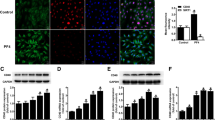
Abstract
Pregnancy-associated plasma protein-A (PAPP-A) is a metalloproteinase with a controversial role in pathophysiology of cardiovascular disease. It seems involved in progression of atherosclerosis and is widely represented in atherosclerotic plaque. PAPP-A plasma levels are elevated in patients with acute coronary syndromes (ACS), thus it has been suggested that it might be a prognostic marker for developing major cardiovascular events. However, the pathophysiological link(s) between PAPP-A and ACS are still unknown. Several studies have indicated that tissue factor (TF) plays a pivotal role in the pathophysiology of ACS by triggering the formation of intracoronary thrombi following endothelial injury. This study investigates whether PAPP-A, at concentrations measurable in ACS patients, might induce TF expression in human endothelial cells in culture (HUVEC). In HUVEC, PAPP-A induced TF-mRNA transcription as demonstrated by real time PCR and expression of functionally active TF as demonstrated by FACS analysis and pro-coagulant activity assay. PAPP-A induced TF expression through the activation of Akt/NF-κB axis, as demonstrated by luciferase assay and by suppression of TF-mRNA transcription as well as of TF expression/activity by Akt and NF-κB inhibitors. These data indicate that PAPP-A promotes TF expression in human endothelial cells and support the hypothesis that this proteinase, besides being involved in progression of atherosclerosis, does not represent an independent risk factor for adverse cardiovascular events, but it rather might play an “active” role in the pathophysiology of ACS as an effector molecule able to induce a pro-thrombotic phenotype in endothelial cells.





Similar content being viewed by others
References
Lin TM, Galbert SP, Kiefer D, Spellacy WN, Gall S (1974) Characterization of four human pregnancy-associated plasma proteins. Am J Obstet Gynecol 118(2):223–236
Oxving C, Sand O, Kristensen T, Kristensen L, Sottrup-Jensen L (1994) Isolation and characterization of circulating complex between pregnancy-associated plasma protein A and proform of eosinophil major basic protein. Biochim Biophys Acta 1201:415–423
Bayes-Genis A, Conover CA, Overgaard MT, Bailey KR, Christiansen M, Holmes DR Jr, Virmani R, Oxvig C, Schwartz RS (2001) Pregnancy-associated plasma protein A as a marker of acute coronary syndromes. N Engl J Med 345:1022–1029
Lund J, Qin QP, Ilva T, Pettersson K, Voipio-Pulkki LM, Porela P, Pulkki K (2003) Circulating pregnancy-associated plasma protein a predicts outcome in patients with acute coronary syndrome but no troponin I elevation. Circulation 108:1924–1926
Lund J, Qin QP, Ilva T, Nikus K, Eskola M, Porela P, Kokkala S, Pulkki K, Pettersson K, Voipio-Pulkki LM (2006) Pregnancy-associated plasma protein A. A biomarker in acute ST-elevation myocardial infarction (STEMI). Ann Med 38:221–228
Elesber AA, Conover CA, Denktas AE, Lennon RJ, Holmes DR Jr, Overgaard MT, Christiansen M, Oxvig C, Lerman LO, Lerman A (2006) Prognostic value of circulating pregnancy-associated plasma protein levels in patients with chronic stable angina. Eur Heart J 27:1678–1684
Harrington SC, Simari RD, Conover CA (2007) Genetic deletion of pregnancy-associated plasma protein-A is associated with resistance to atherosclerotic lesion development in apolipoprotein E-deficient mice challenged with a high-fat diet. Circ Res 100(12):1696–1702
