Abstract
Intraoperative antithrombotic drug removal by haemoadsorption is a novel strategy to reduce perioperative bleeding in patients on antithrombotic drugs undergoing cardiac surgery. The international STAR registry reports real-world clinical outcomes associated with this application. All patients underwent cardiac surgery before completing the recommended washout period. The haemoadsorption device was incorporated into the cardiopulmonary bypass (CPB) circuit. Patients on P2Y12 inhibitors comprised group 1, and patients on direct-acting oral anticoagulants (DOAC) group 2. Outcome measurements included bleeding events according to standardised definitions and 24-hour chest-tube-drainage (CTD). 165 patients were included from 8 institutions in Austria, Germany, Sweden, and the UK. Group 1 included 114 patients (62.9 ± 11.6years, 81% male) operated at a mean time of 33.2 h from the last P2Y12 inhibitor dose with a mean CPB duration of 117.1 ± 62.0 min. Group 2 included 51 patients (68.4 ± 9.4years, 53% male), operated at a mean time of 44.6 h after the last DOAC dose, with a CPB duration of 128.6 ± 48.4 min. In Group 1, 15 patients experienced a BARC-4 bleeding event (13%), including 3 reoperations (2.6%). The mean 24-hour CTD was 651 ± 407mL. In Group 2, 8 patients experienced a BARC-4 bleeding event (16%) including 4 reoperations (7.8%). The mean CTD was 675 ± 363mL. This initial report of the ongoing STAR registry shows that the intraoperative use of a haemoadsorption device is simple and safe, and may potentially mitigate the expected high bleeding risk of patients on antithrombotic drugs undergoing cardiac surgery before completion of the recommended washout period.
Clinical registration number: ClinicalTrials.gov identifier: NCT05077124.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antithrombotic drugs are cornerstone therapies for patients with cardiovascular disease. Millions of patients receive chronic treatment with direct-acting oral anticoagulants (DOACs) to reduce stroke risk associated with atrial fibrillation or reduce recurrent events after venous thromboembolism [1]. In addition, P2Y12 inhibitors are routinely used in patients with acute coronary syndromes and after percutaneous coronary interventions [2,3,4]. The major safety risk associated with antithrombotic drugs is bleeding which can be either spontaneous or iatrogenic when such patients require urgent or emergent interventions including cardiac surgery [5].
Many of these antithrombotic drugs (e.g. DOACs and the reversibly binding P2Y12 receptor antagonist ticagrelor) can be effectively removed from the circulation with the use of a polymer bead haemoadsorption device and this novel approach is increasingly used to reduce perioperative bleeding in patients on a broad range of antithrombotic drugs undergoing urgent or emergency cardiac surgery [6,7,8,9]. The international Safe and Timely Antithrombotic Removal (STAR) registry is designed to collect real-world clinical outcomes associated with this application.
Methods
The international Safe and Timely Antithrombotic Removal (STAR) registry is designed to collect high-fidelity data on patients who underwent intraoperative antithrombotic drug removal during cardiac surgery as part of their routine care. Participating institutions can include both prospective and retrospective cases. Collected clinical and resource utilisation data are entered in an electronic case report form (CRF). Safety is assessed by collection of definite, probable, or possible device-related adverse events. Data collection is done up to 30 days post-operation. The sponsor and funding source of the registry is CytoSorbents Inc., Princeton, NJ, USA.
Ethical statement
This registry complies with the Declaration of Helsinki. National central or local approvals of respective Ethics committees were granted for the STAR registry according to local regulations (complete list available in the Supplement). Written consent was obtained before or after surgery from prospective patients and was waived for retrospective patients.
Inclusion criteria
CytoSorb® utilisation for antithrombotic removal and informed consent for prospective registry participation.
Exclusion criteria
Use of CytoSorb® for purposes other than antithrombotic removal.
Patient groups
Group 1 included patients on oral P2Y12 inhibitors, and group 2 patients on DOACs.
