Abstract
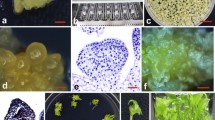
Efficient Agrobacterium tumefaciens-mediated transformation was achieved using embryogenic suspension cultures of sweetpotato (Ipomoea batatas (L.) Lam.) cv. Lizixiang. Cell aggregates from embryogenic suspension cultures were cocultivated with the A. tumefaciens strain EHA105 harboring a binary vector pCAMBIA1301 with gusA and hygromycin phosphotransferase II gene (hpt II) genes. Selection culture was conducted using 25 mg l−1 hygromycin. A total of 2,218 plants were regenerated from the inoculated 1,776 cell aggregates via somatic embryogenesis. β-glucuronidase (GUS) assay and PCR, dot blot and Southern blot analyses of the regenerated plants randomly sampled showed that 90.37% of the regenerated plants were transgenic plants. The number of integrated T-DNA copies varied from 1 to 4. Transgenic plants, when transferred to soil in a greenhouse and a field, showed 100% survival. No morphological variations were observed in the ex vitro transgenic plants. These results exceed all transformation experiments reported so far in the literature in quantity of independent events per transformation experiment in sweetpotato.






Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- AS:
-
Acetosyringone
- Carb:
-
Carbenicillin
- CTAB:
-
Cetyltrimethylammonium bromide
- 2,4-d :
-
2,4-Dichlorophenoxyacetic acid
- GUS:
-
β-Glucuronidase
- hpt II:
-
Hygromycin phosphotransferase II gene
- Hyg:
-
Hygromycin
- LB:
-
Luria–Bertani
- MS:
-
Murashige and Skoog
- npt II:
-
Neomycin phosphotransferase II gene
References
Al-Mazrooei S, Bhatti MH, Henshaw GG (1997) Optimisation of somatic embryogenesis in fourteen cultivars of sweetpotato [Ipomoea batatas (L.)Lam.]. Plant Cell Rep 16:710–714
Cipriani G, Fuentes S, Bello V, Salazar LF, Ghislain M, Zhang DP (2001) Transgene expression of rice cysteine proteinase inhibitors for the development of resistance against sweetpotato feathery mottle virus. CIP Program Rep 1999–2000:267–271
Cipriani G, Michaud D, Brunelle F, Golmirzaie A, Zhang DP (1999) Expression of soybean proteinase inhibitor in sweetpotato. CIP Program Rep 1997–1998:271–277
Dhir SK, Oglesby J, Bhagsari AS (1998) Plant regeneration via embryogenesis, and transient gene expression in sweetpotato protoplasts. Plant Cell Rep 17:665–669
Duncan DB (1955) Multiple range and multiple F tests. Biometrics 11:1–42
Gama MICS, Leite RP Jr, Cordeiro AR, Cantliffe DJ (1996) Transgenic sweetpotato plants obtained by Agrobacterium tumefaciens-mediated transformation. Plant Cell Tiss Organ Cult 46:237–244
Guo JM, Liu QC, Zhai H, Wang YP (2006) Regeneration of plants from Ipomoea cairica L. protoplasts and production of somatic hybrids between I.cairica L. and sweetpotato, I. batatas (L.) Lam. Plant Cell Tiss Organ Cult 87:321–327
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusion: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Kimura T, Otani M, Noda T, Ideta O, Shimada T, Saito A (2001) Absence of amylose in sweetpotato [Ipomoea batatas (L.) Lam.] following the introduction of granule-bound starch synthase cDNA. Plant Cell Rep 20:663–666
Lawton R, Winfield S, Daniell H, Bhagsagi AS, Dhir SK (2000) Expression of green-fluorescent protein gene in sweetpotato tissues. Plant Mol Biol Rep 18:139a–139i
Liu QC, Mi KX, Lu DH, Zhou HY, Fu Z (1997) Establishment of embryogenic cell suspension cultures in sweetpotato, Ipomoea batatas (L.) Lam. Acta Agric Sin 23:22–26
Liu QC, Zhai H, Wang Y, Zhang DP (2001) Efficient plant regeneration from embryogenic suspension cultures of sweetpotato. In Vitro Cell Dev Biol-Plant 37:564–567
