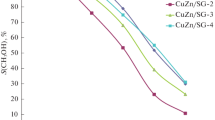
The conversion of dihydroxyacetone solution in methanol to methyl lactate has been studied in flow regime using xSnO2/Al2O3-supported catalysts that have been characterized by XRD, low-temperature nitrogen (ad)desorption analysis, and UV-Vis spectroscopy. It is found that Lewis and Brønsted acid sites of the surface of SnO2-containing catalysts play a crucial role in the selective conversion of dihydroxyacetone to methyl lactate. The formation of methyl lactate with a selectivity of 90% is achieved on 5%SnO2/Al2O3 catalyst at 160°C, 1.0 MPa, and under feed rate of 4 mmol C3H6O3/(gcat·h).



Similar content being viewed by others
References
A. Gupta, V. K. Singh, G. N. Qazi, and A. Kuma, J. Mol. Microbiol. Biotechnol., 3, No. 3, 445-456 (2001).
Yu. Hayashi and Yo. Sasaki, Chem. Commun., 21, 2716-2718 (2005), https://doi.org/10.1039/B501964H.
F. de Clippel, M. Dusselier, R.Van Rompaey, et al., J. Am. Chem. Soc., 134, No. 24, 10089-10101 (2012), https://doi.org/10.1021/ja301678w.
E. Pighin, V.K. Driez, and J. I. Di Cosimo, Appl. Catal. A., 517, 151-160 (2016), https://doi.org/10.1016/j.apcata.2016.03.007.
B. M. Murphy, M. P. Letterio, and B. Xu, ACS Catal., 6, No. 8, 5117-5131 (2016), https://doi.org/10.1021/acscatal.6b00723.
A. M. Mylin, S. I. Levytska, M. E. Sharanda, and V. V. Brei, Catal. Commun., 47, 36-39 (2014), https://doi.org/10.1016/j.catcom.2014.01.004.
S. V. Prudius, N. L. Hes, and V. V. Brei, Colloids Interfaces, 3, No. 1, 16 (2019), https://doi.org/10.3390/colloids3010016.
K. Tanabe, Solid Acids And Bases, Their Catalytic Properties, Acad. Press, New-York, London (1970).
P. Manjunathan, V. S. Marakatti, P. Chandra, et al., Catal. Today, 309, 61-76 (2018), https://doi.org/10.1016/j,cattod.2017.10.009.
R. Bermejo-Deval, R. S. Assary, E. Nikolla, et al., PNAS, 109, No. 25, 9727-9732 (2012), https://doi.org/10.1073/pnas,1206708109.
L. Bricotte, K. Chougrani, V. Alard, et al., Molecules, 28, No. 6, 2724, (2023), https://doi.org/10.3390/molecules28062724.
J. Kùnigstein, Collect. Czechoslov. Chem. Commun., 43, 1152-1158 (1978), https://doi.org/10.1135/cccc19781152.
E. Taarning, S. Saravanamurugan, M. Spangsberg Holm, et al., ChemSusChem., 2, No., 8, 625-627 (2009), https://doi.org/10.1002/cssc.200900099.
E. Jolimaitre, D. Delcroix, N. Essayem, et al., Catal. Sci. Technol., 8, 1349-1356 (2018), https://doi.org/10.1039/C7CY02385E.
G. Innocenti, E. Papadopoulos, G. Fornasari, et al., ACS Catal., 10, No. 20, 11936-11950 (2020), https://doi.org/10.1021/acscatal.0c03761.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Teoretychna ta Eksperymentalna Khimiya, Vol. 59, No. 4, pp. 261-264, July-August, 2023.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hes, N.L., Mylin, A.M. & Prudius, S.V. Catalytic Conversion of Dihydroxyacetone to Methyl Lactate Over SnO2/Al2O3 Catalysts. Theor Exp Chem 59, 302–306 (2023). https://doi.org/10.1007/s11237-024-09789-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11237-024-09789-6