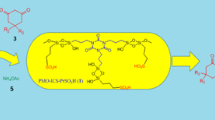
Abstract
Sulfonated mesoporous polydivinylbenzene was prepared by different sulfonation methods and then as a solid acid to catalyze the condensation of phenol and cyclohexanone. The physicochemical properties of the solid acid were characterized by BET, FT-IR, SEM, TG, XRD and acid–base titration; then, the structure–activity relationship of the solid acid was studied. It was found that the catalyst had a sponge like microstructure with the specific surface area of 395 m2/g and average pore diameter of 14.1 nm. The acid–base titration result showed that the acid density of mesoporous polydivinylbenzene solid acid prepared by double sulfonation (D-SPDVB) was the highest, reaching 4.62 mmol H+/g. The catalytic result showed that D-SPDVB had better catalytic performance than Amberlyst-15, sulfuric acid and other traditional Brønsted acids due to its high acid density and suitable pore structure. More importantly, D-SPDVB had good stability, and its catalytic performance did not decrease significantly after reused 5 times.








Similar content being viewed by others
Availability of data and materials
The authors confirm that the data supporting the findings of this study are available within the article.
References
B.G. Manwar, S.H. Kavthia, P.H. Parsania, Eur. Polym. J. 40, 315 (2004)
K. Yoshizawa, S. Toyota, F. Toda, M. Kato, I. Csöregh, CrystEngComm 9, 786 (2007)
Q. Zeng, D. Wu, C. Liu, H. Ma, J. Lu, S. Xu, Y. Li, C. Wang, C. Bai, Crys. Growth Des. 5, 1041 (2005)
C.P. Yang, R.S. Chen, C.W. Yu, J. Appl. Polym. Sci. 82, 2750 (2001)
M.R. Caira, A. Horne, L.R. Nassimbeni, K. Okuda, F. Toda, J. Chem. Soc. Perkin Trans. 2(6), 1063 (1995)
P.H. Parsania, P.P. Shah, K.C. Patel, R.D. Patel, J. Macromol. Sci. A 22, 1495 (1985)
J.A. Desai, U. Dayal, P.H. Parsania, J. Macromol. Sci. A 33, 1113 (1996)
Y.V. Patel, P.H. Parsania, Polym. Test. 21, 711 (2002)
B. Sarma, S. Roy, A. Nangia, Chem. Commun. 47, 4918 (2006)
L. Joshi, P.H. Parsania, Des. Monomers Polym. 16, 185 (2013)
F.D. Karia, P.H. Parsania, Eur. Polym. J. 35, 121 (1999)
Y.S. Kim, J. Yang, S. Wang, A.K. Banthia, J.E. McGrath, Polymer 43, 7207 (2002)
Y. Zhang, G.M. Yan, G. Zhang, S.L. Liu, J. Yang, Polymer 186, 122047 (2020)
G. Latha, N. M, K. Balaji, M. Sevi, High Perform. Polym. 26, 125 (2013)
G. Rizwan, H. Modi, A. Prajapati, C. Patel, P.H. Parsania, Russ. J. Phys. Chem. A 95, 21 (2021)
R.W. Gregor, J. Chem. Educ. 89, 669 (2012)
L.S. Patil, V.S. Suryawanshi, O.B. Pawar, N.D. Shinde, E. J. Chem. 8, 372673 (2011)
C.K. Chozhan, M. Alagar, P. Gnanasundaram, Acta Mater. 57, 782 (2009)
G. Latha, S.C. Murugavel, Polym. Bull. 73, 3237 (2016)
H. Hattori, Top. Catal. 53, 432 (2010)
N.M. Bertero, A.F. Trasarti, M.C. Acevedo, A.J. Marchi, C.R. Apesteguía, Mol. Catal. 481, 110192 (2020)
J.E. Bruno, K.M. Dooley, Appl. Catal. A 497, 176 (2015)
