Abstract
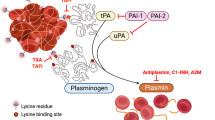
Aminocaproic acid (EACA) availability has recently been decreased whereas tranexamic acid (TXA) is still available as an antifibrinolytic agent to decrease blood loss associated with procedures involving cardiopulmonary bypass (CPB) by inhibiting plasmin mediated platelet activation. Given that the clinical inclination is to substitute TXA for EACA, we sought to compare the antifibrinolytic efficacy of the two agents using the clinically accepted molar ratio of EACA:TXA (7.9:1) that prevents platelet activation in a viscoelastic based system under a variety of conditions in human plasma; 25–50% therapeutic concentration (EACA 32.5–65 µg/ml, TXA 5–10 µg/ml) in the presence of 1500–3000 IU tissue-type plasminogen activator, with 0–50% dilution of plasma with buffer. In all equipotent concentrations, TXA provided superior antifibrinolytic action compared to EACA. It is hoped that this work will serve as a rationale to further investigate these and other similar agents, especially now in a time of unpredictable unavailability of key medications needed to optimize patient care. It is also our wish that these data assist perfusionists, anesthesiologists and cardiothoracic surgeons with their consideration of using an antifibrinolytic agent when managing complex patients with hypercoagulable states (e.g., ventricular assist device explant, infective endocarditis) undergoing CPB.
Similar content being viewed by others
References
Gerstein NS, Brierley JK, Windsor J, Panikkath PV, Ram H, Gelfenbeyn KM, Jinkins LJ, Nguyen LC, Gerstein WH (2017) Antifibrinolytic agents in cardiac and noncardiac surgery: a comprehensive overview and update. J Cardiothorac Vasc Anesth 31:2183–2205
Alkjaersig N, Fletcher AP, Sherry S (1959) ε-Aminocaproic acid: an inhibitor of plasminogen activation. J Biol Chem 234:832–837
Iwamoto M (1975) Plasminogen-plasmin system IX. Specific binding of tranexamic acid to plasmin. Thromb Diath Haemorrh 33:573–585
Lu H, Soria C, Li H, Soria J, Lijnen HR, Perrot JY, Caen JP (1991) Role of active center and lysine binding sites of plasmin in plasmin-induced platelet activation and disaggregation. Thromb Haemost 65:67–72
Soslau G, Horrow J, Brodsky I (1991) Effect of tranexamic acid on platelet ADP during extracorporeal circulation. Am J Hematol 38:113–119
Yurka HG, Wissler RN, Zanghi CN, Liu X, Tu X, Eaton MP, Congenital Heart Surgery Research Interest Group (2010) The effective concentration of epsilon-aminocaproic acid for inhibition of fibrinolysis in neonatal plasma in vitro. Anesth Analg 111:180–184
Yee BE, Wissler RN, Zanghi CN, Feng C, Eaton MP (2013) The effective concentration of tranexamic acid for inhibition of fibrinolysis in neonatal plasma in vitro. Anesth Analg 117:767–772
Nielsen VG, Cankovic L, Steenwyk BL (2007) Epsilon-aminocaproic acid inhibition of fibrinolysis in vitro: should the ‘therapeutic’ concentration be reconsidered? Blood Coagul Fibrinolysis 18:35–39
Nielsen VG, Steenwyk BL, Gurley WQ (2006) Contact activation prolongs clot lysis time in human plasma: role of thrombin-activatable fibrinolysis inhibitor and Factor XIII. J Heart Lung Transplant 25:1247–1252
Myles PS, Smith JA, Forbes A, Silbert B, Jayarajah M, Painter T, Cooper DJ, Marasco S, McNeil J, Bussières JS, McGuinness S, Byrne K, Chan MT, Landoni G, Wallace S, ATACAS Investigators of the ANZCA Clinical Trials Network (2017) Tranexamic acid in patients undergoing coronary-artery surgery. N Engl J Med 376:136–148
van Diepen S, Merrill PD, Carrier M, Tardif JC, Podgoreanu M, Alexander JH, Lopes RD (2017) Association between CK-MB area under the curve and tranexamic acid utilization in patients undergoing coronary artery bypass surgery. J Thromb Thrombolysis 43:446–453
Williams B, Wehman B, Mazzeffi MA, Odonkor P, Harris RL, Kon Z, Tanaka KA (2018) Acute intracardiac thrombosis and pulmonary thromboembolism after cardiopulmonary bypass: a systematic review of reported cases. Anesth Analg 126:425–434
Yu S, Khalpey ZI, Wong RK, Huynh T, Nielsen VG (2018) Complete antithrombin replacement for anticoagulation for cardiopulmonary bypass to repair severe infective mitral valve endocarditis. Blood Coagul Fibrinolysis 29:123–125
Martin K, Knorr J, Breuer T, Gertler R, Macguill M, Lange R, Tassani P, Wiesner G (2011) Seizures after open heart surgery: comparison of ε-aminocaproic acid and tranexamic acid. J Cardiothorac Vasc Anesth 25:20–25
Pilbrant A, Schannong M, Vessman J (1981) Pharmacokinetics and bioavailability of tranexamic acid. Eur J Clin Pharmacol 20:65–72
Funding
This investigation was supported by the Department of Anesthesiology, College of Medicine, at the University of Arizona.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Both authors declare that they have no conflict of interest.
Ethical approval
This was an in vitro investigation and did not involve any living subjects.
Rights and permissions
About this article
Cite this article
Nielsen, V.G., Ford, P.M. The ratio of concentrations of aminocaproic acid and tranexamic acid that prevent plasmin activation of platelets does not provide equivalent inhibition of plasmatic fibrinolysis. J Thromb Thrombolysis 46, 365–370 (2018). https://doi.org/10.1007/s11239-018-1705-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-018-1705-3