Hemoadsorption therapy
Antithrombotic removal via haemoadsorption therapy was performed with the CytoSorb® adsorber (CytoSorbents Inc., Princeton, NJ, USA). This CE-marked device is based on extracorporeal blood purification and is approved to remove ticagrelor and rivaroxaban. The cartridge is filled with highly biocompatible, porous polymer beads covered with a divinylbenzene coating and can be easily integrated into various extracorporeal circuits, such as e.g., continuous renal replacement therapy, extracorporeal membrane oxygenation (ECMO), or cardio-pulmonary bypass (CPB), as shown in Fig. 1. Each polymer bead is between 300 μm and 800 μm in size and has multiple pores and channels, giving it a large (> 40,000 m2) effective surface area for binding hydrophobic small and medium-sized molecules up to 60 kDa of molecular weight [10].
Outcome measures
Bleeding complications were recorded according to the Bleeding Academic Research Consortium (BARC) and Universal Definition of Perioperative Bleeding (UDPB) definitions. Additional outcomes included 24-hour chest tube drainage (CTD), detailed analysis of blood product transfusions, re-operation for bleeding, and in-hospital mortality. Safety of the device was assessed by investigator-reported adverse device events, including severity and related classifications. A detailed classification list is given in the Supplement.
Results
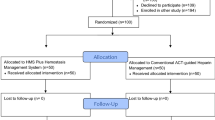
Eight centres from 4 countries (Austria, Germany, Sweden, United Kingdom) enrolled a total of 165 patients by the end of April 2023.
Significant baseline differences were noted between groups and 1 and 2 including age, gender, and qualifying diagnosis (Table 1). Patients on all 3 available P2Y12 inhibitors were included; however, the vast majority were treated with ticagrelor (Fig. 2). In group 2, almost half of the patients were on apixaban, and approximately one-quarter each were on either rivaroxaban or edoxaban (Fig. 2). All antithrombotic drugs were given according to their approved indication with a high prevalence of atrial fibrillation in group 2 (Table 1). The mean washout period in both groups was less than 48 h.
Procedural outcomes are summarised in Table 2 and depicted in Fig. 3A and B. In Group 1, the vast majority of patients underwent isolated coronary artery bypass grafting (i-CABG) (Fig. 3A). In group 2, there was an almost equal distribution between i-CABG, combined procedures (CABG + heart valves), isolated heart valve procedures (i-Valve), and aortic surgery (Fig. 3B). The mean period between the last dose and surgery was shorter in group 1 compared with group 2 (33.2 ± 26.1 vs. 44.6 ± 33.1 h respectively, p = 0.05). Both CPB duration, which also represents device exposure time, and aortic cross clamp times were comparable and not significantly different between the two groups (Table 2). Moreover, in the i-CABG population no significant difference in regard to device exposure time was observed.
Bleeding complications
Serious postoperative bleeding complications according to either BARC or UDPB definitions were not significantly different between the two groups (Fig. 4A). BARC-4 bleeding occurred in 13.2% in group 1 vs. 15.7% in group 2. Total 24-hour CTD also did not differ significantly between groups (Table 2). Blood product transfusions are summarised in Table 3. Packed red blood cells (pRBC) were not needed during the first 24-hours after surgery in 66% of group 1 and in 57% of group 2, while more than 5 units were required in only 2 patients in group 1. 75% and 84% patients in group 1, and 96% and 76% in group 2 did not require platelets or fresh frozen plasma (FFP), respectively. More than 5 units of platelets or FFP were needed in 3% of patients (6% of group 1 patients and no patients in group 2). Surgical revisions within 48 h due to ongoing bleeding were numerically higher in group 2 (7.8% vs. 2.6%; p = 0.2). Finally, patients in group 1 undergoing i-CABG represented the largest uniform subgroup in terms of type of antithrombotic drug and type of surgery. The rates of bleeding complications for this specific cohort are depicted in Fig. 4B. One severe postoperative bleeding event was caused by a bleeding left mammary artery side branch.
A total of 19 patients (17%) in the P2Y12 inhibitor group were treated by clopidogrel as the antithrombotic agent. These patients compared to the other 95 patients in Group 1 showed significant higher CTD (905 ± 433mL vs. 596 ± 386mL, p = 0.012), received numerically higher rates of platelet transfusions (36.8% vs. 23.2%, p = 0.25), and also experienced more UDPB class 3 bleeding events (p = 0.01). Of note, none of the included patients received recombinant factor VIIa (rFVIIa, NovoSeven®).
Mortality
Mortality at 30 days in the overall registry cohort was 9.1% (15/165) with 7.0% in group 1 compared to 13.7% in group 2, showing no significant difference (p = 0.24). The corresponding preoperative EuroSCORE-II for the whole cohort was 7.6 ± 11.2% in group 1 and 8.3 ± 10.5% in group 2 (p = 0.75).
