Abstract
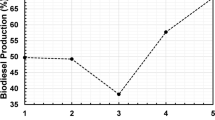
Biomass-derived 5-Hydroxymethylfurfural (HMF) is a vital platform compound for synthesizing biofuel and various high-value chemicals. This work prepared carbonaceous solid acid catalysts with Brønsted acid and Lewis acid using pine biomass as raw materials through chloride salts impregnation, carbonization and sulfonation. The obtained catalysts were characterized by XRD, FTIR, SEM–EDS, BET, and Py-IR. The catalysts were applied to convert glucose into HMF in a biphasic system involving NaCl solution and γ-valerolactone. The results showed that the catalyst of PC-Al-SO3H exhibited larger microspheres and pore sizes compared with the sulfonated catalyst of PC-SO3H without AlCl3 impregnation. Effects of key variables such as reaction temperature, reaction time on conversion of glucose into HMF were examined. By using PC-2Al-SO3H with stronger Lewis acid, glucose conversion and HMF yield achieved 86.53 mol% and 59.62 mol% at 160 °C and 6 h. The cyclic experiments revealed that PC-2Al-SO3H exhibited relatively stable activity.










Similar content being viewed by others
Availability of data and materials
The datasets used or analysed during the current study are available from the corresponding author on reasonable request.
References
A.T. Hoang, H.C. Ong, I.M.R. Fattah, C.T. Chong, C.K. Cheng, R. Sakthivel, Y.S. Ok, Fuel Process. Technol. 223, 106997 (2021)
P. Gautam, Neha, S.N. Upadhyay, S.K. Dubey, Fuel. 273, 117783 (2020)
K. Alper, K. Tekin, S. Karagoz, A.J. Ragauskas, Sustain Energy Fuels 4, 4390 (2020)
Q.D. Hou, X.H. Qi, M.N. Zhen, H.L. Qian, Y.F. Nie, C.Y.L. Bai, S.Q. Zhang, X.Y. Bai, M.T. Ju, Green Chem. 23, 119 (2021)
L.F. Chen, Y.H. Xiong, H. Qin, Z.W. Qi, Chemsuschem. 15, e202102635 (2022)
Q.S. Kong, X.L. Li, H.J. Xu, Y. Fu, Fuel Process. Technol. 209, 106528 (2020)
X. Kong, Y.F. Zhu, Z. Fang, J.A. Kozinski, I.S. Butler, L.J. Xu, H. Song, X.J. Wei, Green Chem. 20, 3657 (2018)
L. Hu, L. Lin, Z. Wu, S.Y. Zhou, S.J. Liu, Renew. Sustain. Energy Rev. 74, 230 (2017)
S.P. Teong, G.S. Yi, Y.G. Zhang, Green Chem. 16, 2015 (2014)
A.I. Torres, M. Tsapatsis, P. Daoutidis, Comput. Chem. Eng. 42, 130 (2012)
V. Choudhary, S.H. Mushrif, C. Ho, A. Anderko, V. Nikolakis, N.S. Marinkovic, A.I. Frenkel, S.I. Sandler, D.G. Vlachos, J. Am. Chem. Soc. 135, 3997 (2013)
I.K.M. Yu, D.C.W. Tsang, Bioresour. Technol. 238, 716 (2017)
H. Li, S. Yang, S. Saravanamurugan, A. Riisager, ACS Catal. 7, 3010 (2017)
W.Q. Wei, S.B. Wu, Chem. Eng. J. 307, 389 (2017)
D. Garces, E. Diaz, S. Ordonez, Ind. Eng. Chem. Res. 56, 5221 (2017)
