Abstract
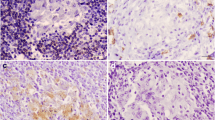
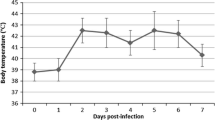
In this study, in order to reveal the immune response against the disease in naturally infected sheep with SPPV, the expressions of various pro- or anti-inflammatory cytokines such as tumour necrosis factor alpha (TNF-α), interferon gamma (IFN-γ), interleukin-1beta (IL-1β), interleukin-2 (IL-2), interleukin-6 (IL-6), interleukin-8 (IL-8), interleukin-10 (IL-10) and interleukin-12 (IL-12) were evaluated immunohistochemically. The material of this study consisted of tissue samples taken from 24 sheep, which were brought as dead for routine histopathological examination to the Department of Pathology. Avidin–biotin–peroxidase method was used for immunohistochemistry. Characteristic pox lesions were observed in the skin, lungs and kidneys. In histopathological examinations, pox cells, which are very characteristic for the diagnosis of the disease, were observed in all three tissues. Capripoxvirus nucleic acid was detected in 8 of the 24 tissues. Samples were sequenced, and a phylogenetic tree was constructed with reference strains from GenBank. Strains from the study clustered with sheeppox virus references. In conclusion, the levels of pro-inflammatory cytokines such as TNF-α, IFN-γ, IL-1β, IL-2, IL-8 and IL12 (Th1) were much more dominant compared to the levels of anti-inflammatory cytokines: IL-10 and IL-6 (Th2). This supported the fact that the cellular immune response is much more effective than the humoral immune response in sheeppox.












Similar content being viewed by others
Data availability
Data are available within the article or its supplementary materials.
References
Abbas, A. K., Lichtman, A. H. and Pillai, S., 2014. Basic Immunology Functions and Disorders of the Immune System, Fourth Edition, (Elsevier Saunders, Philadelphia).
Abu-El-Saad, A. A. and Abdel-Moneim, A. S., 2005. Modulation of macrophage functions by sheeppox virus provides clues to understand interaction of the virus with host immune system. Virology journal, 2, 22.
Alcamí, A., Symons, J. A., Khanna, A. and Smith, G. L., 1998. Poxviruses: Capturing cytokines and chemokines. Seminars in Virology, 5, 419-427.
Alcamí, A. and Smith, G. L., 1996. Soluble interferon-gamma receptors encoded by poxviruses. Comparative immunology, microbiology and infectious diseases, 19(4), 305–317.
Aldaiarov, N., Stahel, A., Nufer, L., Jumakanova, Z., Tulobaev, A. and Ruetten, M., 2016. Outbreak of sheeppox in farmed sheep in Kyrgystan: Histological, eletron microscopical and molecular characterization. Ausbruch von Schafpocken in Kirgistan: eine detaillierte histologische, elektronenmikroskopische und molekulare Charakterisierung. Schweizer Archiv fur Tierheilkunde, 158(7), 529–532.
Arda, M., Minbay, A., Aydin, N., Akay, O. and Diker, S., 1998. Immunoloji, İkinci Baski, (Medisan, Ankara).
Ayari-Fakhfakh, E., Ghram, A., Albina, E. and Cêtre-Sossah, C., 2018. Expression of cytokines following vaccination of goats with a recombinant capripoxvirus vaccine expressing Rift Valley fever virus proteins. Veterinary immunology and immunopathology, 197, 15–20.
Badr, Y., Noreldin, A. E., Elewa, Y. H. A., Ahmed, M. S., Inoshima, Y., Baker, N. M., Aamer, W. N., Abas, O. M., Nayel, M., Rahman, M. M., Elgendy, E., Saleh, A. G. and El-Neweshy, M. S., 2022. Cellular infiltration, cytokines, and histopathology of skin lesions associated with different clinical forms and stages of naturally occurring lumpy skin disease in cattle. Comparative immunology, microbiology and infectious diseases, 90-91, 101894.
Ben Chehida, F., Ayari-Fakhfakh, E., Caufour, P., Amdouni, J., Nasr, J., Messaoudi, L., Haj Ammar, H., Sghaier, S., Bernard, C., Ghram, A. and Cêtre-Sossah, C., 2018. Sheep pox in Tunisia: Current status and perspectives. Transboundary and emerging diseases, 65(1), 50–63.