Non-survivors had very high preoperative risk with a EuroSCORE-II of 27 ± 19% compared to 6.0 ± 7.8% in survivors (p < 0.001), underwent more emergent/urgent procedures and required significantly more postoperative mechanical circulatory support with extracorporeal membrane oxygenation (ECMO) or TandemHeart® compared to survivors (26.7% vs. 6.7%, p = 0.02). In the three patients receiving postoperative mechanical circulatory support, 2 BARC-4 bleeding events occurred, however without any surgical re-exploration. In the i-CABG cohort of group 1, the 4 deaths that occurred were all emergency procedures. A detailed breakdown of the mortalities is presented in Table 4. All deaths were cardiac-related and associated with low output states and multi-organ failure. No fatal bleeding leading to death occurred.
Safety
All participating sites assessed the device as safe according to the ISO 14155:2020 classification (detailed classification given in the Supplement) and did not report any (serious) device related adverse events.
Discussion
This initial report from the ongoing international STAR registry summarises the outcomes and bleeding complications in patients treated with oral antithrombotics undergoing cardiac surgery before the recommended washout period. The following main observations can be derived from the ongoing registry. First, the P2Y12 inhibitor group consisted mainly of ticagrelor patients, whereas in the DOAC group, apixaban was the most frequent drug prescribed. Second, the index operations differed between the groups due to the different underlying conditions requiring treatment with antiplatelets versus anticoagulants. Third, the overall incidence of serious perioperative bleeding complications was acceptable, including the need for surgical re-operations for bleeding control (overall 7 re-operations in 165 patients: 4.2%). Fourth, the overall 30-day mortality of 9% was high and likely related to the very high operative risk of emergent/urgent operations. Importantly, the removal of antithrombotics with intraoperative haemoadsorption was simple and safe, without any device-related adverse events reported.
In the current analysis, we sought to present data on two different groups: First, the group treated by preoperative P2Y12 inhibitors (consisting mainly of CABG patients) and a second group with preoperative DOAC treatment (consisting mainly of complex or combined surgeries). Both antithrombotic groups differed regarding their surgical indication and demographics. Group 2 included mainly elderly, high-risk patients undergoing more complex, long-lasting surgeries (including combination surgeries), or major aortic surgery (including aortic type A dissections). These patients are per se presenting with a high baseline risk for surgery and the pre-existing high risk for postoperative bleeding is further aggravated by the presence of antithrombotic agents.
Of note, 19 patients on clopidogrel have been included into the P2Y12 inhibitor group. It has to be acknowledged that since clopidogrel, in contrast to ticagrelor, is irreversibly bound to platelets, it remains unclear whether removal via haemoadsorption is to be expected. The serious bleeding rates reported in this analysis compare favourably to historical benchmarks. Patients on dual anti-platelet therapy (DAPT) with ticagrelor and acetylsalicylic acid undergoing isolated CABG surgery within 24 h after drug discontinuation have been reported with an incidence of BARC-4 bleeding of 38% and a 24-hour CTD of 813 ± 554mL, resulting in a re-exploration rate of 6.1% [11]. In a more recent study, a postoperative 24-hour CTD of 698 ± 409mL and a surgical re-exploration rate of 8.3% was reported [12] after a washout period of 24 h. In the current analysis, patients in group 1 undergoing isolated CABG had an incidence of BARC-4 bleeding of 4.5%, mean 24-hour CTD of 651 ± 407mL and a re-exploration rate of only 2.6%, therefore suggesting that intraoperative haemoadsorption may lower the high baseline bleeding risk of patients on P2Y12 inhibitors undergoing cardiac surgery before completing the recommended washout period. It should be acknowledged, however, that in the current analysis the washout period of the isolated CABG group was 33.2 h compared to 24 h in the dataset of Hansson et al. [11].
The European Multicenter Registry on Coronary Artery Bypass Grafting (E-CABG) reported a higher incidence of UDPB severe or massive bleeding in ticagrelor-treated patients when ticagrelor was discontinued 0–2 days, which corresponds to the current analysis with 1.4 days of washout in group 1. Holm et al. reported a UDPB class 3/4 bleeding rate of 16.0%, which was lower in the current analysis (UDPB class > 3: 10.1%). In addition, the current analysis also showed a lower incidence of BARC-4 bleeding events (6.7%) in the isolated CABG group compared to the E-CABG registry (11.8%) [5].
