Abstract
Objectives
To investigate the long-term effects of Floseal® on erectile function recovery (EFR) after nerve-sparing robot-assisted radical prostatectomy (RALP).
Methods
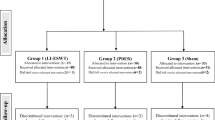
We prospectively collected results of the self-administered International Index Erectile Function Questionnaire 1–5 and 15 (IIEF 1–5 and 15) of 532 consecutive patients who underwent RALP for prostate cancer in our institution between October 2007 and December 2015. Patients were divided into two groups according to Floseal® application after prostatectomy. They were enrolled according to the following criteria: (a) bilateral nerve-sparing procedure; (b) preoperative IIEF ≥ 17; adherence to our erectile rehabilitation protocol; (c) 1-year follow-up. Outcomes were measured as mean IIEF score, EFR (IIEF < 17 or ≥17), grade of ED: severe (IIEF < 17), moderate (17–21), mild (22–25) and no ED (>25).
Results
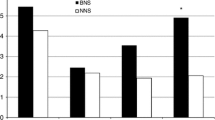
A total of 120 patients were enrolled. Group A included 40 consecutive patients who received traditional hemostasis, and Group B included 80 consecutive patients in which Floseal® was additionally used. No differences were observed in terms of preoperative mean IIEF score (p = 0.65). Group B patients showed a trend toward a higher mean IIEF score 3 months after surgery (p = 0.06) but no differences in terms of EFR (p = 1.000). Long-term results (6, 9, 12 months after surgery) showed a significantly and progressively higher mean IIEF score (p = 0.04, 0.003, 0.003) and EFR (p = 0.043, 0.027, 0.004) in Group A patients. Comparison between the groups in terms of severe, moderate, mild and no ED becomes significant at 9 and 12 months (p = 0.002, 0.006).
Conclusion
The results of our study suggest that local use of Floseal® worsens the long-term erectile function recovery in patients selected for nerve-sparing RALP.


Similar content being viewed by others
Abbreviations
- ED:
-
Erectile dysfunction
- EFR:
-
Erectile function recovery
- NVB:
-
Neurovascular bundles
- RALP:
-
Robot-assisted radical prostatectomy
- IIEF:
-
International Index Erectile Function Questionnaire
References
Tiroli M, Conti A, Muzzonigro G (2012) The use of haemostatic agents in urology. Urologia 79(suppl 19):99–106
Achneck HE, Sileshi B, Jamiolkowski RM et al (2010) A comprehensive review of topical hemostatic agents: efficacy and recommendations for use. Ann Surg 251:217–228
Sileshi B, Achneck HE, Lawson JH (2008) Management of surgical hemostasis: topical agents. Vascular 16(suppl 1):S22–S28
David G, Lim S, Gunnarsson C et al (2015) Similar patient outcomes yet different hospital costs between flowable hemostatic agents. J Med Econ 18:735–745
Altun I (2016) An experimental study on histopathological effects of hemostatic agents used in spinal surgery. World Neurosurg 90:147–153
Rigopoulos C, Kyriazis I, Kallidonis P et al (2013) Assessing the use of haemostatic sealants in tubeless percutaneous renal access and their effect on renal drainage and histology: an experimental porcine study. BJU Int 112(2):E11421
Cağlar M, Yavuzcan A, Yıldız E et al (2014) Increased adhesion formation after gelatin-thrombin matrix application in a rat model. Arch Gynecol Obstet 290(3):501–506
Suzuki Y, Vellinga TT, Istre O et al (2010) Small bowel obstruction associated with use of a gelatin-thrombin matrix sealant (FloSeal) after laparoscopic gynecologic surgery. J Minim Invasive Gynecol 17(5):641–645
Thomas PJ, Tawfic SN (2009) Eosinophil-rich inflammatory response to FloSeal hemostatic matrix presenting as postoperative pelvic pain. Am J Obstet Gynecol 200(4):e10–e11
Hoffmann NE, Siddiqui SA, Agarwal S et al (2009) Choice of hemostatic agent influences adhesion formation in a rat cecal adhesion model. J Surg Res 155(1):77–81
Martorana E, Ghaith A, Micali S et al (2016) A retrospective analysis of the hemostatic effect of FloSeal in patients undergoing robot-assisted laparoscopic radical prostatectomy. Urol Int 96(3):274–279