M.M. Garnica, A.E. Torres, G.E. Ramírez-Caballero, P.B. Balbuena, Microporous Mesoporous Mater. 265, 241 (2018)
Y. Román-Leshkov, M. Moliner, J.A. Labinger, M.E. Davis, Angew. Chem Int. Ed. 49, 8954 (2010)
S. Singhal, S. Agarwal, S. Arora, N. Singhal, A. Kumar, Catal. Sci. Technol. 7, 5810 (2017)
X. Liang, Chem. Eng. J. 264, 251 (2015)
Y. Liu, E. Baráth, H. Shi, J. Hu, D.M. Camaioni, J.A. Lercher, Nat. Catal. 1, 141 (2018)
K. Jacobson, R. Gopinath, L.C. Meher, A.K. Dalai, Appl. Catal. B 85, 86 (2008)
P. Kasinathan, C. Lang, S. Radhakrishnan, J. Schnee, C. D’Haese, E. Breynaert, J.A. Martens, E.M. Gaigneaux, A.M. Jonas, A.E. Fernandes, Chem. Eur. J. 25, 6753 (2019)
F. Su, Y. Guo, Green Chem. 16, 2934 (2014)
Y.L. Zhang, S. Wei, F.J. Liu, Y.C. Du, S. Liu, Y.Y. Ji, T. Yokoi, T. Tatsumi, F.S. Xiao, Nano Today 4, 135 (2009)
R.L. Mou, X.J. Wang, Z.Q. Wang, D.Y. Zhang, Z.L. Yin, Y. Lv, Z. Wei, Fuel 302, 121175 (2021)
D.P. Yuan, N. Zhao, Y.X. Wang, K. Xuan, F. Li, Y.F. Pu, F. Wang, L. Li, F.K. Xiao, Appl. Catal. B 240, 182 (2019)
F. Mohajer, G.M. Ziarani, A. Badiei, L.G. Voskressensky, R. Luque, Res. Chem. Intermed. 48, 3739 (2022)
Z.M. Sun, X.Y. Yang, X.P. Huang, M.M. Zhang, G.M. Bian, Y.L. Qi, X.L. Yang, W.Q. Zhang, New J. Chem. 43, 16676 (2019)
M. Halder, P. Bhanja, M.M. Islam, S. Chatterjee, A. Khan, A. Bhaumik, S.M. Islam, Mol. Catal. 494, 111119 (2020)
X. Wang, Q. Deng, Y. Zhang et al., Res. Chem. Intermed. 49, 1369 (2023)
D.Y. Zhao, Q.S. Huo, J.L. Feng, B.F. Chmelka, G.D. Stucky, J. Am. Chem. Soc. 120, 6024 (1998)
F.H. Richter, L. Sahraoui, F. Schuth, Chem. Eur. J. 22, 13563 (2016)
D. Das, J.F. Lee, S.F. Cheng, J. Catal. 223, 152 (2004)
K. Nowinska, W. Kaleta, Appl. Catal. A 203, 91 (2000)
L.J. Hou, Q.H. Cai, B. Lu, X.M. Li, X. Xiao, Y.Y. Han, S.H. Cui, Catal. Lett. 111, 153 (2006)
Funding
This work was supported by the Startup Foundation for Doctors of Yan'an University (YAU202303842 and YAU202213104) and National Natural Science Foundation of China (Nos. 22168040 and 22162025) and the Project of Science and Technology Office of Shaanxi Province (No. 2022JM-062).
Author information
Authors and Affiliations
Contributions
NW was involved in investigation, material preparation, data collection and analysis, writing the original draft and funding acquisition. LG and DW took part in review and funding acquisition. TC and GW participated in data analysis, methodology and review. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Ethical approval
Not applicable for this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, N., Guo, L., Wang, D. et al. A double sulfonated mesoporous polydivinylbenzene as a solid acid to catalyze the condensation of phenol and cyclohexanone. Res Chem Intermed 50, 839–852 (2024). https://doi.org/10.1007/s11164-023-05175-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05175-4