In a previous analysis of cardiac surgery patients under DOAC medications, it was demonstrated that an increased incidence of bleeding is observed up to 10 days after drug cessation [6]. Specifically in patients undergoing urgent aortic surgery, DOAC use was independently associated with increased perioperative mortality [13]. Current guidelines recommend that, when possible, patients should discontinue DOACs at least 2 days prior to surgery; however, surgeons routinely report that their standard washout period is longer [14].
Although surgical re-operation for bleeding control was required in only 7 out of 165 patients (4.2%) in the overall cohort, it appeared to be slightly more frequent in DOAC patients who underwent more complex and combined procedures. In a previous study, it was demonstrated that in patients on apixaban discontinued < 48 h before surgery, the 24-hour CTD was 893 ± 579mL with a re-thoracotomy rate of 8.3% [8]. In group 2 of the current STAR registry analysis, we observed a lower 24-hour CTD of 675 ± 363mL but noted a similar surgical re-exploration for bleeding rate of 7.8%. Since 24-hr CTD is a strong independent predictor of surgical outcomes including mortality [15, 16], the current results again support the use of intraoperative haemoadsorption in patients on DOAC undergoing cardiac surgery before completing the recommended washout period.
An overall mortality at 30 days of 9% in the overall cohort must be viewed in the context of a high percentage of urgent/emergent indications and also the complexity of the index surgical procedures. In addition, a fairly high number of patients in the current analysis required mechanical extracorporeal circulatory support in the perioperative period which historically defines patients with high perioperative mortality ranging between 15 and 25% [17, 18]. The preoperative assessed EuroSCORE-II in both groups tended to reflect, per its definition, a high-risk cohort (EuroSCORE-II 7.6 ± 11.2% in group 1 and 8.3 ± 10.5% in group 2). Specifically, among non-survivors the baseline EuroSCORE-II was 27 ± 19% and much higher compared to a value of 6.0 ± 7.8% in survivors, supporting the notion that mortality was directly linked to very high baseline risk. A recent published analysis analysed the bleeding events in patients undergoing surgical revascularisation in patients receiving dual-antiplatelet therapy < 72 h before surgery. They reported major bleeding events according to BARC-4 of 44.3% (10.5% surgical revisions) resulting in an in-hospital mortality of 9.0% [19].
It has been previously shown in a benchtop model that the CytoSorb® haemoadsorption device (CytoSorbents, Inc., Princeton, NJ, USA) effectively reduces DOAC (apixaban and rivaroxaban) and ticagrelor levels in a time-dependent fashion [20]. It is therefore postulated that actively reducing DOAC or ticagrelor levels can lower the bleeding risk in such patients operated on before complete drug washout. These observations were recently validated by the results of a clinical study measuring ticagrelor levels before and after CPB during which haemoadsorption was utilised for antithrombotic removal [21]. In this first-in-human analysis, it was demonstrated that a mean CPB + haemoadsorption time of 97 ± 43 min led to a significant reduction in ticagrelor levels by 67% (p < 0.001). An additional important benefit of reduced circulating ticagrelor levels is that it may allow platelet transfusions to work more effectively. Previous reports have shown that platelet reactivity remained unchanged following transfusion of platelets to ticagrelor-treated patients [22], an observation likely explained by the reversible mode of binding of ticagrelor to platelets that renders newly transfused platelets also vulnerable to inhibition.
Whether intraoperative ticagrelor removal with haemoadsorption reduces perioperative bleeding in patients on ticagrelor undergoing cardiac surgery is currently investigated in the pivotal, double blind, randomised Safe and Timely Antithrombotic Removal – Ticagrelor (STAR-T) trial in the US and Canada (ClinicalTrials.gov Identifier: NCT04976530) [23].
In a case report, Dalmastri et al. described the successful preoperative reduction of apixaban levels in a patient scheduled for emergency bilateral nephrostomy by 48% after 150 min. of haemoadsorption during renal replacement therapy [24]. Therefore, analogous results are to be expected in further clinical trials aiming at significant DOAC removal before major surgery.