Luo HR, Santa Maria M, Benavides J, Zhang DP, Zhang YZ, Ghislain M (2006) Rapid genetic transformation of sweetpotato (Ipomoea batatas (L.) Lam.) via organogenesis. Afr J Biotechnol 5:1851–1857
Morán R, García R, López A, Zaldúa Z, Mena J, García M, Armas R, Somonte D, Rodríguez J, Gómez M, Pimentel E (1998) Transgenic sweetpotato plants carrying the delta-endotoxin gene from Bacillus thuringiensis var. tenebrionis. Plant Sci 139:175–184
Newell CA, Lowe JM, Merryweather A, Rooke LM, Hamilton WDO (1995) Transformation of sweetpotato (Ipomoea batatas (L.) Lam.) with Agrobacterium tumefaciens and regeneration of plants expressing cowpea trypsin inhibitor and snowdrop lectin. Plant Sci 107:215–227
Okada Y, Saito A, Nishiguchi M, Kimura T, Mori M, Hanada K, Sakai J, Miyazaki C, Matsuda Y, Murata T (2001) Virus resistance in transgenic sweetpotato [Ipomoea batatas L. (Lam.)] expressing the coat protein gene of sweetpotato feathery mottle virus. Theor Appl Genet 103:743–751
Otani M, Mii M, Handa T, Kamada H, Shimada T (1993) Transformation of sweetpotato (Ipomoea batatas (L.) Lam.) plants by Agrobacterium rhizogenes. Plant Sci 94:151–159
Otani M, Shimada T, Kimura T, Saito A (1998) Transgenic plant production from embryogenic callus of sweetpotato (Ipomoea batatas (L.) Lam.) using Agrobacterium tumefaciens. Plant Biotechol 15:11–16
Otani M, Wakita Y, Shimada T (2001) Genetic transformation of sweetpotato (Ipomoea batatas (L.) Lam.) by Agrobacterium tumefaciens. Acta Hortic 560:193–196
Otani M, Wakita Y, Shimada T (2003) Production of herbicide-resistant sweetpotato (Ipomoea batatas (L.) Lam.) plants by Agrobacterium tumefaciens-mediated transformation. Breed Sci 53:145–148
Prakash CS, Varadarajan U (1992) Genetic transformation of sweetpotato by particle bombardment. Plant Cell Rep 11:53–57
Saghai-Maroof MA, Soliman KM, Jorgensen RA, Allard RW (1984) Ribosomal DNA space length polymophisms in barly: mendelian inheritance, chromosomal locations and population dynamics. Proc Natl Acad Sci 81:8014–8018
Shimada T, Otani M, Hamada T, Kim SH (2006) Increase of amylose content of sweetpotato starch by RNA interference of the starch branching enzyme II gene (IbSBEII). Plant Biotechnol 23:85–90
Song GQ, Honda H, Yamaguchi KI (2004) Efficient Agrobacterium tumefaciens-mediated transformation of sweetpotato (Ipomoea batatas (L.) Lam.) from stem explants using a two-step kanamycin-hygromycin selection method. In Vitro Cell Dev Biol-Plant 40:359–365
Trick HN, Finer JJ (1998) Sonication-assisted Agrobacterium-mediated transformation of soybean [Glycine max (L.) Merrill] embryogenic suspension culture tissue. Plant Cell Rep 17:482–488
Wakita Y, Otani M, Hamada T, Mori M, Iba K, Shimada T (2001) A tobacco microsomal ω−3 fatty acid desaturase gene increases the linolenic acid content in transgenic sweetpotato (Ipomoea batatas). Plant Cell Rep 20:244–249
Wang JS, Sato M, Taura S, Kokubu T (1998) Efficient embryogenic callus formation and plant regeneration in shoot tip cultures of sweetpotato. Mem Fac Agric Kagoshima Univ 34:61–64
Zhai H, Liu QC (2003) Studies on the genetic transformation of embryogenic suspension cultures in sweetpotato. Sci Agric Sin 36:487–491
Acknowledgments
This work was supported by Outstanding Younger Science Foundation of China (no. 30225028) and The National Science and Technology Project of China (no. 2006BAD01A06).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, B., Zhai, H., Wang, Y. et al. Efficient Agrobacterium tumefaciens-mediated transformation using embryogenic suspension cultures in sweetpotato, Ipomoea batatas (L.) Lam.. Plant Cell Tiss Organ Cult 90, 265–273 (2007). https://doi.org/10.1007/s11240-007-9265-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-007-9265-9