M. Nahavandi, T. Kasanneni, Z.S. Yuan, C.C. Xu, S. Rohani, Acs Sustain. Chem. Eng. 7, 11970 (2019)
W.Z. Guo, H.J. Heeres, J. Yue, Chem. Eng. J. 381, 122754 (2020)
Y.J. Pagan-Torres, T.F. Wang, J.M.R. Gallo, B.H. Shanks, J.A. Dumesic, ACS Catal. 2, 930 (2012)
J. Zhang, J.Z. Chen, J. Energy Chem. 25, 747 (2016)
A. Munyentwali, H. Li, Q.H. Yang, Appl. Catal. A Gen. 633, 118525 (2022)
J.J. Wang, J.W. Ren, X.H. Liu, J.X. Xi, Q.N. Xia, Y.H. Zu, G.Z. Lu, Y.Q. Wang, Green Chem. 14, 2506 (2012)
I. Jimenez-Morales, M. Moreno-Recio, J. Santamaria-Gonzalez, P. Maireles-Torres, A. Jimenez-Lopez, Appl. Catal. B-Environ. 164, 70 (2015)
T.D. Swift, H. Nguyen, Z. Erdman, J.S. Kruger, V. Nikolakis, D.G. Vlachos, J. Catal. 333, 149 (2016)
H.T. Kreissl, K. Nakagawa, Y.K. Peng, Y. Koito, J.L. Zheng, S.C.E. Tsang, J. Catal. 338, 329 (2016)
Y. Shen, Y.R. Kang, J.K. Sun, C. Wang, B. Wang, F. Xu, R.C. Sun, Chin. J. Catal. 37, 1362 (2016)
M.N. Catrinck, E.S. Ribeiro, R.S. Monteiro, R.M. Ribas, M.H.P. Barbosa, R.F. Teofilo, Fuel 210, 67 (2017)
A. Rezayan, K. Wang, R.F. Nie, J.S. Wang, T.L. Lu, Y.S. Zhang, C.C. Xu, J. Catal. 414, 186 (2022)
A. Mahajan, P. Gupta, Environ. Chem. Lett. 18, 299 (2020)
Z.K. Zhang, Z.Y. Zhu, B.X. Shen, L.N. Liu, Energy 171, 581 (2019)
X. Sun, H.K. Atiyeh, M.X. Li, Y. Chen, Bioresour. Technol. 295, 122252 (2020)
M.G. Mazzotta, D. Gupta, B. Saha, A.K. Patra, A. Bhaumik, M.M. Abu-Omar, Chemsuschem 7, 2342 (2014)
K. Ke, H.R. Ji, X.N. Shen, F.O. Kong, B. Li, Polymers 13, 2096 (2021)
F.R. Tao, H.L. Song, L.J. Chou, RSC Adv. 1, 672 (2011)
T.M.C. Hoang, E.R.H. van Eck, W.P. Bula, J.G.E. Gardeniers, L. Lefferts, K. Seshan, Green Chem. 17, 959 (2015)
G. Tsilomelekis, M.J. Orella, Z.X. Lin, Z.W. Cheng, W.Q. Zheng, V. Nikolakis, D.G. Vlachos, Green Chem. 18, 1983 (2016)
D. Jung, P. Korner, A. Kruse, Biomass Convers. Biorefinery 11, 1155 (2021)
S.S. Xiong, C. Luo, Z.H. Yu, N. Ji, L.J. Zhu, S.R. Wang, Green Chem. 23, 8458 (2021)
Y. Yang, C.W. Hu, M.M. Abu-Omar, Green Chem. 14, 509 (2012)
C.D. Jin, N. Xiang, X. Zhu, E. Shuang, K.C. Sheng, X.M. Zhang, Appl. Catal. B-Environ. 285, 119799 (2021)
P.X. Yan, M. Xia, S.S. Chen, W.Y. Han, H.L. Wang, W.B. Zhu, Green Chem. 22, 5274 (2020)
F. Shen, S. Sun, X. Zhang, J.R. Yang, M. Qiu, X.H. Qi, Cellulose 27, 3013 (2020)
F. Huang, W.Z. Li, T.W. Zhang, D.W. Li, Q.Y. Liu, X.F. Zhu, L.L. Ma, Res. Chem. Intermed. 44, 5439 (2018)
A.K. Das, S. Nandy, S. Bhar, Appl. Organomet. Chem. 35, e6282 (2021)
D. Zeng, Q. Zhang, S. Chen, S. Liu, G. Wang, Microporous Mesoporous Mater. 219, 54 (2016)
M.H. Zong, Z.Q. Duan, W.Y. Lou, T.J. Smith, H. Wu, Green Chem. 9, 434 (2007)