Importantly, no serious adverse device-related events were observed as classified by all investigators. This is in line with previous observations using the haemoadsorption device in different clinical settings [25]. Hence, the most recent ESAIC Guidelines for the management of severe perioperative bleeding [26] provided a class 2 C recommendation for the use of haemoadsorption as an adjuvant in patients on ticagrelor or rivaroxaban undergoing emergency cardiac/aortic surgery on cardiopulmonary bypass to reduce bleeding complications.
Limitations
Our study has three major limitations that have to be considered when interpreting the results. First, since the current data is derived from a single-arm observational registry, a control group (without adsorber use) is missing. Future trials should analyse the current findings in a randomized or propensity-score matched fashion. Moreover, future trials should consider including coagulation or platelet function testing to determine the potential return of haemostatic activity after surgery. Second, comprehensive coagulation or platelet function testing results were not available to accurately determine the impact of the residual oral antithrombotics following cardiac surgery. Third, due to the “open” and all-comers real-world inclusion intention of the registry, results might be biased by the inclusion of many patients that would routinely be excluded from studies with strict inclusion and exclusion criteria (i.e. emergencies, high risk cases). Finally, we only presented the mandatory and complete data available for packed red blood cells or platelet transfusions without having detailed data on other blood products given (e.g. tranexamic acid, fibrinogen etc.).
Conclusion
This initial report of the ongoing STAR registry shows that the intraoperative use of a haemoadsorption device may potentially mitigate the expected high bleeding risk of patients on antithrombotic drugs undergoing cardiac surgery before completion of the recommended washout period. Moreover, in patients on antithrombotic drugs undergoing cardiac surgery before the recommended washout period, the intraoperative use of hemoadsorption was reported by investigators to be easy to implement and generally safe. Whether active antithrombotic removal can reduce serious perioperative bleeding in patients undergoing urgent cardiac surgery compared to control subjects who are not treated with the device is currently being evaluated in the double-blind, randomized Safe and Timely Antithrombotic Removal-Ticagrelor (STAR-T) trial.
Data availability
The data underlying this article were provided by CytoSorbents Inc., Princeton, NJ, USA. Data will be shared on request to the corresponding author with permission of CytoSorbents.
Abbreviations
- ACC:
-
Aortic Cross Clamp
- BARC:
-
Bleeding Academic Research Consortium
- BMI:
-
Body Mass Index
- CABG:
-
Coronary Artery Bypass Grafting
- CPB:
-
Cardio-pulmonary bypass
- CRF:
-
Case Report Form
- CTD:
-
Chest tube drainage
- DOAC:
-
Direct Oral Anticoagulant drug
- ESAIC:
-
European Society of Anaesthesiology and Intensive Care
- HTx:
-
Heart Transplantation
- i-CABG:
-
Isolated CABG
- i-Valve:
-
Isolated heart valve procedure
- STAR:
-
Safe and Timely Antithrombotic Removal
- UDPB:
-
Universal Definition of Perioperative Bleeding
References
Chan NC, Weitz JI (2019) Antithrombotic agents. Circ Res 124(3):426–436. https://doi.org/10.1161/CIRCRESAHA.118.313155
Sanderson NC, Parker WAE, Storey RF (2021) Ticagrelor: clinical development and future potential. Rev Cardiovasc Med 22(2):373–394. https://doi.org/10.31083/j.rcm2202044
Wallentin L, Becker RC, Budaj A, Cannon CP, Emanuelsson H, Held C et al (2009) Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 361(11):1045–1057. https://doi.org/10.1056/NEJMoa0904327
Bonaca MP, Bhatt DL, Cohen M, Steg PG, Storey RF, Jensen EC et al (2015) Long-term use of ticagrelor in patients with prior myocardial infarction. N Engl J Med 372(19):1791–1800. https://doi.org/10.1056/NEJMoa1500857