Conover CA, Mason MA, Bale LK, Harrington SC, Nyegaard M, Oxvig C, Overgaard MT (2010) Transgenic overexpression of pregnancy-associated plasma protein-A in murine arterial smooth muscle accelerates atherosclerotic lesion development. Am J Physiol Heart Circ Physiol 299(2):H284–H291
Tang SL, Chen WJ, Yin K, Zhao GJ, Mo ZC, Lv YC, Ouyang XP, Yu XH, Kuang HJ, Jiang ZS, Fu YC, Tang CK (2012) PAPP-A negatively regulates ABCA1, ABCG1 and SR-B1 expression by inhibiting LXRα through the IGF-I-mediated signaling pathway. Atherosclerosis 222(2):344–354
Loftus IM, Naylor AR, Bell RF (2002) Matrix metalloproteinases and atherosclerotic plaque instability. Br J Surg 89:680–694
Sangiorgi G, Mauriello A, Bonanno E, Oxvig C, Conover CA, Christiansen M, Trimarchi S, Rampoldi V, Holmes DR Jr, Schwartz RS, Spagnoli LG (2006) Pregnancy-associated plasma protein-A is markedly expressed by monocyte-macrophage cells in vulnerable and ruptured carotid atherosclerotic plaques: a link between inflammation and cerebrovascular events. J Am Coll Cardiol 47:2201–2211
Libby P, Ridker PM, Maseri A (2002) Inflammation and atherosclerosis. Circulation 105:1135–1143
Ross R (1986) The pathogenesis of atherosclerosis-an update. N Engl J Med 314:488–500
Annex BH, Denning SM, Channon KM, Sketch MH Jr, Stack RS, Morrissey JH, Peters KG (1995) Differential expression of tissue factor protein in directional atherectomy specimens from patients with stable and unstable coronary syndromes. Circulation 91:619–622
Pawashe AB, Golino P, Ambrosio G, Migliaccio F, Ragni M, Chiariello M, Bach R, Garen A, Kinisberg WK (1994) A monoclonal antibody against rabbit tissue factor inhibits thrombus formation in stenotic injured rabbit carotid arteries. Circ Res 74:56–63
Ragni M, Cirillo P, Pascucci I, Scognamiglio A, D’Andrea D, Eramo N, Ezekowitz MD, Pawashe AB, Chiariello M, Golino P (1996) Monoclonal antibody against tissue factor shortens tissue plasminogen activator lysis time and prevents reocclusion in a rabbit model of carotid artery thrombosis. Circulation 93:1913–1918
Wilcox JN, Smith KM, Schwartz SM, Gordon D (1989) Localization of tissue factor in the normal vessel wall and in the atherosclerotic plaque. Proc Natl Acad Sci USA 86:2839–2843
Bierhaus A, Chen J, Liliensiek B, Nawroth PP (2000) LPS and cytokine activated endothelium. Semin Thromb Hemost 26:571–587
Golino P, Ragni M, Cirillo P, Avvedimento VE, Feliciello A, Esposito N, Scognamiglio A, Trimarco B, Iaccarino G, Condorelli M, Chiariello M, Ambrosio G (1996) Effects of tissue factor induced by oxygen free radicals on coronary flow during reperfusion. Nat Med 2:35–40
Cirillo P, Golino P, Calabrò P, Calì G, Ragni M, De Rosa S, Cimmino G, Pacileo M, De Palma R, Forte L, Gargiulo A, Corigliano FG, Angri V, Spagnuolo R, Nitsch L, Chiariello M (2005) C-reactive protein induces tissue factor expression and promotes smooth muscle and endothelial cell proliferation. Cardiovasc Res 68(1):47–55
Cirillo P, Angri V, De Rosa S, Calì G, Petrillo G, Maresca F, D’Ascoli GL, Maietta P, Brevetti L, Chiariello M (2010) Pro athero-thrombotic effects of leptin in human coronary endothelial cells. Thromb Haemost 103(5):1065–1075