Beytut, E., 2010. Sheep pox virus induces proliferation of type II pneumocytes in the lungs. Journal of comparative pathology, 143(2-3), 132–141.
Beytut, E., 2017. Pathological and immunohistochemical evaluation of skin and teat papillomas in cattle. Turkish journal of veterinary & animal sciences, 41(2), 204-212.
Boshra, H., Truong, T., Nfon, C., Bowden, T. R., Gerdts, V., Tikoo, S., Babiuk, L. A., Kara, P., Mather, A., Wallace, D. B. and Babiuk, S., 2015. A lumpy skin disease virus deficient of an IL-10 gene homologue provides protective immunity against virulent capripoxvirus challenge in sheep and goats. Antiviral research, 123, 39–49.
Campanella, J. J., Bitincka, L., and Smalley, J., 2003. MatGAT: an application that generates similarity/identity matrices using protein or DNA sequences. BMC bioinformatics, 4, 29.
Chahota, R., Sharma, P., Kumar, R., Gupta, T. and Sharma, M., 2022. Investigation of an outbreak of sheeppox among native sheep breeds in the Western Himalayas of India. Veterinary research communications, 46(1), 101–107.
Chibssa, T. R., Kangethe, R. T., Berguido, F. J., Settypalli, T. B. K., Liu, Y., Grabherr, R., Loitsch, A., Sassu, E. L., Pichler, R., Cattoli, G., Diallo, A., Wijewardana, V. and Lamien, C. E., 2021. Innate immune responses to wildtype and attenuated sheeppox virus mediated through RIG-1 sensing in PBMC in-vitro. Frontiers in immunology, 12, 666543.
Diker, K. S., 2005. Immunoloji. İkinci Baski, (Medisan, Ankara).
Embury-Hyatt, C., Babiuk, S., Manning, L., Ganske, S., Bowden, T. R., Boyle, D. B. and Copps, J., 2012. Pathology and viral antigen distribution following experimental infection of sheep and goats with capripoxvirus. Journal of comparative pathology, 146(2-3), 106–115.
Gulbahar, M. Y., Davis, W. C., Yuksel, H. and Cabalar, M., 2006. Immunohistochemical evaluation of inflammatory infiltrate in the skin and lung of lambs naturally infected with sheeppox virus. Veterinary pathology, 43(1), 67–75.
Hall, T.A., 1999. BioEdit: A user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic acids symp ser, 41:95–98
Kara, P. D., Mather, A. S., Pretorius, A., Chetty, T., Babiuk, S. and Wallace, D. B., 2018. Characterisation of putative immunomodulatory gene knockouts of lumpy skin disease virus in cattle towards an improved vaccine. Vaccine, 36(31), 4708–4715.
Madhavan, A., Venkatesan, G., Kumar, A., Arya, S. and Pandey, A. B., 2020. Comparative sequence and structural analysis of the ORF095 gene, a vaccinia virus A4L homolog of capripoxvirus in sheep and goats. Archives of Virology, 165(6), 1419-1431.
Maxie, M. G., 2016. Jubb, Kennedy, and Palmer’s Pathology of Domestic Animals, Sixth Edition, (Elsevier, St. Louis, Missouri).
Oğuzoğlu, T. C., Alkan, F., Ozkul, A., Vural, S. A., Güngör, A. B. and Burgu, I., 2006. A sheeppox virus outbreak in Central Turkey in 2003: isolation and identification of capripoxvirus ovis. Veterinary research communications, 30(8), 965–971.
Oreiby, A., Seada, A. S., Abou Elazab, M. F., Abdo, W., Kassab, M., Hegazy, Y., Khalifa, H. O. and Matsumoto, T., 2022. Emergency vaccination as a control strategy against sheeppox outbreak in a highly susceptible population. Animals : an open access journal from MDPI, 12(16), 2084.
Ozmen, O., Kale, M., Haligur, M. and Yavru, S., 2009. Pathological, serological, and virological findings in sheep infected simultaneously with Bluetongue, Peste-des-petits-ruminants, and Sheeppox viruses. Tropical animal health and production, 41(6), 951–958.
Pikor, L. A., Enfield, K. S., Cameron, H. and Lam, W. L., 2011. DNA extraction from paraffin embedded material for genetic and epigenetic analyses. JoVE (Journal of Visualized Experiments), (49), e2763.