Holm M, Biancari F, Khodabandeh S, Gherli R, Airaksinen J, Mariscalco G et al (2019) Bleeding in patients treated with Ticagrelor or Clopidogrel before Coronary Artery Bypass Grafting. Ann Thorac Surg 107(6):1690–1698. https://doi.org/10.1016/j.athoracsur.2019.01.086
Hassan K, Bayer N, Schlingloff F, Oberhoffer M, Wohlmuth P, Schmoeckel M, Geidel S (2018) Bleeding complications after use of novel oral anticoagulants in patients undergoing cardiac surgery. Ann Thorac Surg 105(3):702–708. https://doi.org/10.1016/j.athoracsur.2017.11.066
Hassan K, Kannmacher J, Wohlmuth P, Budde U, Schmoeckel M, Geidel S (2019) Cytosorb Adsorption during Emergency Cardiac operations in patients at high risk of bleeding. Ann Thorac Surg 108(1):45–51. https://doi.org/10.1016/j.athoracsur.2018.12.032
Hassan K, Thielmann M, Easo J, Kamler M, Wendt D, Haidari Z et al (2022) Removal of apixaban during emergency cardiac surgery using hemoadsorption with a porous polymer bead sorbent. J Clin Med 11(19):5889. https://doi.org/10.3390/jcm11195889
Cao D, Amabile N, Chiarito M, Lee VT, Angiolillo DJ, Capodanno D et al (2023) Reversal and removal of oral antithrombotic drugs in patients with active or perceived imminent bleeding. Eur Heart J 44(20):1780–1794. https://doi.org/10.1093/eurheartj/ehad119
Nierhaus A, Morales J, Wendt D, Scheier J, Gutzler D, Jarczak D et al (2022) Comparison of the CytoSorb((R)) 300 mL and Jafron HA380 hemoadsorption devices: an in vitro study. Minim Invasive Ther Allied Technol 31(7):1058–1065. https://doi.org/10.1080/13645706.2022.2104617
Hansson EC, Jideus L, Aberg B, Bjursten H, Dreifaldt M, Holmgren A et al (2016) Coronary artery bypass grafting-related bleeding complications in patients treated with ticagrelor or clopidogrel: a nationwide study. Eur Heart J 37(2):189–197. https://doi.org/10.1093/eurheartj/ehv381
Nardi P, Pisano C, Turturici M, Bertoldo F, Maggio VR, Bassano C et al (2021) The impact of dual antiplatelet therapy administration on the risk of bleeding complications during coronary artery bypass surgery. Kardiochir Torakochirurgia Pol 18(3):145–151. https://doi.org/10.5114/kitp.2021.109407
Bjornstad JL, Khan AM, Roed-Undlien H, Bendz B, Nygard S, Hoel TN, Lingaas PS (2020) Operative survival in patients with acute aortic disease in the era of newer oral anticoagulants. Open Heart 7(2):e001278. https://doi.org/10.1136/openhrt-2020-001278
Task Force on Patient Blood Management for Adult Cardiac Surgery of the European Association for, Cardio-Thoracic S, the European Association of, Cardiothoracic A, Boer C, Meesters MI, Milojevic M, Benedetto U et al (2018) 2017 EACTS/EACTA Guidelines on patient blood management for adult cardiac surgery. J Cardiothorac Vasc Anesth. ;32(1):88–120. https://doi.org/10.1053/j.jvca.2017.06.026
Christensen MC, Dziewior F, Kempel A, von Heymann C (2012) Increased chest tube drainage is independently associated with adverse outcome after cardiac surgery. J Cardiothorac Vasc Anesth 26(1):46–51. https://doi.org/10.1053/j.jvca.2011.09.021
Dixon B, Santamaria JD, Reid D, Collins M, Rechnitzer T, Newcomb AE et al (2013) The association of blood transfusion with mortality after cardiac surgery: cause or confounding? (CME). Transfusion 53(1):19–27. https://doi.org/10.1111/j.1537-2995.2012.03697.x
Liakopoulos OJ, Schlachtenberger G, Wendt D, Choi YH, Slottosch I, Welp H et al (2019) Early clinical outcomes of Surgical Myocardial revascularization for Acute Coronary syndromes complicated by cardiogenic shock: a Report from the North-Rhine-Westphalia Surgical Myocardial Infarction Registry. J Am Heart Assoc 8(10):e012049. https://doi.org/10.1161/JAHA.119.012049
Thielmann M, Wendt D, Slottosch I, Welp H, Schiller W, Tsagakis K et al (2021) Coronary artery bypass graft surgery in patients with Acute Coronary syndromes after primary percutaneous coronary intervention: a current report from the North-Rhine Westphalia Surgical Myocardial Infarction Registry. J Am Heart Assoc 10(18):e021182. https://doi.org/10.1161/JAHA.121.021182