Lawrence JB, Oxvig C, Overgaard MT, Sottrup-Jensen L, Gleich GJ, Hays LG, Yates JR 3rd, Conover CA (1999) The insulin-like growth factor (IGF) dependent IGF binding protein-4 protease secreted by human fibroblasts is pregnancy-associated plasma protein-A. Proc Natl Acad Sci USA 96:3149–3153
Boldt HB, Overgaard MT, Laursen LS, Weyer K, Sottrup-Jensen L, Oxvig C (2001) Mutational analysis of the proteolytic domain of pregnancy-associated plasma protein-A (PAPP-A): classification as a metzincin. Biochem J 358:359–367
Cirillo P, Ziviello F, Pellegrino G, Conte S, Cimmino G, Giaquinto A, Pacifico F, Leonardi A, Golino P, Trimarco B (2015) The adipokine apelin-13 induces expression of prothrombotic tissue factor. Thromb Haemost 113(2):363–372
Spirina LV, Bochkareva NV, Kondakova IV, Kolomiets LA, Shashova EE, Koval’ VD, Chernysheva AL, Asadchikova ON (2012) Regulation of insulin-like growth factor and NF-kB by proteasome system in endometrial cancer. Mol Biol 46:452–460
Bayes-Genis A, Conover CA, Schwartz RS (2000) The insulin-like growth factor axis: a review of atherosclerosis and restenosis. Circ Res 86:125–130
Crea F, Andreotti F (2005) Pregnancy associated plasma protein-A and coronary atherosclerosis: marker, friend or foe? Eur Heart J 26:2075–2076
Andreotti F, Rio T, Conti E (2009) Role of PAPP-A in atherothrombosis: message to take home. Atherosclerosis 203:352–354
Parveen N, Subhakumari KN, Krishnan S (2015) Pregnancy associated plasma protein-A (PAPP-A) levels in acute coronary syndrome: a case control study in a tertiary care centre. Indian J Clin Biochem 30(2):150–154
Kane LP, Shapiro VS, Stokoe D, Weiss A (1999) Induction of NF-kappa B by the Akt/PKB kinase. Curr Biol 9:601–604
Ozes ON, Mayo LD, Gustin JA, Pfeffer SR, Pfeffer LM, Donner DB (1999) NF-kappaB activation by tumor necrosis factor requires the Akt serine-threonine kinase. Nature 401:82–85
Barnes PJ, Karin M (1997) Nucler factor-kappa B: a pivotal transcription factor in chronic inflammatory disease. N Engl J Med 336:1066–1071
Collins T, Read MA, Neish AS, Whitley MZ, Thanos D, Maniatis T (1995) Transcriptional regulation of endothelial cell adhesion molecules: NF-κB and cytokine-inducible enhancers. FASEB J 9:899–909
Mackman N (1997) Regulation of the tissue factor gene. Thromb Haemost 78:747–754
Ritchie ME (1998) Nuclear factor-κB is selectively and markedly activated in humans with unstable angina pectoris. Circulation 98:1707–1713
Wilson SH, Best PJ, Edwards WD, Holmes DR Jr, Carlson PJ, Celermajer DS, Lerman A (2002) Nuclear factor-kappaB immunoreactivity is present in human coronary plaque and enhanced in patients with unstable angina pectoris. Atherosclerosis 160:147–153
Bode M, Mackman N (2014) Regulation of tissue factor gene expression in monocytes and endothelial cells: thromboxane A2 as a new player. Vascul Pharmacol 62:57–62
Antoniak S, Mackman N (2016) Tissue factor expression by the endothelium: coagulation or inflammation? Trends Cardiovasc Med
Acknowledgment
This study is dedicated to my daughter Camilla. Honey, you are my “endless love”.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None.
Rights and permissions
About this article
Cite this article
Cirillo, P., Conte, S., Pellegrino, G. et al. Pregnancy-associated plasma protein-A promotes TF procoagulant activity in human endothelial cells by Akt–NF-κB axis. J Thromb Thrombolysis 42, 225–232 (2016). https://doi.org/10.1007/s11239-016-1353-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-016-1353-4