016/j.transproceed.2004.08.122
Ozkurt S, Karavelioglu Y, Kalcik M, Musmul A (2017) Assessment of long-term cardiovascular effects of unilateral nephrectomy. Int Urol Nephrol 49(5):867–873. doi:10.1007/s11255-017-1553-7
Ibrahim HN, Foley R, Tan L, Rogers T, Bailey RF, Guo H, Gross CR, Matas AJ (2009) Long-term consequences of kidney donation. N Engl J Med 360(5):459–469. doi:10.1056/NEJMoa0804883
Lentine KL, Patel A (2012) Risks and outcomes of living donation. Adv Chronic Kidney Dis 19(4):220–228. doi:10.1053/j.ackd.2011.09.005
Reese PP, Boudville N, Garg AX (2015) Living kidney donation: outcomes, ethics, and uncertainty. Lancet 385(9981):2003–2013. doi:10.1016/S0140-6736(14)62484-3
Benoit T, Game X, Roumiguie M, Sallusto F, Doumerc N, Beauval JB, Rischmann P, Kamar N, Soulie M, Malavaud B (2017) Predictive model of 1-year postoperative renal function after living donor nephrectomy. Int Urol Nephrol 49(5):793–801. doi:10.1007/s11255-017-1559-1
Collins GS, Reitsma JB, Altman DG, Moons KG (2015) Transparent reporting of a multivariable prediction model for individual prognosis or diagnosis (TRIPOD): the TRIPOD statement. Ann Intern Med 162(1):55–63. doi:10.7326/M14-0697
Levey AS, Coresh J, Greene T, Marsh J, Stevens LA, Kusek JW, Van Lente F, Chronic Kidney Disease Epidemiology C (2007) Expressing the modification of diet in renal disease study equation for estimating glomerular filtration rate with standardized serum creatinine values. Clin Chem 53(4):766–772. doi:10.1373/clinchem.2006.077180
Hanley JA, McNeil BJ (1982) The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 143(1):29–36. doi:10.1148/radiology.143.1.7063747
Dols LF, Kok NF, Ijzermans JN (2010) Live donor nephrectomy: a review of evidence for surgical techniques. Transpl Int 23(2):121–130. doi:10.1111/j.1432-2277.2009.01027.x
Lewis GR, Brook NR, Waller JR, Bains JC, Veitch PS, Nicholson ML (2004) A comparison of traditional open, minimal-incision donor nephrectomy and laparoscopic donor nephrectomy. Transpl Int 17(10):589–595. doi:10.1007/s00147-004-0770-z
Saito T, Uchida K, Ishida H, Tanabe K, Nitta K (2015) Changes in glomerular filtration rate after donation in living kidney donors: a single-center cohort study. Int Urol Nephrol 47(2):397–403. doi:10.1007/s11255-014-0861-4
Funding
This work was supported by a grant from the German Federal Ministry of Education and Research (reference number: 01EO1302).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All authors of this manuscript declare that they have no conflicts of interest to disclose.
Human and animal rights
This article does not contain any studies with animals performed by any of the authors.
Informed consent
Regarding the patients’ clinical data, as an observational retrospective study, according to the Professional Code of the German Medical Association (article B.III. § 15.1), neither informed consent nor approval of the ethics committee was needed for this study.
Rights and permissions
About this article
Cite this article
Kulik, U., Gwiasda, J., Oldhafer, F. et al. External validation of a proposed prognostic model for the prediction of 1-year postoperative eGFR after living donor nephrectomy. Int Urol Nephrol 49, 1937–1940 (2017). https://doi.org/10.1007/s11255-017-1683-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-017-1683-y