M. Nasrollahzadeh, N.S.S. Bidgoli, N. Shafiei, F. Momenbeik, Int. J. Biol. Macromol. 182, 59 (2021)
G.P. Perez, M.J. Dumont, Chem. Eng. J. 382, 122766 (2020)
R.S. Thombal, V.H. Jadhav, Appl. Catal. A Gen. 499, 213 (2015)
H.Y. Song, C. Xing, B. Li, W.Z. Shen, ChemistrySelect 1, 301 (2016)
M. Kruk, M. Jaroniec, Chem. Mater. 13, 3169 (2001)
A. Ates, Biomass Convers. Biorefinery 13, 9075 (2021)
C.W. Jiang, J.D. Zhu, B. Wang, L. Li, H. Zhong, Chin. J. Chem. Eng. 26, 1270 (2018)
A. Taguchi, F. Schuth, Microporous Mesoporous Mater. 77, 1 (2005)
L. Li, T.I. Koranyi, B.F. Sels, P.P. Pescarmona, Green Chem. 14, 1611 (2012)
F.F. Wang, J. Liu, H. Li, C.L. Liu, R.Z. Yang, W.S. Dong, Green Chem. 17, 2455 (2015)
A. Penkova, L.F. Bobadilla, F. Romero-Sarria, M.A. Centeno, J.A. Odriozola, Appl. Surf. Sci. 317, 241 (2014)
R. Khumho, S. Yousatit, C. Ngamcharussrivichai, Catalysts 11, 887 (2021)
M. Rezaie, M. Dinari, A.N. Chermahini, M. Saraji, A. Shahvar, Int. J. Biol. Macromol. 165, 1129 (2020)
S.H. Dhawane, E.G. Al-Sakkari, G. Halder, Catal. Lett. 149, 3508 (2019)
S.M. Kang, J.X. Fu, G. Zhang, Renew. Sust. Energ. Rev. 94, 340 (2018)
X. Ye, X.Y. Shi, J.Y. Li, B.B. Jin, J. Cheng, Z.H. Ren, H. Zhong, L.W. Chen, X. Liu, F.M. Jin, T.F. Wang, Chem. Eng. J. 440, 135844 (2022)
Y. Su, G.G. Chang, Z.G. Zhang, H.B. Xing, B.G. Su, Q.W. Yang, Q.L. Ren, Y.W. Yang, Z.B. Bao, AIChE J. 62, 4403 (2016)
F. Huang, W.Z. Li, Q.C. Liu, T.W. Zhang, S.X. An, D.W. Li, X.F. Zhu, Fuel Process. Technol. 181, 294 (2018)
F. Guo, Z. Fang, T.J. Zhou, Bioresour. Technol. 112, 313 (2012)
Funding
This work was financially supported by the National Key Research and Development Program of China (2018YFB1501402).
Author information
Authors and Affiliations
Contributions
HG contributed to Investigation, Data curation, Formal analysis, and Writing—original draft. LH contributed to Supervision, Writing—review and editing, Conceptualization, and Funding acquisition. HB contributed to Writing—review and editing. ZW contributed to Investigation, Data curation. JX contributed to Investigation, Data curation. CZ contributed to Data curation. XY contributed to Data curation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no conflict of interests.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, H., Han, L., Baloch, H.A. et al. Conversion of glucose into 5-hydroxymethylfurfural by carbonaceous solid acid catalysts loaded with Brønsted acid and Lewis acid in biphasic system. Res Chem Intermed 49, 4523–4539 (2023). https://doi.org/10.1007/s11164-023-05100-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05100-9