Abboudi H, Khan MS, Guru KA et al (2014) Learning curves for urological procedures: a systematic review. BJU Int 114(4):617–629
Costello AJ, Brooks M, Cole OJ (2004) Anatomical studies of the neurovascular bundle and cavernosal nerves. BJU Int 94(7):1071–1076
Reeves F, Preece P, Kapoor J et al (2015) Preservation of the neurovascular bundles is associated with improved time to continence after radical prostatectomy but not long-term continence rates: results of a systematic review and meta-analysis. Eur Urol 68(4):692–704
Seddon HJ (1942) A classification of nerve injuries. Br Med J 2(4260):237–239
Sunderland S (1981) The anatomic foundation of peripheral nerve repair techniques. Orthop Clin North Am 12(2):245–266
Lee SK, Wolfe SW (2000) Peripheral nerve injury and repair. J Am Acad Orthop Surg 8(4):243–252
Ong AM, Su LM, Varkarakis I et al (2004) Nerve sparing radical prostatectomy: effects of hemostatic energy sources on the recovery of cavernous nerve function in a canine model. J Urol 172(4 Pt 1):1318–1322
Zeltser I, Dugi D, Gupta A et al (2008) Does topical haemostatic agent have an adverse effect on the function of the prostatic neurovascular bundle? BJU Int 102(8):1005–1007
Ahlering TE, Eichel L, Skarecky D (2005) Rapid communication: early potency outcomes with cautery-free neurovascular bundle preservation with robotic laparoscopic radical prostatectomy. J Endourol 19:715–718
Ahlering TE, Eichel L, Chou D et al (2005) Feasibility study for robotic radical prostatectomy cautery-free neurovascular bundle preservation. Urology 65(994–7):4
Oz MC, Rondinone JF, Shargill NS (2003) FloSeal Matrix: new generation topical hemostatic sealant. J Card Surg 18:486–493
Arand AG, Sawaya R (1986) Intraoperative chemical hemostasis in neurosurgery. Neurosurgery 18(2):223–233
Nishino A, Suzuki M, Ohtani H et al (1993) Thrombin may contribute to the pathophysiology of central nervous system injury. J Neurotrauma 10:167–179
Mandel P, Preisser F, Graefen M et al (2016) High Chance of late recovery of urinary and erectile function beyond 12 months after radical prostatectomy. Eur Urol 71(6):848–850
Akassoglou K, Akpinar P, Murray S et al (2003) Fibrin is a regulator of Schwann cell migration after sciatic nerve injury in mice. Neurosci Lett 338(3):185–188
Lee P, Spector JG, Derby A et al (1998) Effects of thrombin and protease nexin-1 on peripheral nerve regeneration. Ann Otol Rhinol Laryngol. 107(1):61–69
Lee DY, Park KW, Jin BK (2006) Thrombin induces neurodegeneration and microglial activation in the cortex in vivo and in vitro: proteolytic and non-proteolytic actions. Biochem Biophys Res Commun 346(3):727–738
Turgeon VL, Milligan CE, Houenou LJ (1999) Activation of the protease-activated thrombin receptor (PAR)-1 induces motoneuron degeneration in the developing avian embryo. J Neuropathol Exp Neurol 58(5):499–504
Fujimoto S, Katsuki H, Kume T et al (2006) Thrombin-induced delayed injury involves multiple and distinct signaling pathways in the cerebral cortex and the striatum in organotypic slice cultures. Neurobiol Dis 22(1):130–142
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they do not have any kind of conflict of interest and that they have no affiliations with or involvement in any organization or entity with any financial interest.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments. All patients signed a specific informed consent before their inclusion into the study.
Rights and permissions
About this article
Cite this article
Martorana, E., Rocco, B., Kaleci, S. et al. Does topical hemostatic agent (Floseal®) have a long-term adverse effect on erectile function recovery after nerve-sparing robot-assisted radical prostatectomy?. Int Urol Nephrol 49, 1519–1526 (2017). https://doi.org/10.1007/s11255-017-1645-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-017-1645-4