Schlachtenberger G, Deppe AC, Gerfer S, Choi YH, Zeriouh M, Liakopoulos O, Wahlers TCW (2020) Major bleeding after Surgical revascularization with dual antiplatelet therapy. Thorac Cardiovasc Surg 68(8):714–722. https://doi.org/10.1055/s-0040-1710508
Tripathi R, Morales J, Lee V, Gibson CM, Mack MJ, Schneider DJ et al (2022) Antithrombotic drug removal from whole blood using haemoadsorption with a porous polymer bead sorbent. Eur Heart J Cardiovasc Pharmacother 8(8):847–856. https://doi.org/10.1093/ehjcvp/pvac036
Hassan K, Geidel S, Zamvar V, Tanaka K, Knezevic-Woods Z, Wendt D et al (2023) Intraoperative ticagrelor removal via hemoadsorption during on-pump coronary artery bypass grafting. JTCVS Open 15:190–196. https://doi.org/10.1016/j.xjon.2023.04.013
Laine M, Panagides V, Frere C, Cuisset T, Gouarne C, Jouve B et al (2021) On-Ticagrelor platelet reactivity and clinical outcome in patients undergoing percutaneous coronary intervention for Acute Coronary Syndrome. Thromb Haemost 121(7):923–930. https://doi.org/10.1055/a-1326-5110
Gibson CM, Mack MJ, Lee VT, Schneider DJ, Sellke FW, Ohman EM et al (2022) Rationale and design of the safe and timely antithrombotic removal - ticagrelor (STAR-T) trial: a prospective, multi-center, double-blind, randomized controlled trial evaluating reductions in postoperative bleeding with intraoperative removal of ticagrelor by the drugsorb-ATR device in patients undergoing cardiothoracic surgery within 48 hours from last ticagrelor dose. Am Heart J 245:19–28. https://doi.org/10.1016/j.ahj.2021.10.188
Dalmastri V, Angelini A, Minerva V, Ballarini M, Grammatico F, Todeschini P et al (2023) Extracorporeal hemoadsorption therapy as a potential therapeutic option for rapid removal of Apixaban in high risk-surgical patients: a case report. J Med Case Rep 17(1):283. https://doi.org/10.1186/s13256-023-03949-3
Diab M, Lehmann T, Bothe W, Akhyari P, Platzer S, Wendt D et al (2022) Cytokine hemoadsorption during cardiac surgery Versus Standard Surgical Care for Infective Endocarditis (REMOVE): results from a Multicenter Randomized Controlled Trial. Circulation 145(13):959–968. https://doi.org/10.1161/CIRCULATIONAHA.121.056940
Kietaibl S, Ahmed A, Afshari A, Albaladejo P, Aldecoa C, Barauskas G et al (2023) Management of severe peri-operative bleeding: guidelines from the European Society of Anaesthesiology and Intensive Care: second update 2022. Eur J Anaesthesiol 40(4):226–304. https://doi.org/10.1097/EJA.0000000000001803
Acknowledgements
We would like to thank Weihong Fan for her important statistical support that made this analysis possible.
Funding
The international STAR registry is funded by CytoSorbents Inc., Princeton, NJ, USA.
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Michael Schmoeckel, Matthias Thielmann, Kambiz Hassan, Stephan Geidel, Sandra Lindstedt, received speaker honoraria and travel fees. Marijana Matejic-Spasic, Daniel Wendt are full-time employees of CytoSorbents Europe GmbH, Berlin, Germany, Efthymios Deliargyris is a full-time employee of CytoSorbents Inc., Princeton, NJ, USA. Robert Storey reports institutional research grants/support from Cytosorbents and AstraZeneca; and personal fees from Alfasigma, AstraZeneca, Boehringer Ingelheim/Lilly, Chiesi, Cytosorbents, Daiichi Sankyo, Idorsia, Novartis, Novo Nordisk, Pfizer, PhaseBio and Tabuk.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schmoeckel, M., Thielmann, M., Hassan, K. et al. Intraoperative haemoadsorption for antithrombotic drug removal during cardiac surgery: initial report of the international safe and timely antithrombotic removal (STAR) registry. J Thromb Thrombolysis (2024). https://doi.org/10.1007/s11239-024-02996-x
Accepted:
Published:
DOI: https://doi.org/10.1007/s11239-024-02996-x