Rasool, T. J., Hosamani, M., Premraj, A., Sreekumar, E. and Singh, R. K., 2006. Ovine interferon-γ gene of indigenous sheep: Cloning, sequencing and expression studies in Escherichia coli. Indian journal of biotechnology, 5, 486-490.
Roy, P., Purushothaman, V., Sreekumar, C., Tamizharasan, S. and Chandramohan, A., 2008. Sheep pox disease outbreaks in Madras Red and Mechery breeds of indigenous sheep in Tamilnadu, India, Research in veterinary science, 85(3), 617–621.
Sareyyüpoğlu, B., Gülyaz, G., Saraç, F., Uzar, S., Kabaklı, Ö., Çokçalışkan, C., Gündüzalp, C., Uzun, E. A. and Camli, O., 2022. Antibody response of sheep to simultaneous vaccination of foot-and-mouth disease, peste des petits ruminants, sheep pox and goat pox, and bluetongue vaccines. Tropical biomedicine, 39(1), 47–54.
Tamura, K., Peterson, D., Peterson, N., Stecher, G., Nei, M. and Kumar, S., 2011. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molecular biology and evolution, 28, 2731–2739.
Varshovi, H. R., Norian, R., Azadmehr, A. and Ahangaran, N. A., 2017. Immune response characteristics of capri pox virus vaccines following emergency vaccination of cattle against lumpy skin disease virus. Iranian journal of veterinary science and technology, 9(2), 33-40.
WOAH., 2023. Manual of diagnostic tests and vaccines for terrestrial animals, World organisation of animal health. https://www.woah.org/fileadmin/Home/eng/Health_standards/tahm/A_summry.htm. Accessed 18 July 2023
Zachary, J. F., 2017. Pathologic Basis of Veterinary Disease, Sixth Edition, (Elsevier, St. Louis, Missouri).
Zeng, X., Wang, S., Chi, X., Chen, S. L., Huang, S., Lin, Q., Xie, B. and Chen, J. L., 2016. Infection of goats with goatpox virus triggers host antiviral defense through activation of innate immune signaling. Research in veterinary science, 104, 40–49.
Zewdie, G., Derese, G., Getachew, B., Belay, H. and Akalu, M., 2021. Review of sheep and goat pox disease: current updates on epidemiology, diagnosis, prevention and control measures in Ethiopia. Animal diseases, 1(1), 28.
Zhang, D., Yang, B., Zhang, T., Shi, X., Shen, C., Zheng, H., Liu, X. and Zhang, K., 2021. In vitro and in vivo analyses of co-infections with peste des petits ruminants and capripox vaccine strains. Virology journal, 18(1), 69.
Zhu, X. L., Yang, F., Li, H. X., Dou, Y. X., Meng, X. L., Li, H., Luo, X. N. and Cai, X. P., 2013. Identification and phylogenetic analysis of a sheep pox virus isolated from the Ningxia Hui Autonomous Region of China. Genetics and molecular research : GMR, 12(2), 1670–1678.
Zro, K., Zakham, F., Melloul, M., El Fahime, E. and Ennaji, M. M., 2014. A sheeppox outbreak in Morocco: isolation and identification of virus responsible for the new clinical form of disease. BMC veterinary research, 10, 31.
Author information
Authors and Affiliations
Contributions
E.K.: idea, concept and writing. N.C.: molecular studies and interpretation of data. E.B.: immunohistochemical and histopathological analysis. S.D.: immunohistochemical and histopathological analysis. V.Y.: molecular studies and interpretation of data. H.N.: immunohistochemical and histopathological staining. A.Y.: immunohistochemical and histopathological staining. E.K.: immunohistochemical and histopathological staining.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by Kafkas University Animal Experiments Local Ethics Committee (decision number: KAU-HADYEK-2021/065) and the General Directorate of Food and Control (E-71037622-325.01-7907977) of the Republic of Türkiye Ministry of Agriculture and Forestry.
Consent to participate
All participants gave their consent before participating.
Consent for publication
All authors revised and approved the final manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karakurt, E., Coskun, N., Beytut, E. et al. Cytokine profile in lambs naturally infected with sheeppox virus. Trop Anim Health Prod 55, 401 (2023). https://doi.org/10.1007/s11250-023-03823-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11250-023-03823-w